Therapeutic Uses of Botulinum Toxin in Neuroophthalmology Janette

Therapeutic Uses of Botulinum Toxin in Neuro-ophthalmology Janette I. Lindley, MD FRCSC St. Paul’s Hospital University of British Columbia

Overview 1. On label uses: n hemifacial spasm n blepharospasm injection techniques n other: cervical dystonia strabismus wrinkles (glabella)

Overview 2. Off label uses: n headache (chronic daily) Phase II Trial Results injection technique n crocodile tears, protective ptosis n other: writer’s cramp, hyperhydrosis, head tremor, focal spasticity, drooling

Botulinum Toxin in Neuro-ophthalmology neurotransmission inhibition (ACH, other) at NMJ n chemical denervation striated muscle peaks @ 2 weeks n Neuronal sprouting heralds return of function @ 3 – 6 mos. n

Botulinum Toxin n Serotype A (Botox , Dysport) n Serotype B (Myobloc)

Hemifacial spasm Unilateral n Periocular and lower facial +/-platysma stapedius (clicking at hs) n R/O facial nerve compression (MRI) n

Hemifacial spasm movie Click here

Blepharospasm tonic/ clonic lid closure n may present unilaterally n uncontrollable n functional cause for visual loss n (apraxia of lid opening) n

Blepharospasm movie (Click here)
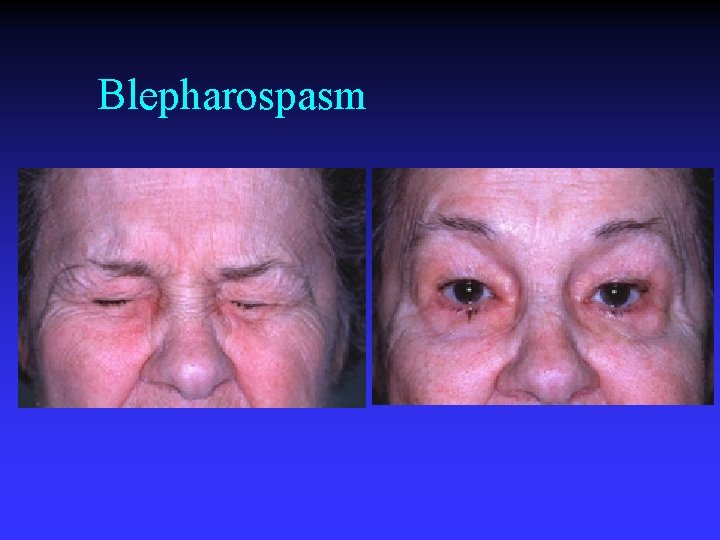
Blepharospasm

Pathological Pain Inhibition observed (Binder et al) after Rx hyperfunctional facial lines n inhibition of neuromuscular activity and n substance P, glutamate, & calcitonin peptide release n results in analgesic effect n

Headache Disorders n n n heterogeneous group of conditions recent results Phase II trials in chronic daily headache (CDH)/ transformed migraine randomized, double blind placebo controlled 75% completion at 11 mos.

CDH or Transformed Migraine HA 15 d/m > 1(3)m n each HA 4 h/d n no primary cause n H/O episodic migraine (>50% pr migr) n 4% of pop ~1. 2 -1. 5 million in Canada n significant disability/resource use n

Chronic Daily Headache Studies -Common Design Primary analysis Placebo Non. Responder (PNR) Baseline -30 270 *Allergan, Botox®, USA Bo. NTA* Placebo Placebo Responder (pr) -60 Final analysis Placebo 0 Day 90 180 14
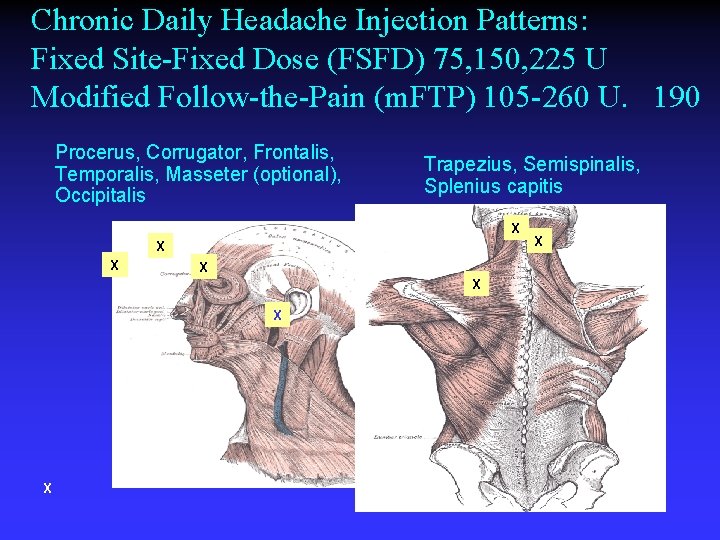
Chronic Daily Headache Injection Patterns: Fixed Site-Fixed Dose (FSFD) 75, 150, 225 U Modified Follow-the-Pain (m. FTP) 105 -260 U. 190 Procerus, Corrugator, Frontalis, Temporalis, Masseter (optional), Occipitalis Trapezius, Semispinalis, Splenius capitis X X X X
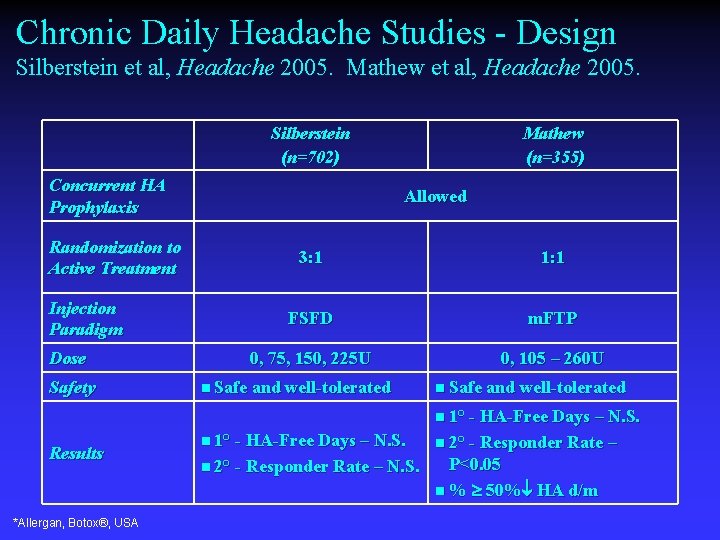
Chronic Daily Headache Studies - Design Silberstein et al, Headache 2005. Mathew et al, Headache 2005. Silberstein (n=702) Concurrent HA Prophylaxis Randomization to Active Treatment Injection Paradigm Dose Safety Mathew (n=355) Allowed 3: 1 1: 1 FSFD m. FTP 0, 75, 150, 225 U 0, 105 – 260 U n Safe and well-tolerated n 1° - HA-Free Days – N. S. Results *Allergan, Botox®, USA n 1° - HA-Free Days – N. S. n 2° - Responder Rate – N. S. P<0. 05 n % 50% HA d/m
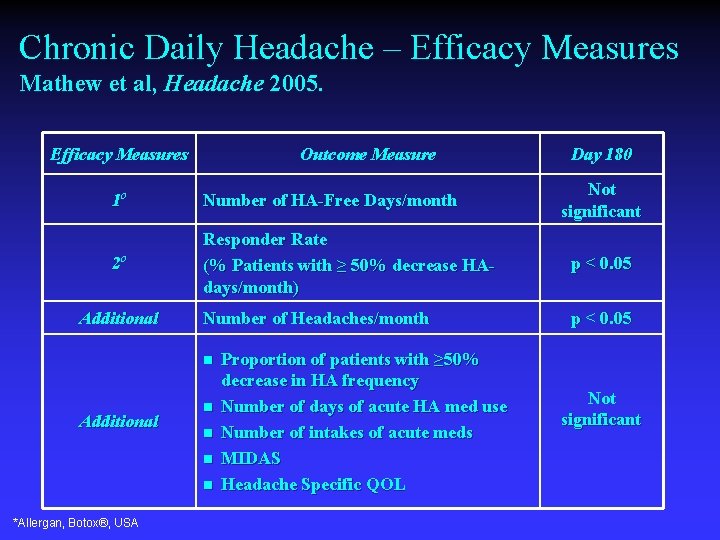
Chronic Daily Headache – Efficacy Measures Mathew et al, Headache 2005. Efficacy Measures Outcome Measure Day 180 Not significant 1 o Number of HA-Free Days/month 2 o Responder Rate (% Patients with ≥ 50% decrease HAdays/month) p < 0. 05 Number of Headaches/month p < 0. 05 Additional n n n n *Allergan, Botox®, USA Proportion of patients with ≥ 50% decrease in HA frequency Number of days of acute HA med use Number of intakes of acute meds MIDAS Headache Specific QOL Not significant
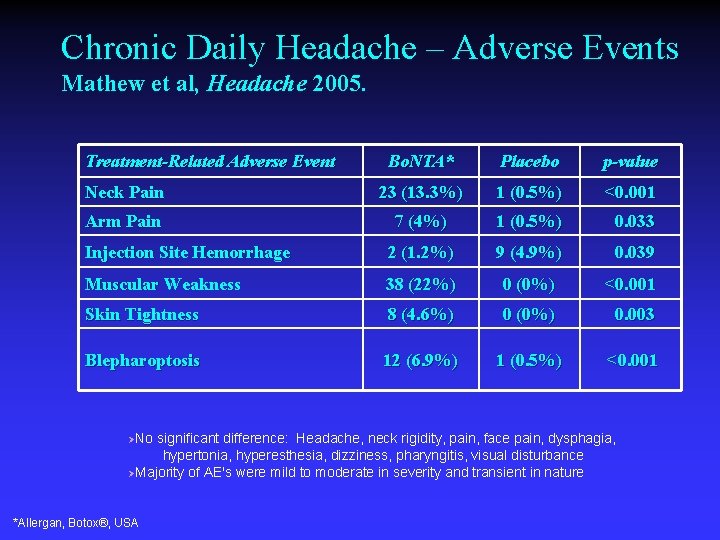
Chronic Daily Headache – Adverse Events Mathew et al, Headache 2005. Treatment-Related Adverse Event Bo. NTA* Placebo p-value Neck Pain 23 (13. 3%) 1 (0. 5%) <0. 001 Arm Pain 7 (4%) 1 (0. 5%) 0. 033 Injection Site Hemorrhage 2 (1. 2%) 9 (4. 9%) 0. 039 Muscular Weakness 38 (22%) 0 (0%) <0. 001 Skin Tightness 8 (4. 6%) 0 (0%) 0. 003 Blepharoptosis 12 (6. 9%) 1 (0. 5%) <0. 001 ØNo significant difference: Headache, neck rigidity, pain, face pain, dysphagia, hypertonia, hyperesthesia, dizziness, pharyngitis, visual disturbance ØMajority of AE's were mild to moderate in severity and transient in nature *Allergan, Botox®, USA

Chronic Daily Headache – Safety & Results Mathew et al, Headache 2005. Repeat treatment (up to 3 treatment cycles) with Bo. NTA* is safe and well-tolerated at doses up to 260 U n No neutralizing antibodies n No benefit of placebo run-in pool PNR and PR groups n Although the 1° endpoint was not met, significant & clinically meaningful improvements were seen following Bo. NTA* vs placebo: u Responder rates u Headache frequency n No significant change in proportion of patients with ≥ 50% Decrease in HA Frequency, Number of Days of Acute HA Med Use, Number of Uses of Acute Meds, MIDAS, Headache Specific QOL n *Allergan, Botox®, USA 19

CDH – 1° Outcome Measure Mathew et al, Headache 2005. Number of Headache-Free Days PNR Bo. NTA* (n=134) PNR PBO (n=145) PR Bo. NTA* (n=39) PR PBO (n=37) Δ = 1. 5 HA-free days at Day 180 Blinded Treatment Days After Placebo Run - In *Allergan, Botox®, USA 20

CDH – 2° Outcome Measure Mathew et al, Headache 2005. Responder Rate % Patients with > 50% Decrease Headache Days *p<0. 027 Blinded Treatment * 33 15 Days After Placebo Run - In *Allergan, Botox®, USA 21

CDH – Number of HA’s Pooled Mathew et al, Headache 2005. (PNR + pr) Number of Headaches – Change from Baseline *p<0. 05 § p=0. 001 * * * Blinded Treatment 3. 4 * § Baseline * * * Bo. NTA* = 13. 5 Placebo = 12. 7 Days After Placebo Run - In *Allergan, Botox®, USA 22

CDH – % Decrease HA Frequency Dodick et al, Headache 2005. Subgroup Analysis - No Concomitant Prophylaxis % Decrease in Number of Headaches ≥ 30% ≥ 50% *p<0. 05 * * *p<0. 05 * Days After Placebo Run - In Bo. NTA* *Allergan, Botox®, USA * Days After Placebo Run - In Placebo Blinded Treatment 23

Chronic Daily Headache m. FTP n HA free days - NS n 50% in HA d/m - S n #HA/mo - S n
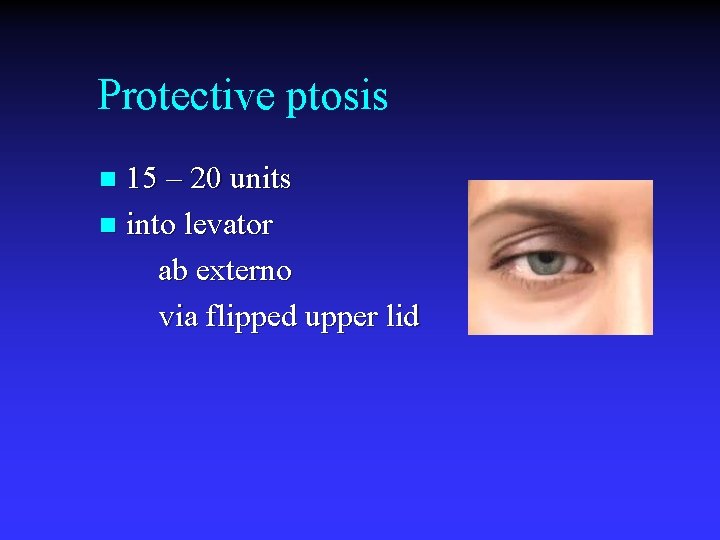
Protective ptosis 15 – 20 units n into levator ab externo via flipped upper lid n

Autonomic Nerve Inhibition – Ach Release Blocked glands n lumen post injection (Swartling) n smooth muscle n
Injection of BTX-A

Conclusions n Onset of effect occurred within 2 – 3 days following injection and lasted for 3 - 4 months. n Side effects are infrequent, mild and transient. n Subjective and objective evidence for reduction in tear production. n Effectiveness needs to be established with a randomised clinical trial.

Thanks to: SPH Staff: Cynchia, Maureen, Kathy Residents: Leah, Paul, Briar Allergan: Botox Therapeutic Div G. Davidovic D. Hoppenbrouwer
- Slides: 29