THERAPEUTIC AGENTS 1 Recombinant Proteins 2 Nucleic Acids

THERAPEUTIC AGENTS 1. Recombinant Proteins 2. Nucleic Acids 1
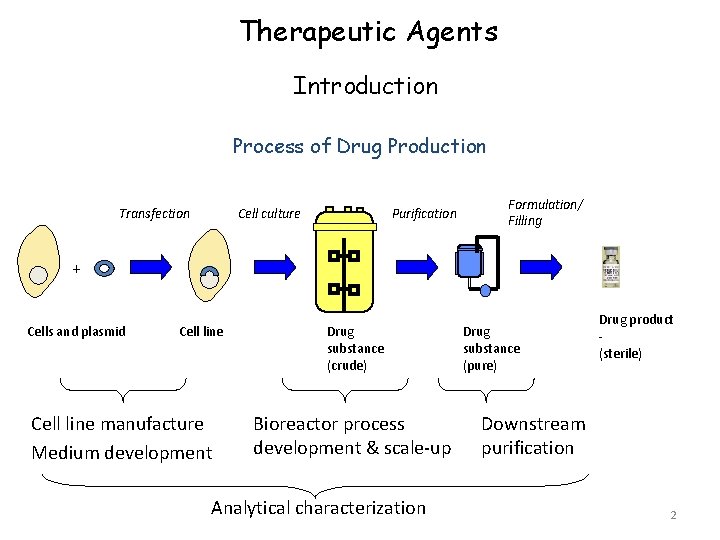
Therapeutic Agents Introduction Process of Drug Production Transfection Cell culture Purification Formulation/ Filling + Cells and plasmid Cell line manufacture Medium development Drug substance (crude) Bioreactor process development & scale-up Analytical characterization Drug substance (pure) Drug product (sterile) Downstream purification 2
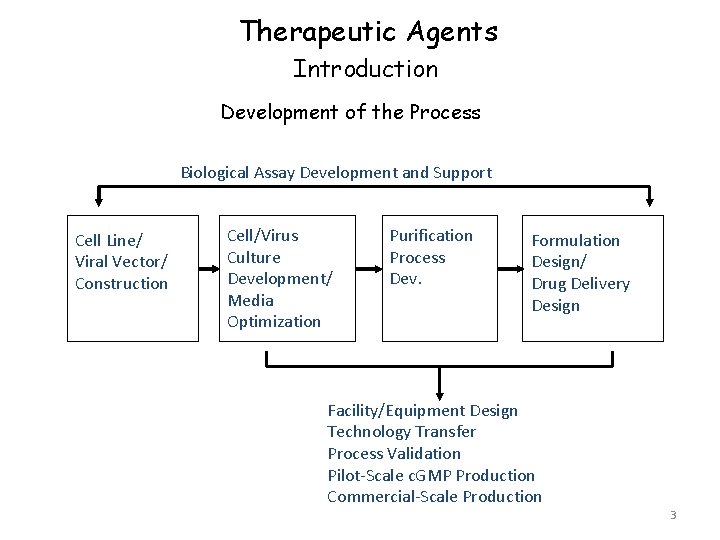
Therapeutic Agents Introduction Development of the Process Biological Assay Development and Support Cell Line/ Viral Vector/ Construction Cell/Virus Culture Development/ Media Optimization Purification Process Dev. Formulation Design/ Drug Delivery Design Facility/Equipment Design Technology Transfer Process Validation Pilot-Scale c. GMP Production Commercial-Scale Production 3

PRODUCTION OF THERAPEUTIC PROTEINS OBSTACLES IN PRODUCTION: • Protein solubility, distribution, stability • Delivery route (blood or digestive juice may degrade it) • Physiologically active-post translational modifications • Cost

SOURCES Various types of cells like • bacterial cells, • yeast cells, • insect cells, • microbes (Bacillus sp. , Actinomycetes sp. , E coli), • plants, • animals, • also mammalian cells are used for producing therapeutic proteins under defined conditions.
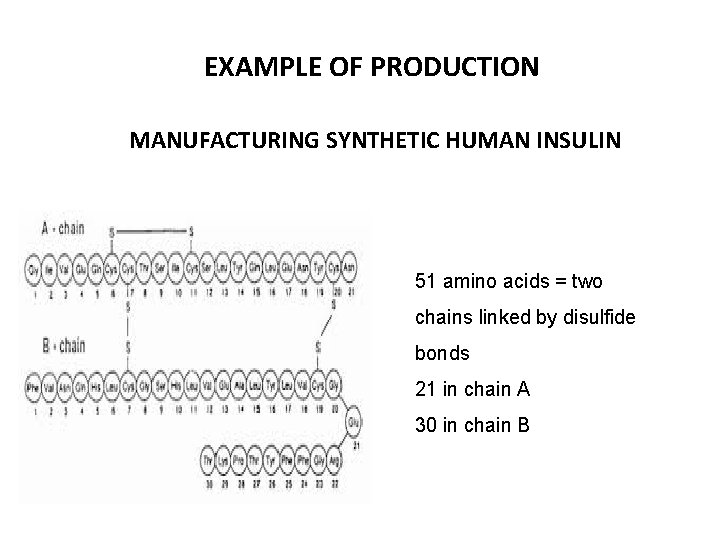
EXAMPLE OF PRODUCTION MANUFACTURING SYNTHETIC HUMAN INSULIN 51 amino acids = two chains linked by disulfide bonds 21 in chain A 30 in chain B
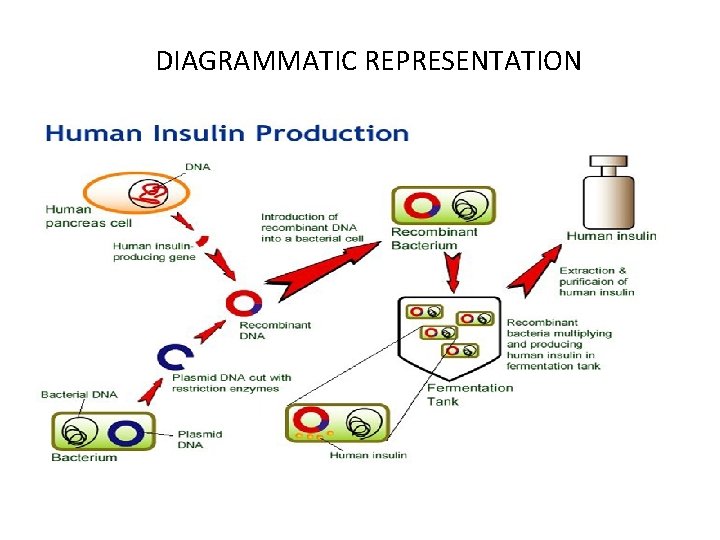
DIAGRAMMATIC REPRESENTATION

Summary of Steps • • Cell fractionation - yield 1 ug of protein, which can be obtained in a pure form by electrophoresis. The protein is analyzed to yield the identity of the first 30 amino acids - its amino terminus. The genetic code is used to predict the nucleotide sequence corresponding to this amino acid sequence, probes synthesized. Probes used to identify the complementary m. RNA from which c. DNA is produced using reverse transcriptase. Cloning produces large amounts of c. DNA produced from each clone is hybridized to total cellular m. RNA. Purified m. RNA corresponding to each cloned c. DNA sequence is then produced. Having checked by cell-free protein synthesis procedures which m. RNA codes for the desired protein, the appropriate c. DNA is then sequenced and used to determine the protein's amino acid sequence. Finally, the cloned c. DNA is incorporated into a plasmid vector and transferred to bacterial or yeast cells. This is the starting point for scaled-up production of large amounts of the purified protein. 8

Therapeutic Agents Recombinant Proteins Human Interferons -> to fight viral infections -> Scientists discovered an antiviral protein in 1957 that inhibited growth of influenza virus in chicken embryos. It was named interferon because it interfered with the growth of influenza virus. • Anti viral proteins released by host cells (part of the immune system) • Interfere with viral multiplication • Host cell specific but not virus specific • Different types of cells in animals produce different interferons 9

Therapeutic Agents Recombinant Proteins Human Interferons -> to fight viral infections • 3 types of human interferon: – alpha interferon (13 genes) – beta interferon (2 genes) – gamma interferon (1 gene) • Alpha & beta usually produced early in viral infections (viruses or viral RNA) - Gamma appears later -> Presence of double-stranded RNA indicates cell is infected -> Viral infected cells release alpha and beta interferons Diffuse to neighboring cells -> Virus can’t replicate 10

Therapeutic Agents Recombinant Proteins Human Interferons -> to fight viral infections Antiviral Treatment: • Interferon therapy – Limited lifetime, short lasting effect – Recombinant interferons • Pure and fast • Hybrid genes for enhanced/new activity – Oral administration 11

Therapeutic Agents Recombinant Proteins Human Interferons -> to fight viral infections Why are Side Effects Common and Severe for Injectable Interferon? • • • Injectable interferon (beta) is approved world-wide (FDA) for the treatment of various cancers and viral diseases. Interferon is a protein readily eliminated from the blood by the kidney. To counteract the kidney’s clearance of interferon from the blood injectable interferon must be given in doses much higher than what occur naturally. Side effects include flu-like symptoms, poor results on liver function tests, and blood cell abnormalities. More serious side effects include depression, epileptic seizures, or liver problems. 12

Why is Oral Interferon Different? • • Low-dose oral interferon is given in doses 10 thousand times less than injectable interferon. Therefore, side effects are dramatically reduced. Oral interferon is human interferon alpha administered in a small tablet (lozenge) to humans or in powder to animals. Oral interferon binds to surface (mucosal) cells in the mouth and throat resulting in stimulation of white blood cells and activates hundreds of genes affecting the immune system in the peripheral blood of man, cattle and mice. Studies show oral interferon is effective against disorders such as cancer, viral diseases and autoimmunity. 13
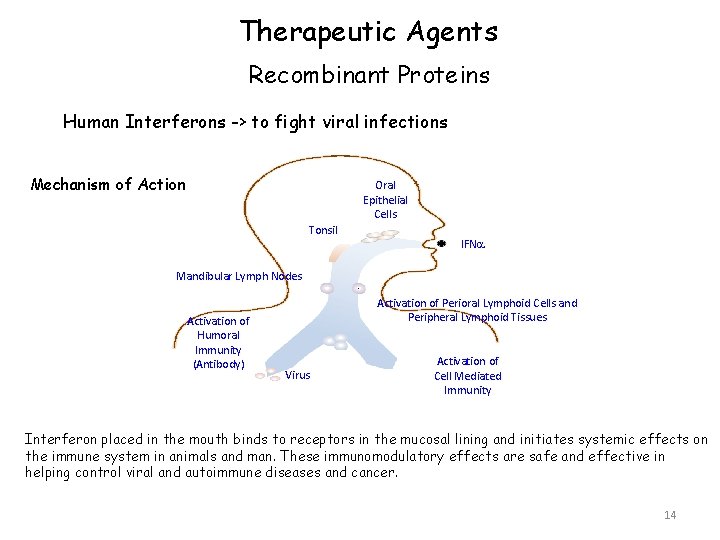
Therapeutic Agents Recombinant Proteins Human Interferons -> to fight viral infections Mechanism of Action Oral Epithelial Cells Tonsil IFNa Mandibular Lymph Nodes Activation of Humoral Immunity (Antibody) Activation of Perioral Lymphoid Cells and Peripheral Lymphoid Tissues Virus Activation of Cell Mediated Immunity Interferon placed in the mouth binds to receptors in the mucosal lining and initiates systemic effects on the immune system in animals and man. These immunomodulatory effects are safe and effective in helping control viral and autoimmune diseases and cancer. 14
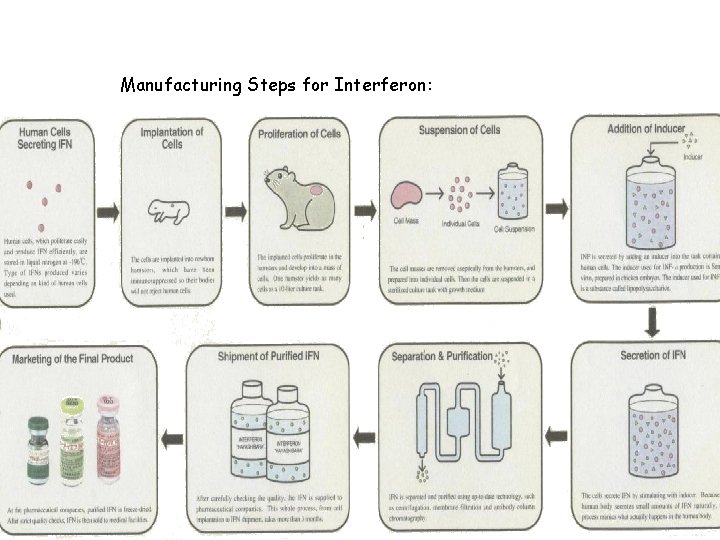
Manufacturing Steps for Interferon: 15

Therapeutic Agents Recombinant Proteins Human Interferons -> to fight viral infections Human diseases in which oral interferon has been tested and reported to be safe 16
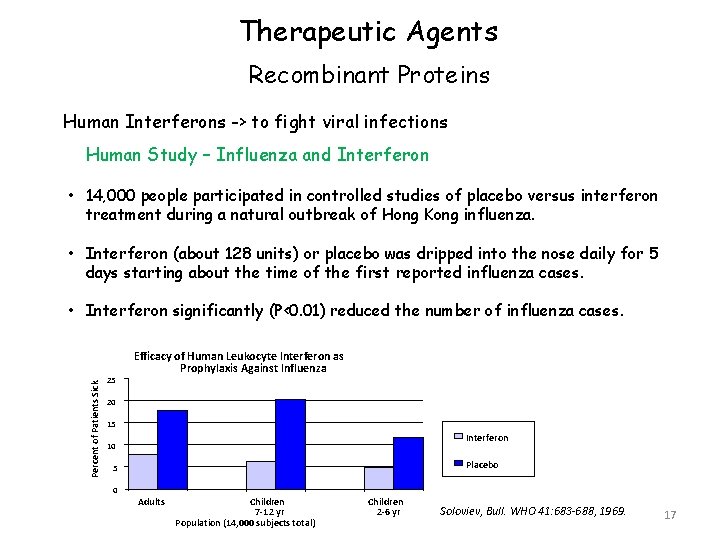
Therapeutic Agents Recombinant Proteins Human Interferons -> to fight viral infections Human Study – Influenza and Interferon • 14, 000 people participated in controlled studies of placebo versus interferon treatment during a natural outbreak of Hong Kong influenza. • Interferon (about 128 units) or placebo was dripped into the nose daily for 5 days starting about the time of the first reported influenza cases. Percent of Patients Sick • Interferon significantly (P<0. 01) reduced the number of influenza cases. 25 Efficacy of Human Leukocyte Interferon as Prophylaxis Against Influenza 20 15 Interferon 10 Placebo 5 0 Adults Children 7 -12 yr Population (14, 000 subjects total) Children 2 -6 yr Soloviev, Bull. WHO 41: 683 -688, 1969. 17

Therapeutic Agents Recombinant Proteins Strategies for Optimisation of Recombinant Production • Screening libraries of recombinant genes (IFNs, human growth hormone, TNF-a…) • Screening of recombinant expression systems (E coli, fungi, Mammalian cells…) • Delivery by intestinal bacteria (lactobacilli) 18

Principles of Drug Delivery

Drug Delivery • Definition – The appropriate administration of drugs through various routes in the body for the purpose of improving health – It is highly interdisciplinary – It is not a young field – It has recently evolved to take into consideration • • Drug physico-chemical properties Body effects and interactions Improvement of drug effect Patient comfort and well being Controlled Drug Delivery
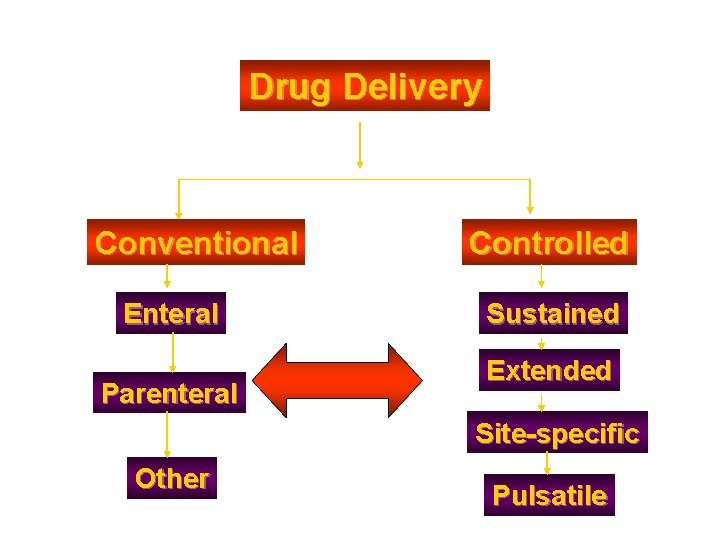
Drug Delivery Conventional Enteral Parenteral Controlled Sustained Extended Site-specific Other Pulsatile

Oral Administration • Advantages – Patient: Convenience, not invasive, higher compliance – Manufacture: well established processes, available infrastructure • Disadvantages – Unconscious patients cannot take dose – Low solubility – Low permeability – Degradation by GI enzymes or flora – First pass metabolism – Food interactions – Irregular absorption

Oral Administration • Traditional oral delivery systems – – – Tablets Capsules Soft gelatin capsules Suspensions Elixirs
Buccal/Sublingual • Advantages – By-pass First pass metabolism – Rapid absorption – Low enzymatic activity • Disadvantages – Discomfort during dissolution – Probability of swallowing- lost of effect – Small doses • Traditional delivery system/devices – Tablets – Chewing gum
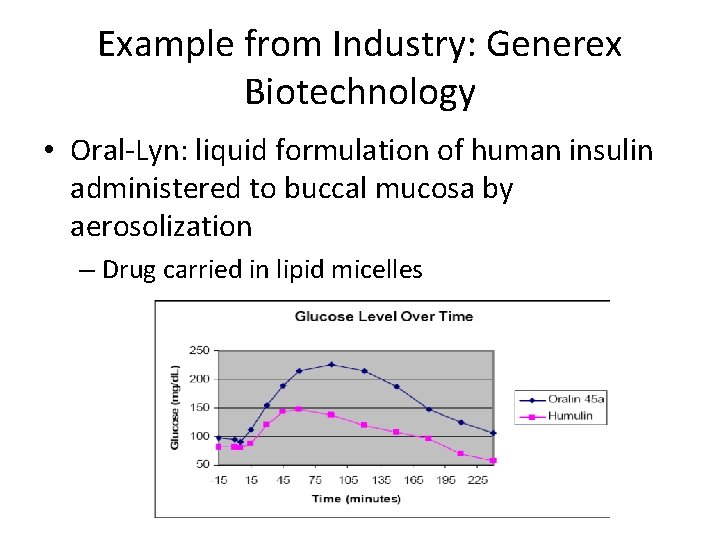
Example from Industry: Generex Biotechnology • Oral-Lyn: liquid formulation of human insulin administered to buccal mucosa by aerosolization – Drug carried in lipid micelles

Rectal • Advantages – By-pass first pass metabolism – Useful for children • Disadvantages – Absorption depends on disease state – Degradation by bacterial flora – Uncomfortable • Traditional delivery system/devices – Suppository – Enema
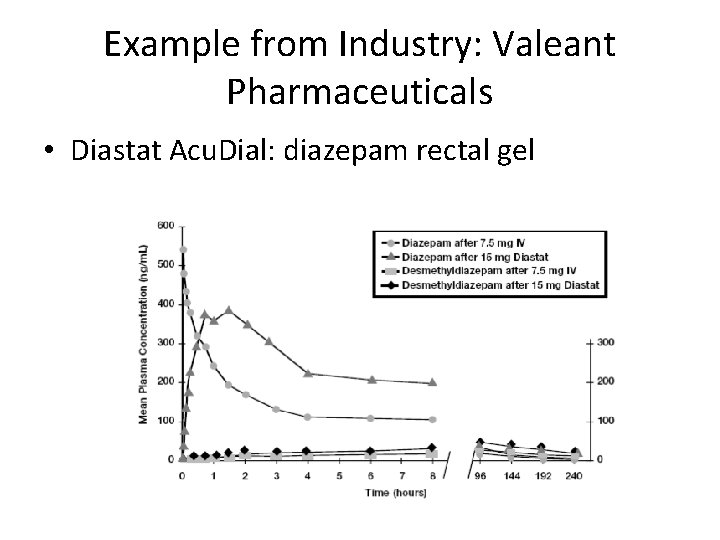
Example from Industry: Valeant Pharmaceuticals • Diastat Acu. Dial: diazepam rectal gel
Intravenous (IV) • Advantages – Drug 100% bioavailable – Rapid response – Total control of blood concentration – Maximize incorporation of degradable drugs – By-pass FPM • Disadvantages – Invasive – Trained personnel – Possible toxicity due to incorrect dosing – sterility • Traditional delivery system/devices – Injection-bolus – IV bag - infusion

Subcutaneous (SC) • Advantages – Patient selfadministration – Slow, complete absorption – By-pass FPM • Disadvantages – Invasive – Irritation, inflammation – Maximum dose volume - 2 m. L

Intramuscular (IM) • Advantages – Patient can administer the drug himself – Larger volume than subcutaneous – By-pass first pass metabolism • Disadvantages – Invasive – patient disconfort – Irritation, inflamation – May require some training

Inhalers • Advantages – By-pass FPM – Gases are rapidly absorbed • Disadvantages – Solids and liquids can be absorbed if size is below 0. 5 um
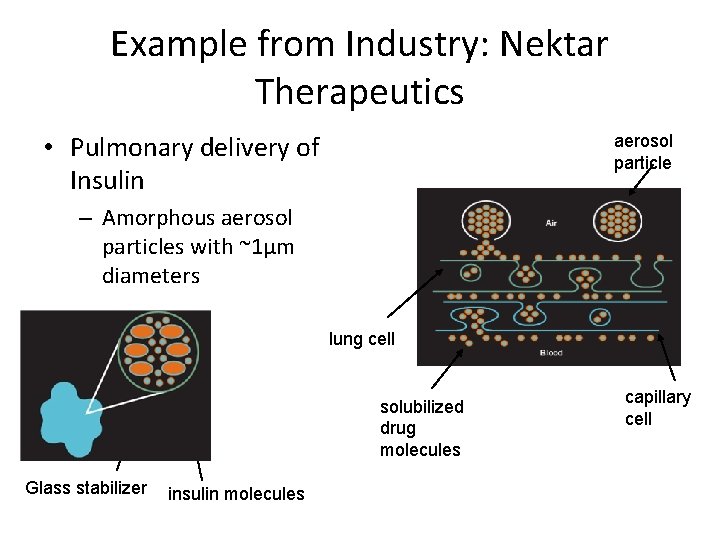
Example from Industry: Nektar Therapeutics • Pulmonary delivery of Insulin aerosol particle – Amorphous aerosol particles with ~1μm diameters lung cell solubilized drug molecules Glass stabilizer insulin molecules capillary cell

Transdermal • Advantages – Local effect – Ease of administration • Disadvantages – Low absorption for some drugs – May cause allergic reactions • Requirements – Low dosage <10 mg/m. L – MW< 1, 000
Factors Influencing the Selection of the Delivery Route • Drug physico-chemical properties – Drug molecular size (molecular weight) – Half-life – Chemical stability – Loss of biological activity in aqueous solution • Proteins – Denaturation, degradation

Example from Industry: 3 M Company • Microstructured Transdermal System: MTS – Microneedle system – Drug-in-adhesive technology platform (a) 3 M microneedle system and (b) histological section of microneedles in guinea pig skin

Factors Influencing the Selection of the Delivery Route – Solubility in aqueous solution (hydrophobicity/hydrophilicity) • • p. H p. Ka - ionization Temperature Concentration Crystalinity Particle size State of hydration

Factors Influencing the Selection of the Delivery Route • Drug biological interactions – Sensitive to FPM – Low membrane permeabiltiy • Efflux pumps (MRP, MDR) – cancer drugs • Hydrophilicity • High-density charge – – Enzymatic degradation Bacterial degradation Half-life Side effects • Irritation

Factors Influencing the Selection of the Delivery Route • Desired pharmacological effect – Local • topical, vaginal – Systemic • oral, buccal, IV, SC, IM, rectal, nasal – Immediate response • IV, SC, IM, nasal – Dose size – Drug molecular size

Manufacture of Classical Oral Delivery Systems • Formulation – combination of active ingredients with the appropriate excipients • Excipients – inactive ingredients employed for the purpose of dilution, protection, stability, controlled release, taste, fillers, coloring, disintegration, etc
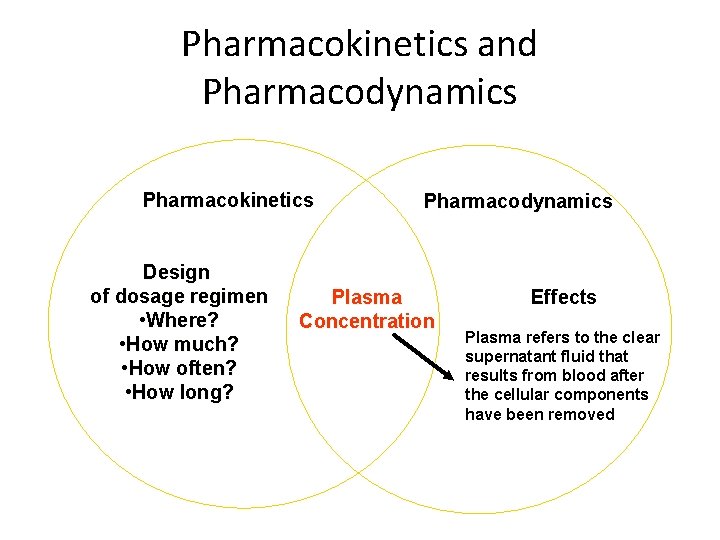
Pharmacokinetics and Pharmacodynamics Pharmacokinetics Design of dosage regimen • Where? • How much? • How often? • How long? Pharmacodynamics Plasma Concentration Effects Plasma refers to the clear supernatant fluid that results from blood after the cellular components have been removed
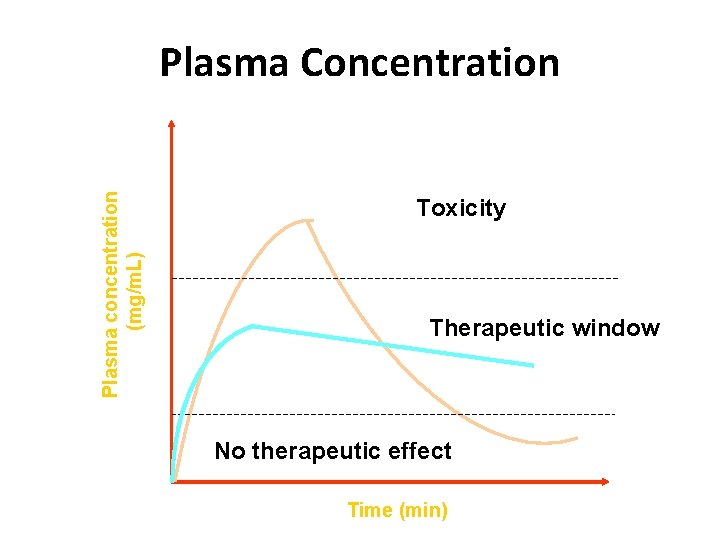
Plasma concentration (mg/m. L) Plasma Concentration Toxicity Therapeutic window No therapeutic effect Time (min)
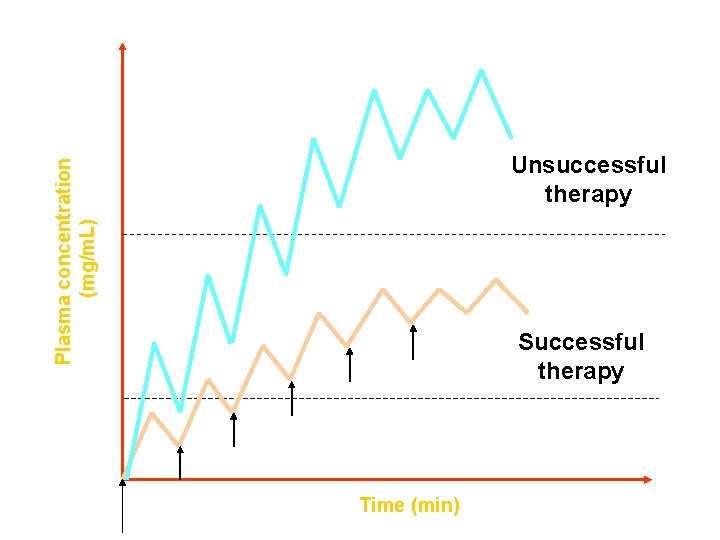
Plasma concentration (mg/m. L) Unsuccessful therapy Successful therapy Time (min)

Magnitude of Drug Response • Depends upon concentration achieved at the site of action – Dosage – Extent of absorption – Distribution to the site – Rate/extent of elimination
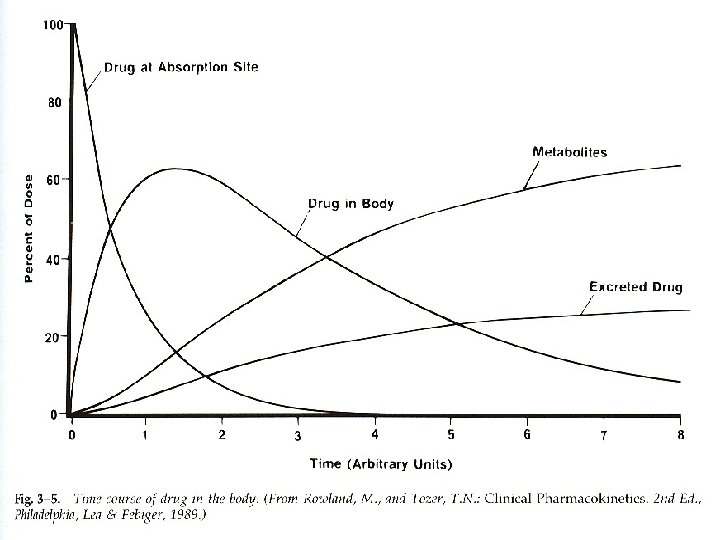

From the Site of Delivery to Elimination… steps in drug delivery, absorption, distribution and elimination • Drug Delivery – Selection of drug delivery route • Knowledge of physicochemical properties – Design of dosing regimen • Absorption – Knowledge of PK and PD • First pass effect • MDR or MRP

From the Site of Delivery to Elimination… steps in drug delivery, absorption, distribution and elimination • Distribution – Drugs must reach the site of action • Tissue • Plasma Depends upon drug binding capabilities • Elimination • Metabolism – Liver, kidneys, cells • Excretion – Kidneys – Feces
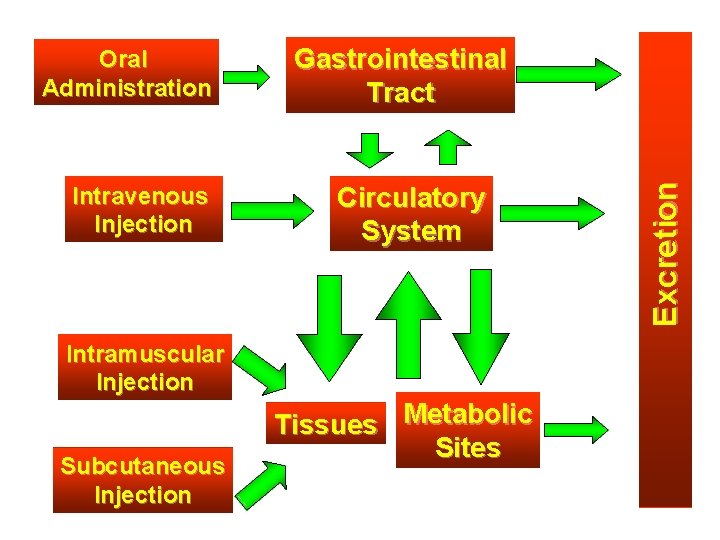
Intravenous Injection Gastrointestinal Tract Circulatory System Intramuscular Injection Subcutaneous Injection Tissues Metabolic Sites Excretion Oral Administration
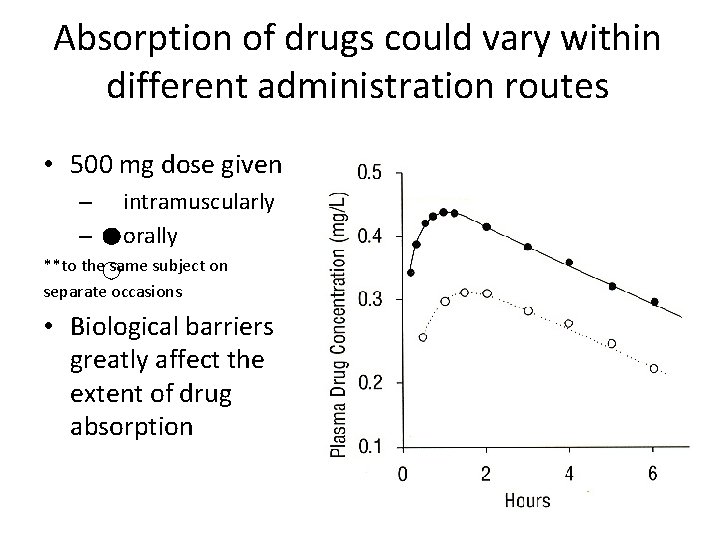
Absorption of drugs could vary within different administration routes • 500 mg dose given – intramuscularly – orally **to the same subject on separate occasions • Biological barriers greatly affect the extent of drug absorption
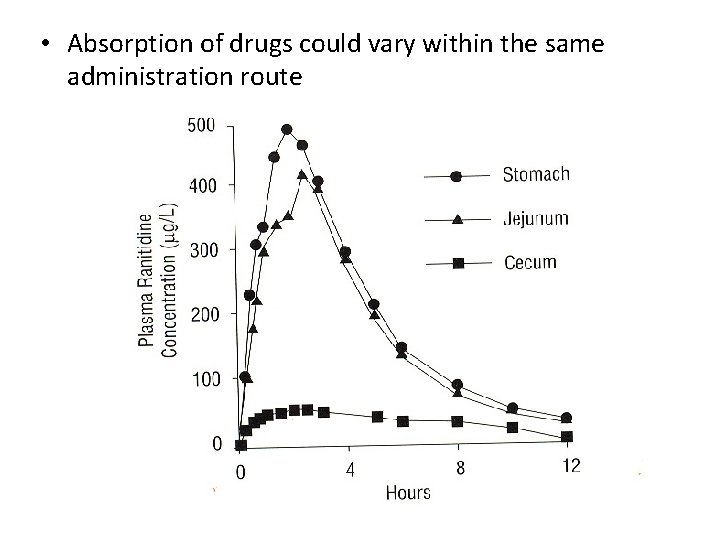
• Absorption of drugs could vary within the same administration route

Mathematical Modeling of Drug Disposition • • • Single compartment with absorption Two compartments with absorption Physiological Models

Determination of the Efficacy of the Delivery Route • Bioavailability (F) – Fraction of the drug that reached the systemic circulation – According to the FDA, Food, Drug, and Cosmetic Act • “The rate and extent to which an active ingredient or active moiety is absorbed from a drug product and becomes available at the site of action. For drugs that are not intended to be absorbed in the bloodstream, bioavailability may be assessed by measurements intended to reflect the rate and extent to which the active ingredient or active moiety becomes available at the site of action. ”

Factors Influencing Bioavailabilty • • Delivery route The site of measurement Type of animal employed Physiological state of the animal/human – Disease – Anesthesia

Implications of PK and PD in Drug Delivery • The PK and PD of a drug may be affected when administered via different routes – Examples • Proteins – oral vs. intramuscular • Morphine – oral vs. intramuscular • The PK and PD of a drug delineates its therapeutic window – Degree of absorption – Degree of elimination and/or metabolism • Example – Tetracycline (infection) – given 6 to 8 hours – Digoxin (cardiac failure)– given daily

DELIVERY SYSTEM • Proteins with a peptide sequence that shows the capability to translocate membrane rapidly, termed as ‘‘cell penetrating peptide (CPP) or protein transduction domain(PTD) are covalently modified by methods mentioned below: 1. Recombinant fusion protein from a vector containing DNA sequence of the CPP sequence are directly expressed 2. Linker such as disulfide bond linkage that is cleavable under reductive environment are used for protein or chemical conjugation of CPP to the protein

• Peptide based biomaterial are used for delivery to protect protein from protease degradation & Strategy to improve delivery efficiency for the following reasons : -Easy to synthesize -Easy characterization -Less toxic & has higher immunogenicity than high molecular wt polymers. • Due to its amphipathic character of peptides can associate rapidly with protein cargos in solution in self-assembly manner, possibly through noncovalent hydrophobic interaction.

Protein polymers conjugate for Targeted delivery • Most commonly employed polymer : Polyethylene glycol(PEG) & Poly(Nisopropyl acrylamide) (PNIPAM) • These polymers that alter their solubility or propensity for self-assembly when exposed to changes in p. H or temperature allow their responsive nature to be conferred to the protein to which they are attached. • Functionalizable with active esters & hence can be conjugated with protein amine.
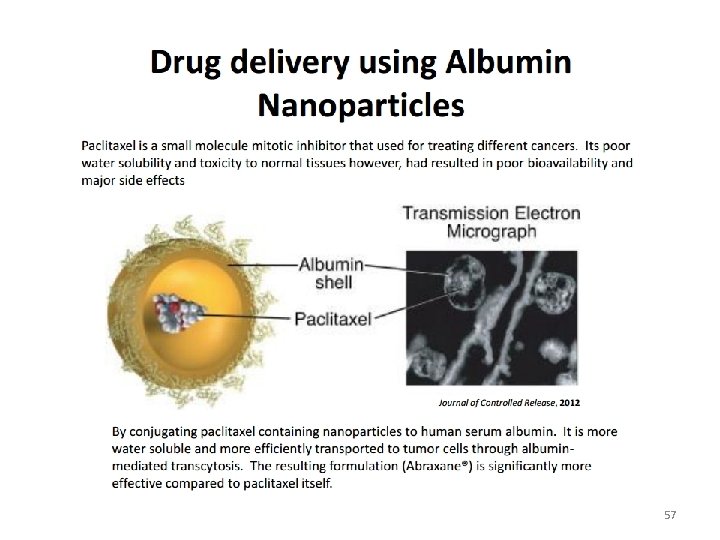
57

58
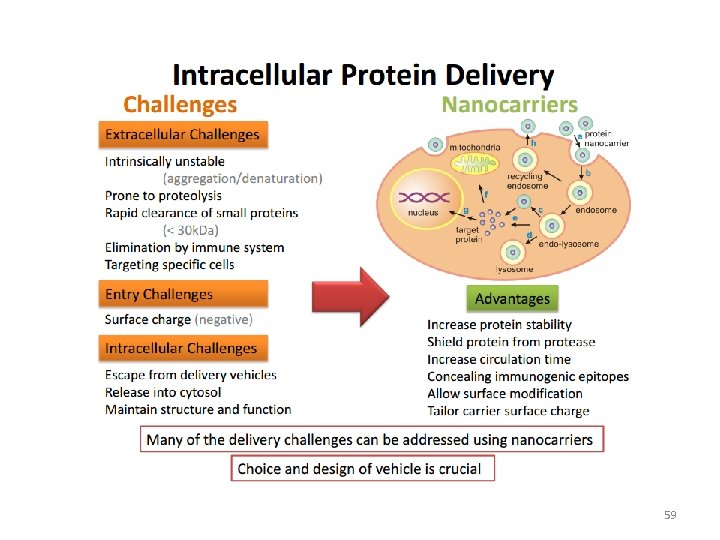
59
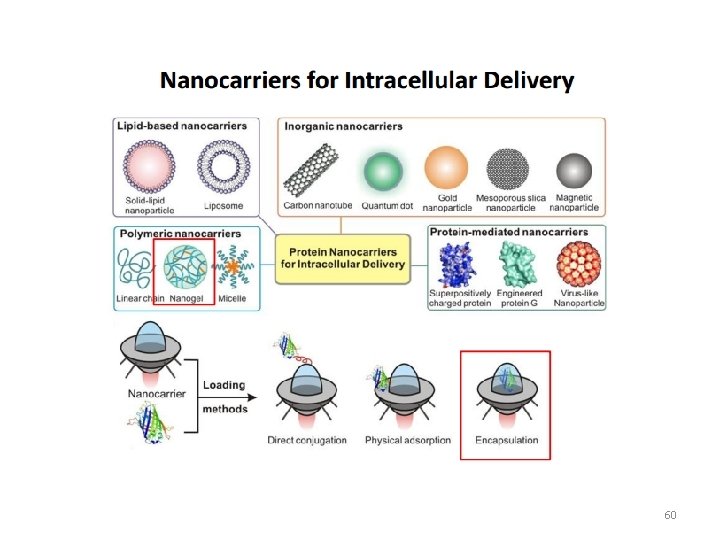
60
- Slides: 60