Thermal Energy Chapter 6 Molecules and Motion n






















- Slides: 24

Thermal Energy Chapter 6

Molecules and Motion n The motion of molecules produces heat n The more motion, the more heat is generated

Heat Transfer n The movement of heat from a warmer object to a colder one

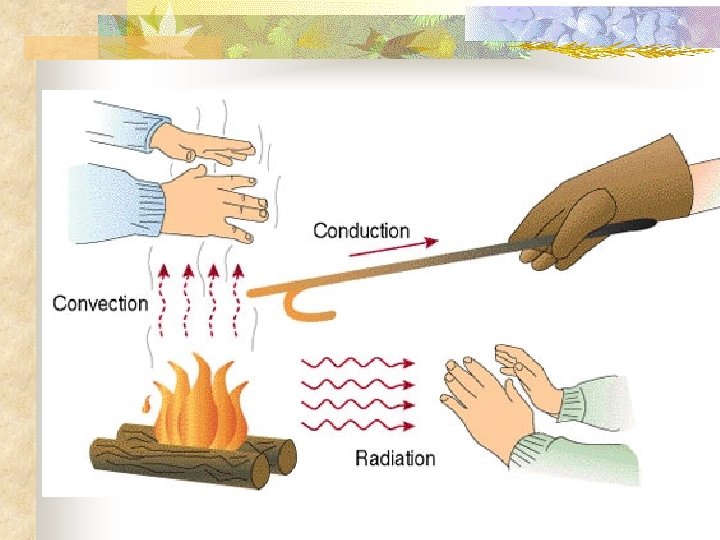
Forms of heat transfer n Three forms of heat transfer: n Conduction n Convection n Radiation


Conduction n Conduction involves the transfer of heat through direct contact n Heat conductors conduct heat well, insulators do not

Convection n n Takes place in liquids and gases as molecules move in currents Heat rises and cold settles to the bottom

Radiation n n Heat is transferred through space Energy from the sun being transferred to the Earth

What type of heat transfer is involved? n n n Heating a room with a fireplace Egg cooking in a frying pan Roof of a house becoming hot

What type of heat transfer? n Warm air mass bringing a change in the weather n Wire getting hot from an electric appliance

Temperature and Heat n Kinetic energy is the energy of motion n Temperature is the measure of the average kinetic energy of an object

Thermometer n n n An instrument used to measure temperature Thermometers commonly have alcohol (with dye) or mercury Digital thermometers have replaced older ones

Celsius Scale Celsius is the metric scale for measuring temperature n Water freezes at 0ºC and boils at 100ºC n

Kelvin scale n n The Kelvin scale is a metric temperature scale measured in Kelvin units (K) Formula (273+ºC)= Kelvin

Absolute zero n The temperature in which all molecular motion stops (0 K)

Calories n Unit for measuring heat n The amount of heat needed to raise 1 gram of water one degree Celsius

Temperature n n Joule is another unit for measuring heat Mass and type of substance determine the amount of temperature change

Specific Heat n n n The ability of a substance to absorb heat energy (specific heat) Different substances absorb heat at different rates The greater the mass of the object the more heat is absorbed

What is a calorimeter? n Device used to measure the heat given off during chemical reactions

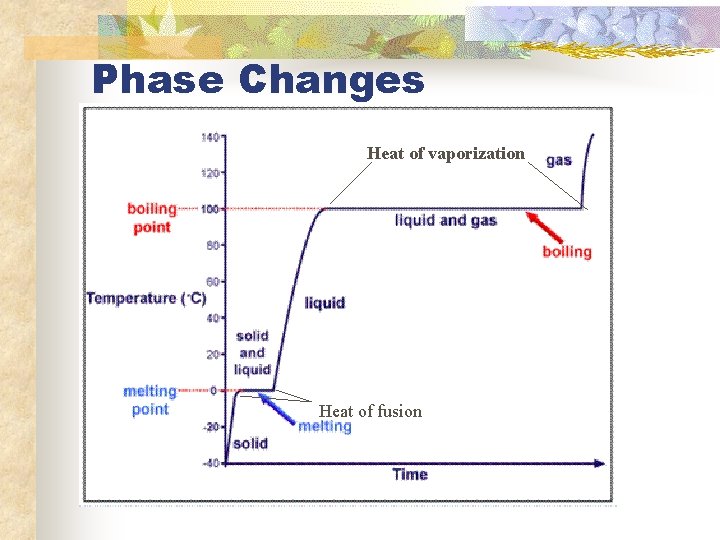
Phase Changes Heat of vaporization Heat of fusion

Heat of Fusion and Vaporization n n Heat of fusion- amount of heat required to change a substance completely from a solid to a liquid Heat of vaporization- amount of heat required to change a substance completely from a liquid to a gas

Heat expansion The expansion of a substance due to heat n Most solids, liquids, and gases expand as they are heated n

Expansion of water n Between 4ºC and 0ºC, water EXPANDS as it cools and turns into a solid

Water has a high specific heat n n 1 cal/ g (degrees C) This is why water stays warm in the winter and cool in the summer