THEORY INSTRUMENTATION OF AFFINITY CHROMATOGRAPHY Contents Introduction Principle

























- Slides: 32

THEORY & INSTRUMENTATION OF AFFINITY CHROMATOGRAPHY.

Contents Introduction Principle Materials Applications 2

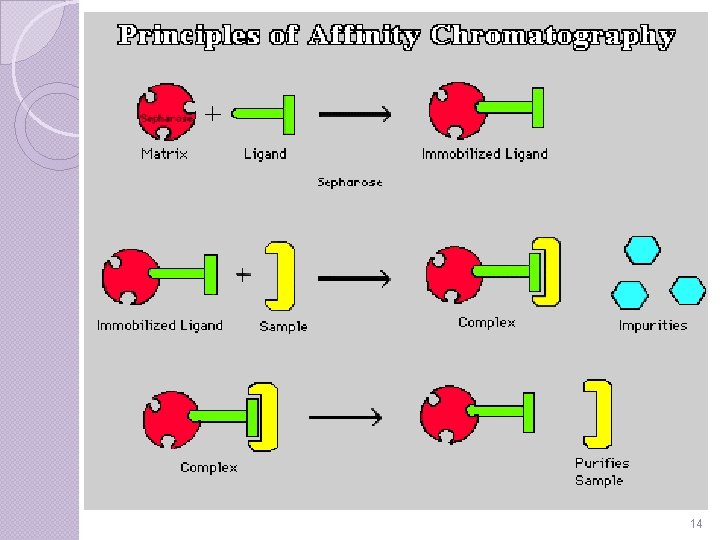
INTRODUCTION Affinity chromatography is a technique enabling purification of a biomolecule with respect to biological function or individual chemical structure. The substance to be purified is specifically and reversibly adsorbed to a ligand(binding substance), immobilized by a covalent bond to a chromatographic bed material(matrix). A ligand which exhibits a specific affinity for a particular compound gets covalently bonded to gel matrix and material is filled in to the column. 3

Choice of gel depends on type of group present in ligand molecule and nature of binding reaction with the substance to be purified. Samples are applied under favorable conditions for their specific binding to the ligand. Substances of interest are consequently bound to the ligand while unbound substances are washed away. Recovery of molecules of interest can be achieved by changing experimental conditions to favour desorption. 4

PRINCIPLE In this, the molecule of interest will have a well known & defined property which can be separated from complex mixtures in single process. The target molecule becoming trapped on a solid or stationary phase or medium. The other molecules in the solution will not become trapped as they do not posses this property. The solid medium can then be removed from the mixture, washed and the target molecule released from the entrapment in a process known as elution. 5

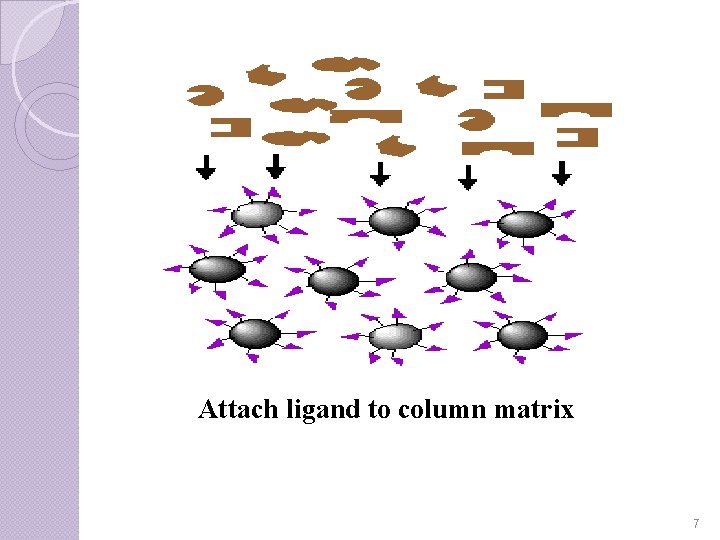
Step-I Binding of the selected ligand to the matrix requires that a covalent bond to be formed between the two. It is important to realize that the substrate might not be able to reach the ligand active site if it is hidden deep within the ligand. Therefore, most ligands are attached first to spacer arms which are then bonded to the matrix. The ligand -matrix gel is then loaded into an elution column. 6

Attach ligand to column matrix 7

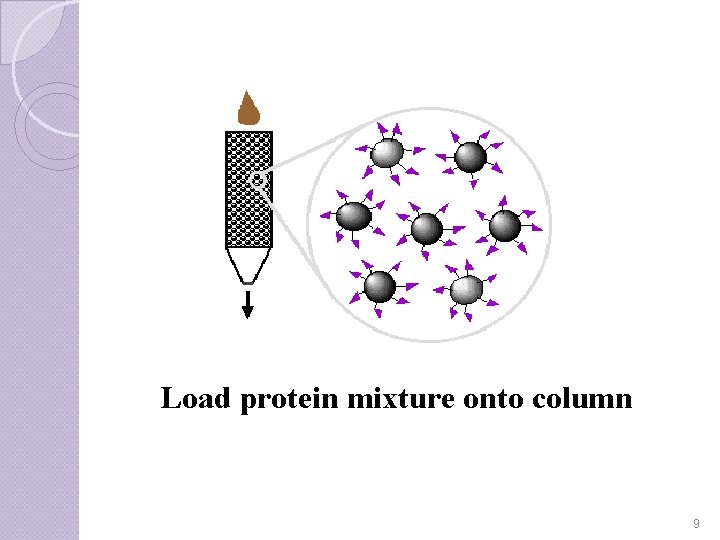
Step-II Once the column has been prepared, the mixture containing isolate is poured into the elution column. Gravity pulls the solution through the gel, because most of the proteins do not bind to the ligand-matrix complex. Some of the impurities flow through the gel, but most remain unbound in the gel column. 8

Load protein mixture onto column 9

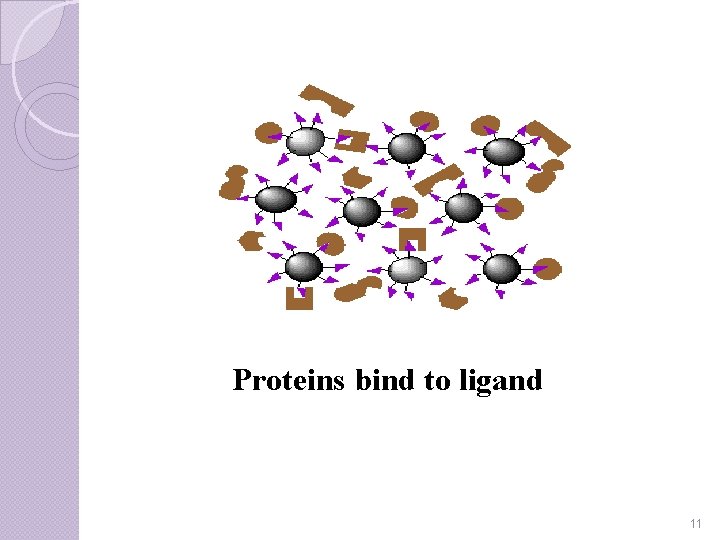
Step-III In order to remove these unbound impurities, a wash of extreme p. H, salt concentration, or temperature is run through the gel. It is important to use a strong wash so that all the impurities are removed. Once the impurities are washed out the only remaining part of the protein mixture should be the desired isolates. 10

Proteins bind to ligand 11

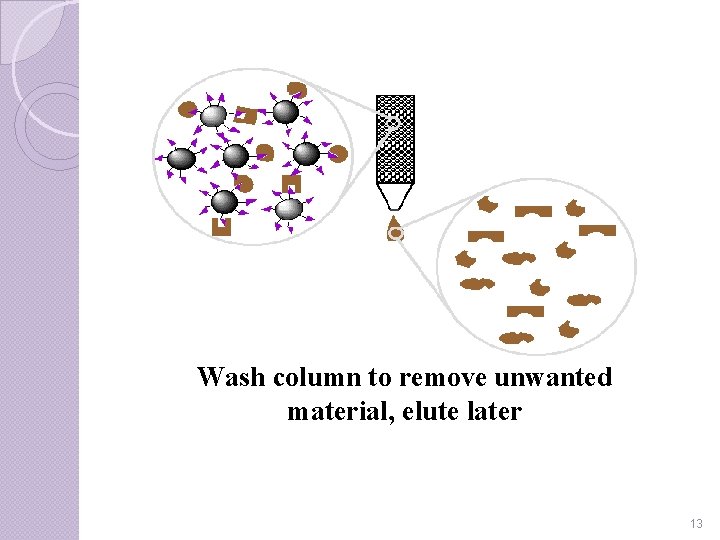
Step-IV Finally to collect isolate, which is still bound to the ligand-matrix in the gel, a stronger second wash is run through the column. The protein is then free to run through the gel and be collected. 12

Wash column to remove unwanted material, elute later 13

14

Materials Major materials required for an affinity chromatography procedure a bead matrix a ligand a solution containing the substrate to be isolated a wash to elute the non bound impurities in the solution a final wash to elute the bound substrate from its ligand 15

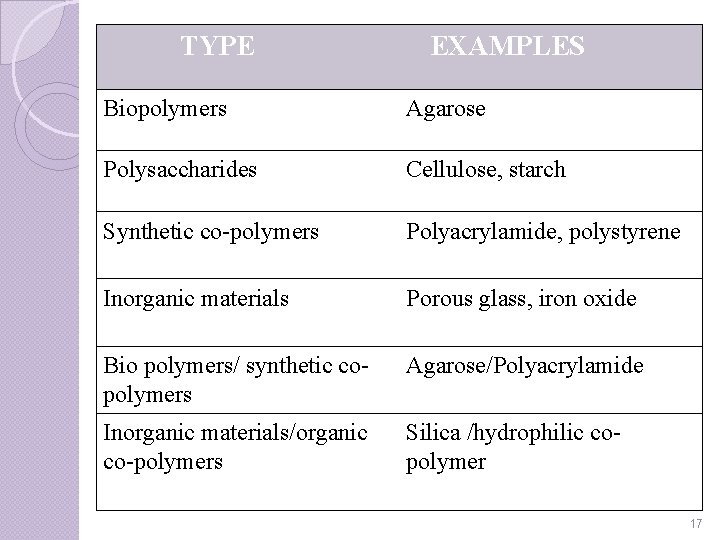
Bead Matrix CHARACTERISTICS � It should exhibit good flow property. � Mechanically & chemically stable and resistance against microorganisms. � Should posses functional group to bind with ligand. � Should be economical. � Adequate particle size and shape. Eg: increase in particle size reduces flow resistance and separation power. 16

TYPE EXAMPLES Biopolymers Agarose Polysaccharides Cellulose, starch Synthetic co-polymers Polyacrylamide, polystyrene Inorganic materials Porous glass, iron oxide Bio polymers/ synthetic copolymers Agarose/Polyacrylamide Inorganic materials/organic co-polymers Silica /hydrophilic copolymer 17

Ligand � The ligand bind only to the desired molecule with in the solution. � The ligand attaches to the matrix which is made up of an inert substance. � The ligand should only interact with the desired molecule and form a temporary bond. � The ligand/molecule complex dissociates by changing the p. H. � It is selected according to the desired isolate. � For e. g. if isolate antibodies specific for antigen A from antiserum, then antigen A acts as ligand. 18

Characteristics of ligand � The ligand must be readily available. � Ligand must be attachable(covalently) to the matrix such that it still retains affinity for protein. � Binding must not be too strong/weak. 19

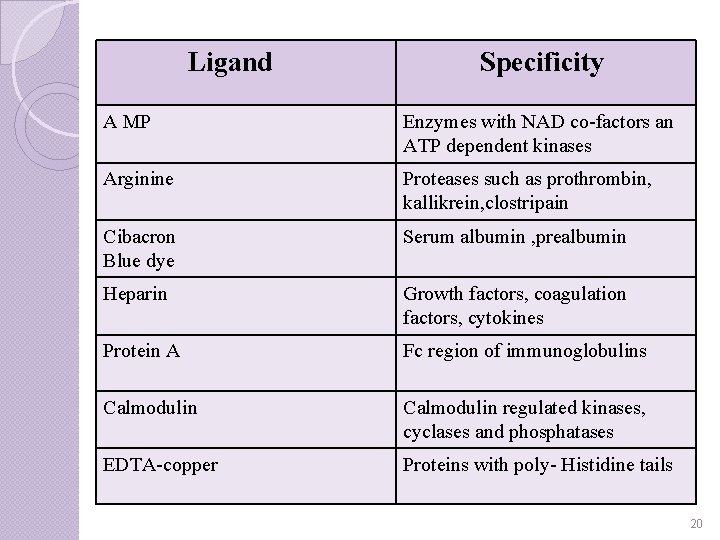
Ligand Specificity A MP Enzymes with NAD co-factors an ATP dependent kinases Arginine Proteases such as prothrombin, kallikrein, clostripain Cibacron Blue dye Serum albumin , prealbumin Heparin Growth factors, coagulation factors, cytokines Protein A Fc region of immunoglobulins Calmodulin regulated kinases, cyclases and phosphatases EDTA-copper Proteins with poly- Histidine tails 20

Solution �The solution usually is a protein rich mixture such as anti-serum, which is poured into the elution column and allowed to run through the gel, at a controlled rate. 21

Prepacked columns: Hi. Trap Heparin HP (High Performance) Column size: 5 x 1 mm 1 x 5 mm 5 x 5 mm Average particle diameter: 34µm Maximum operating flow rate: 4 ml/min ; 20 ml/min 22

Storage � Store the prepacked columns at 2 -8 °C in an upright position with both caps in place. Thiomersal may be added for long term storage. � DONOT FREEZE. 23

ELUTION DIFFERENT METHOD USED FOR ELUTION � Non-specific � Special elution 24

Non specific: Ø Change of p. H, ionic strength, temperature, polarity & using different reagent like deforming eluents, urea, detergent. Ø In many cases change in p. H leads to desorption. Ø E. g. for protease adsorption occurs at 8. 1 p. H & elution at 3. 1 p. H. 25

Specific method: Ø Affinity elution (with substrate, co-factors, inhibitors) Special method: Ø Electrophoresis, cleavage of matrix ligand bond, buffer effect. Ø Electrophoresis is suited for desorption of antibiotics and hormone binding protein. Ø By reductive cleavage of aza-linkage with dithionate, serum estradiol-binding protein are isolated from estradiol- agarose. 26

Advantages ü High degrees of purity can be obtained. ü The process is very reproducible. ü The binding sites of biological molecules can be simply investigated. 27

Disadvantages ü Cost, the difficulties associated with scale-up &high labor intensity. ü With such drawbacks, affinity chromatography tends to be used in the purification of specialty chemicals & pharmaceutical compounds, where the need for high purity samples outweighs the cost of production. 28

Applications ü The goal of affinity chromatography is to separate all the molecules of a particular specificity from the whole amount of molecules in a mixture such as blood serum. For e. g. the antibodies in a serum sample. ü Purification of substances from biological mixtures. ü Removal of small amounts of biomaterial from large amounts of contaminants. ü Purify & concentrate a molecule from a mixture in solution, even at very low concentrations. 29

Industrial Applications ü Affinity chromatography is widely used in the pharmaceutical industry to purify & extract molecules of interest from complex mixtures. ü These molecules tend to be enzymes, proteins or amino acids, but other biological species can be selectively retained. ü Once isolated, these biological species can be selectively amplified to produce larger quantities, although at large concentrations. 30

REFERENCES � Instrumental Methods of Analysis seventh edition: Willard. Meritt. Dean. Settle. Pg. no. 650 -652. � Instrumental Techniques for Analytical Chemistry. FRANK SETTLE. Pg. no: 158. � Principles of Quantitative Chemical Analysis-Robert de Analysis Levie. Pg. no: 284 � Chromatographic Seperations, Wiley series- Peter A. Seperations Sewell, Brian Clarke. Pg. no: 33 � IMAGES: IMAGES www. google. com 31

Thank you …!!! 32