Thermal Decomposition Pathways of Hydroxylamine Theoretical Investigation on



















- Slides: 26

Thermal Decomposition Pathways of Hydroxylamine: Theoretical Investigation on the Initial Steps 作者:Qingsheng Wang, Chunyang Wei, Lisa M. Pérez, William J. Rogers, Michael B. Hall, and M. Sam Mannan* 出處:J. Phys. Chem. A, 2010, 114, 9262 -9269 指導教授:胡維平 教授 報告學生:彭家瑜 報告日期: 2010/10/25 1


Outline Introduction n Computational Details n -Methods -Solvent effect n Results and Discussion -Simple bond dissociations -Unimolecular decomposition pathways of HA -Bimolecular decomposition pathways of HA n Conclusions 3


Introduction Hydroxylamine n 由實驗結果得知,hydroxylamine 在水溶液中分解到最後的產物分別是 NH 3、H 2 O、 N 2、N 2 O 以及少量的 NO 和 H 2,且這個反應大部分研究 都是針對 overall 的反應,缺少反應的 mechanism 。 Papadaki, M. I. ; Pontiki, E. ; Liu, L. ; Rogers, W. J. ; Mannan, M. S. J. Chem. Eng. Data 2009, 54, Cisneros, L. O. ; Wu, X. ; Rogers, W. J. ; Mannan, M. S. ; Park, J. ; North, S. W. Process Saf. Environ. Prot. 2003, 81, 121 n 目前實驗上除了上述那些最終分解出來的氣體,另外還發現了中間產 物 HNO。兩個 hydroxylamine 會發生自身氧化還原反應,產生 H 2 O、 NH 3、HNO,所以作者在此處把兩個 hydroxylamine,也就是雙分子的 分解反應途徑也考慮進去計算。 Nast, R. ; Foppl, I. Z. Anorg. Allg. Chem. 1950, 263, 310 5

Computational Details n Methods Geometry:B 3 LYP、MPW 1 K、BB 1 K、MP 2、CCSD Single-point energies:CCSD(T) n Basis sets cc-p. VDZ → BS 1 6 -31+G(d, p) → BS 2 6 -311+G(3 df, 2 p) → BS 3 n Solvent effects Cluster methods、PCM n Program Gaussian 03 6

Methods (ab initio) n n n Møller-Plesset Purterbation Theory (MPn) Configuration-Interaction Method (CI) Coupled-Cluster Method (CC) Methods (hybrid DFT) n n n Becke 3 -Lee-Yang-Parr (B 3 LYP) Modified Perdew-Wang One-Parameter Model for Kinetics Lynch, B. J. ; Fast, P. L. ; Harris, M. ; Truhlar, D. G. J. Phys. Chem. A, 2000, 104, 4811 (MPW 1 K) Becke 88 -Becke 95 One-Parameter Model for Kinetics Zhao, Y. ; Lynch, B. J. ; Truhlar, D. G. J. Phys. Chem. A, 2004, 108, 2715 (BB 1 K) 7

Solvent Effect n Cluster Method 又稱為 microsolvation,將 solvent 視為獨立的分子,和 solute 一起進行結構最佳化,使用一般量化計算方式去考慮溶劑效應 solvent → solute → ← solvent → 8

Solvent Effect n Polarizable Continuum Model (PCM) 假設 solvent 是一個介質,是連續的,由η(介電常數)表示 ,solute 會受到 solvent 的 dipole 影響。 solute → solvent 9

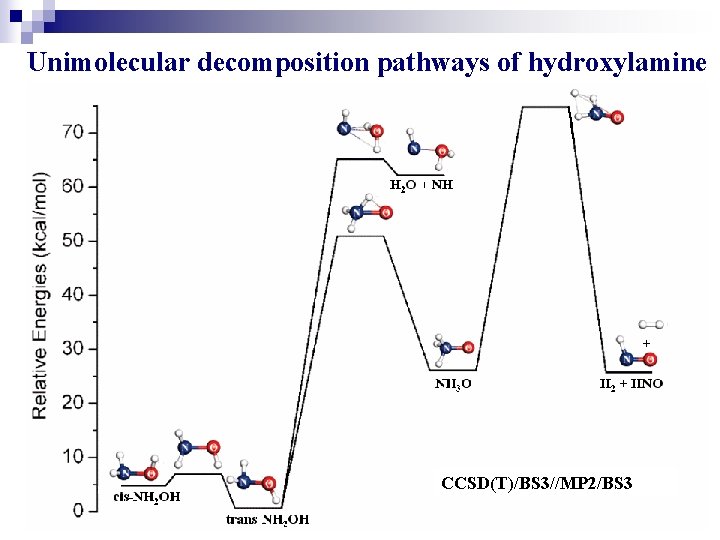
Results and Discussions Hydroxylamine n n Simple bond dissociations Unimolecular decomposition pathways of hydroxylamine cis-NH 2 OH → trans-NH 2 OH → NH 3 O → H 2 + HNO trans-NH 2 OH → H 2 O + NH n Bimolecular decomposition pathways of hydroxylamine 2 NH 2 OH → trans-NH 2 OH + NH 3 O 2 NH 2 OH → cis-NH 2 OH + NH 3 O 10

Results and Discussions Hydroxylamine ※補充 n Bimolecular decomposition pathways of hydroxylamine 2 NH 2 OH → H 2 O + NH 3 + HNO 2 NH 2 OH → NH 2 OH + NH 3 O → HNO + H 2 NH 3 O → NH 2 O· + H· 11

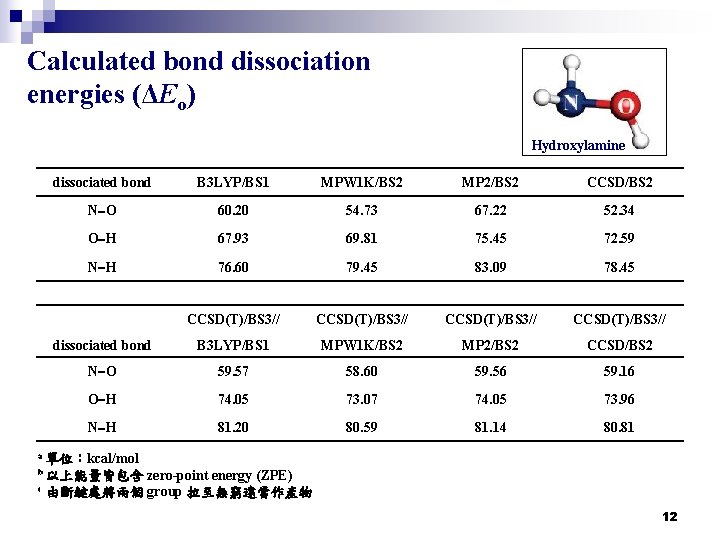
Calculated bond dissociation energies (ΔEo) Hydroxylamine dissociated bond B 3 LYP/BS 1 MPW 1 K/BS 2 MP 2/BS 2 CCSD/BS 2 N-O 60. 20 54. 73 67. 22 52. 34 O-H 67. 93 69. 81 75. 45 72. 59 N-H 76. 60 79. 45 83. 09 78. 45 CCSD(T)/BS 3// dissociated bond B 3 LYP/BS 1 MPW 1 K/BS 2 MP 2/BS 2 CCSD/BS 2 N-O 59. 57 58. 60 59. 56 59. 16 O-H 74. 05 73. 07 74. 05 73. 96 N-H 81. 20 80. 59 81. 14 80. 81 a 單位:kcal/mol zero-point energy (ZPE) c 由斷鍵處將兩個 group 拉至無窮遠當作產物 b 以上能量皆包含 12

Unimolecular decomposition pathways of hydroxylamine CCSD(T)/BS 3//MP 2/BS 3 13

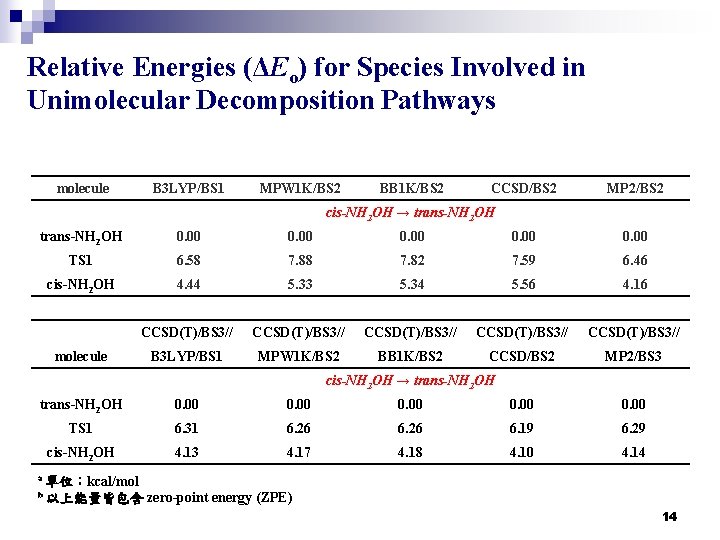
Relative Energies (ΔEo) for Species Involved in Unimolecular Decomposition Pathways molecule B 3 LYP/BS 1 MPW 1 K/BS 2 BB 1 K/BS 2 CCSD/BS 2 MP 2/BS 2 cis-NH 2 OH → trans-NH 2 OH 0. 00 TS 1 6. 58 7. 82 7. 59 6. 46 cis-NH 2 OH 4. 44 5. 33 5. 34 5. 56 4. 16 CCSD(T)/BS 3// CCSD(T)/BS 3// B 3 LYP/BS 1 MPW 1 K/BS 2 BB 1 K/BS 2 CCSD/BS 2 MP 2/BS 3 molecule cis-NH 2 OH → trans-NH 2 OH 0. 00 TS 1 6. 31 6. 26 6. 19 6. 29 cis-NH 2 OH 4. 13 4. 17 4. 18 4. 10 4. 14 a 單位:kcal/mol b 以上能量皆包含 zero-point energy (ZPE) 14

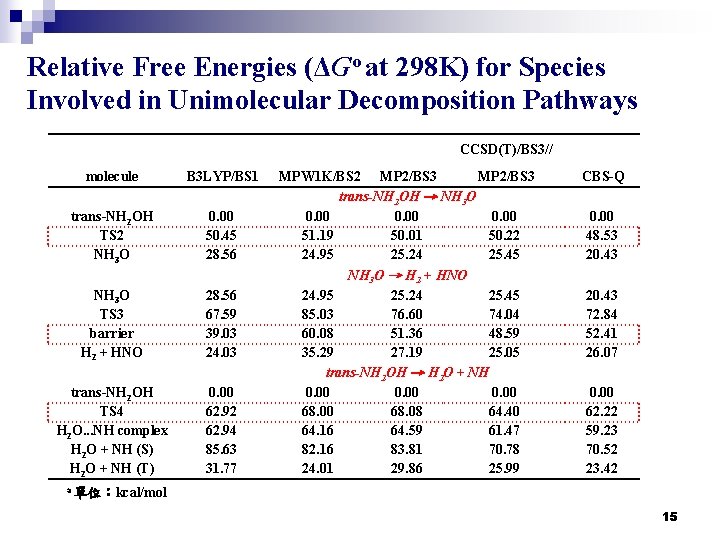
Relative Free Energies (ΔGo at 298 K) for Species Involved in Unimolecular Decomposition Pathways CCSD(T)/BS 3// molecule B 3 LYP/BS 1 trans-NH 2 OH TS 2 NH 3 O 0. 00 50. 45 28. 56 NH 3 O TS 3 barrier H 2 + HNO 28. 56 67. 59 39. 03 24. 03 trans-NH 2 OH TS 4 H 2 O. . . NH complex H 2 O + NH (S) H 2 O + NH (T) 0. 00 62. 92 62. 94 85. 63 31. 77 MPW 1 K/BS 2 MP 2/BS 3 trans-NH 2 OH → NH 3 O 0. 00 51. 19 50. 01 50. 22 24. 95 25. 24 25. 45 NH 3 O → H 2 + HNO 24. 95 25. 24 25. 45 85. 03 76. 60 74. 04 60. 08 51. 36 48. 59 35. 29 27. 19 25. 05 trans-NH 2 OH → H 2 O + NH 0. 00 68. 08 64. 40 64. 16 64. 59 61. 47 82. 16 83. 81 70. 78 24. 01 29. 86 25. 99 CBS-Q 0. 00 48. 53 20. 43 72. 84 52. 41 26. 07 0. 00 62. 22 59. 23 70. 52 23. 42 a 單位:kcal/mol 15

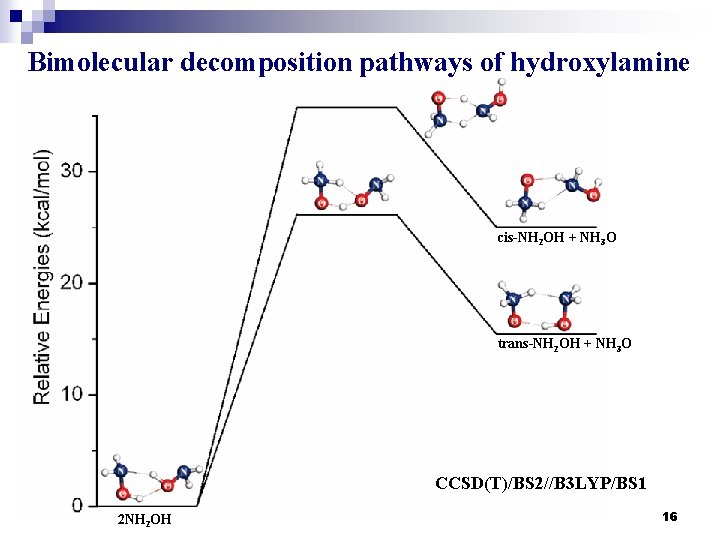
Bimolecular decomposition pathways of hydroxylamine cis-NH 2 OH + NH 3 O trans-NH 2 OH + NH 3 O CCSD(T)/BS 2//B 3 LYP/BS 1 2 NH 2 OH 16

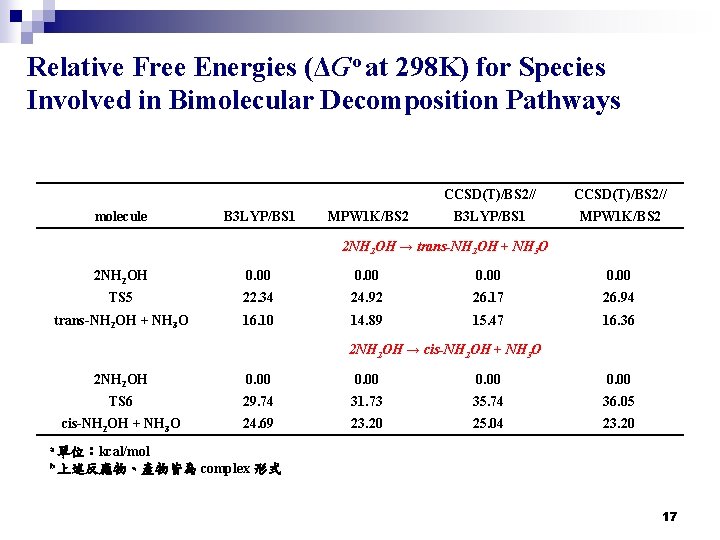
Relative Free Energies (ΔGo at 298 K) for Species Involved in Bimolecular Decomposition Pathways molecule B 3 LYP/BS 1 MPW 1 K/BS 2 CCSD(T)/BS 2// B 3 LYP/BS 1 MPW 1 K/BS 2 2 NH 2 OH → trans-NH 2 OH + NH 3 O 2 NH 2 OH 0. 00 TS 5 22. 34 24. 92 26. 17 26. 94 trans-NH 2 OH + NH 3 O 16. 10 14. 89 15. 47 16. 36 2 NH 2 OH → cis-NH 2 OH + NH 3 O 2 NH 2 OH 0. 00 TS 6 29. 74 31. 73 35. 74 36. 05 cis-NH 2 OH + NH 3 O 24. 69 23. 20 25. 04 23. 20 a 單位:kcal/mol b 上述反應物、產物皆為 complex 形式 17

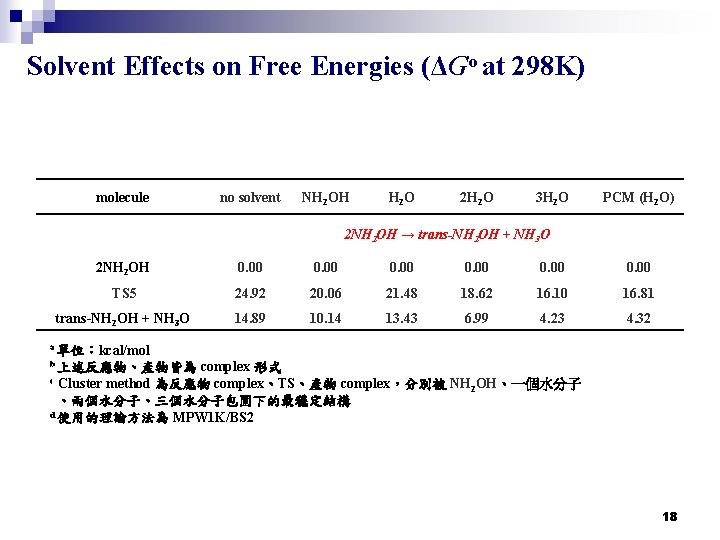
Solvent Effects on Free Energies (ΔGo at 298 K) molecule no solvent NH 2 OH H 2 O 2 H 2 O 3 H 2 O PCM (H 2 O) 2 NH 2 OH → trans-NH 2 OH + NH 3 O 2 NH 2 OH 0. 00 TS 5 24. 92 20. 06 21. 48 18. 62 16. 10 16. 81 trans-NH 2 OH + NH 3 O 14. 89 10. 14 13. 43 6. 99 4. 23 4. 32 a 單位:kcal/mol complex 形式 Cluster method 為反應物 complex、TS、產物 complex,分別被 NH 2 OH、一個水分子 、兩個水分子、三個水分子包圍下的最穩定結構 d 使用的理論方法為 MPW 1 K/BS 2 b 上述反應物、產物皆為 c 18

Solvent Effects (Cluster Method) 2 NH 2 OH TS MPW 1 K/BS 2 n. N n. O NH 2 OH + NH 3 O 19

Solvent Effects (Cluster Method) 2 NH 2 OH TS MPW 1 K/BS 2 n. N n. O NH 2 OH + NH 3 O 20


22


Methods (ab initio) n Møller-Plesset Purterbation Theory (MPn) n Configuration-Interaction Method (CI) 24

Methods (ab initio) n Coupled-Cluster Method (CC) 25

Methods (hybrid DFT) n Becke 3 -Lee-Yang-Parr (B 3 LYP) n Modified Perdew-Wang One-Parameter Model for Lynch, B. J. ; Fast, P. L. ; Harris, M. ; Truhlar, D. G. J. Phys. Chem. A, 2000, 104, 4811 Kinetics (MPW 1 K) n Becke 88 -Becke 95 One-Parameter Model for Kinetics Zhao, Y. ; Lynch, B. J. ; Truhlar, D. G. J. Phys. Chem. A, 2004, 108, 2715 (BB 1 K) 26