Theories of Covalent Bonding Theories of Covalent Bonding

Theories of Covalent Bonding

Theories of Covalent Bonding 11. 1 VSEPR Theory 11. 2 Valence Bond Theory 11. 3 Molecular Orbital (MO)Theory

VSEPR - Valence Shell Electron Pair Repulsion Theory Each group of valence electrons around a central atom is located as far away as possible from the others in order to maximize repulsions. These repulsions maximize the space that each object attached to the central atom occupies. The result is five electron-group arrangements of minimum energy seen in a large majority of molecules and polyatomic ions. The electron-groups are defining the object arrangement, but the molecular shape is defined by the relative positions of the atomic nuclei. Because valence electrons can be bonding or nonbonding, the same electron-group arrangement can give rise to different molecular shapes. A - central atom X -surrounding atom AXm. En integers E -nonbonding valence electron-group

Valence Bond Theory • Lewis structures and VSEPR do not explain why a bond forms. • How do we account for shape in terms of quantum mechanics? • What are the orbitals that are involved in bonding? • We use Valence Bond Theory: Prentice Hall © 2003 Chapter 9

The Central Themes of VB Theory Themes A set of overlapping orbitals has a maximum of two electrons that must have opposite spins. The greater the orbital overlap, the stronger (more stable) the bond. Types of overlap: 1. head-on overlap -formed by sigma bonds 2. Sigma bonds -formed by pi bonds There are two electrons of opposite spin in the orbital overlap
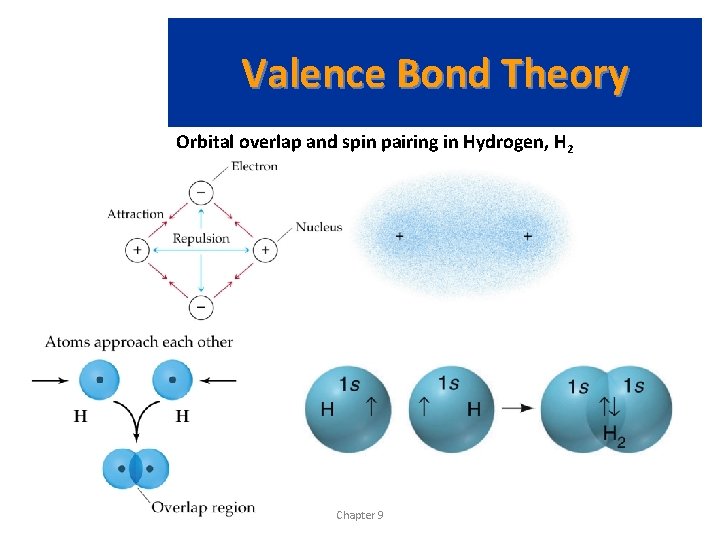
Valence Bond Theory Orbital overlap and spin pairing in Hydrogen, H 2 Prentice Hall © 2003 Chapter 9

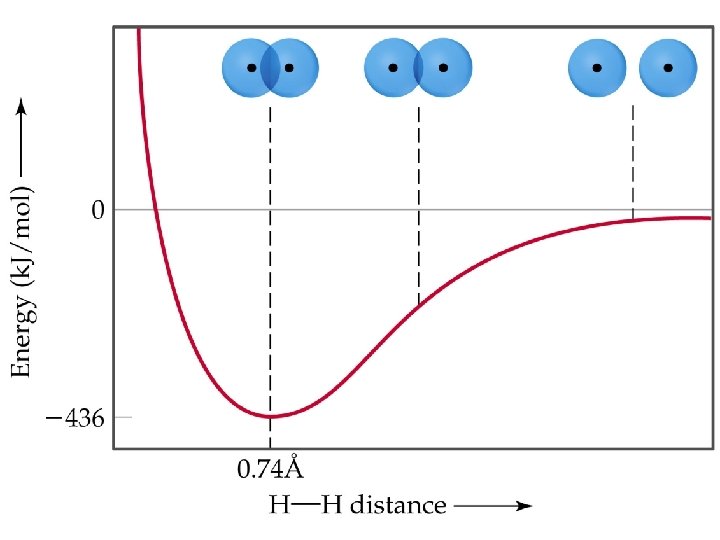
Valence Bond Theory • As two nuclei approach each other their atomic orbitals overlap. • As the amount of overlap increases, the energy of the interaction decreases. • At some distance the minimum energy is reached. • The minimum energy corresponds to the bonding distance (or bond length). • At the bonding distance, the attractive forces between nuclei and electrons just balance the repulsive forces (nucleus-nucleus, electron-electron). • As the two atoms get closer, their nuclei begin to repel and the energy increases. Prentice Hall © 2003 Chapter 9
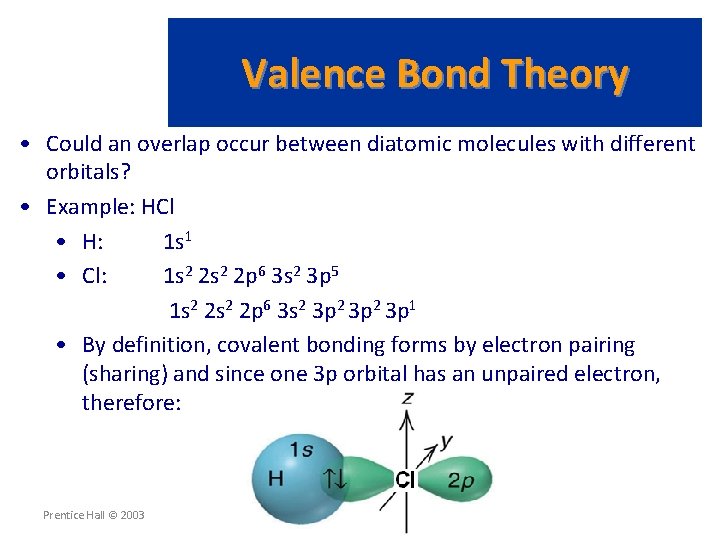
Valence Bond Theory • Could an overlap occur between diatomic molecules with different orbitals? • Example: HCl • H: 1 s 1 • Cl: 1 s 2 2 p 6 3 s 2 3 p 5 1 s 2 2 p 6 3 s 2 3 p 1 • By definition, covalent bonding forms by electron pairing (sharing) and since one 3 p orbital has an unpaired electron, therefore: Prentice Hall © 2003 Chapter 9
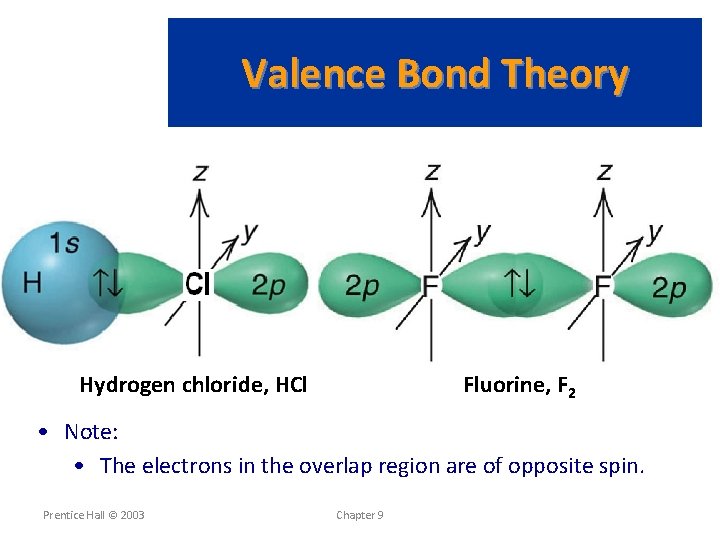
Valence Bond Theory Hydrogen chloride, HCl Fluorine, F 2 • Note: • The electrons in the overlap region are of opposite spin. Prentice Hall © 2003 Chapter 9

Hybrid Orbitals • Atomic orbitals can mix or hybridize in order to adopt an appropriate geometry for bonding. • Key points: • The number of hybrid orbitals obtained equals the number of atomic orbitals mixed. • The type of hybrid orbitals obtained varies with the types of atomic orbitals mixed. Prentice Hall © 2003 Chapter 9

Hybrid Orbitals • Hybridization is determined by the electron domain geometry • Types of Hybrid Orbitals: • sp 2 • sp 3 d 2 Prentice Hall © 2003 Chapter 9

Hybrid Orbitals sp Hybrid Orbitals • Consider the Be. Cl 2 molecule (experimentally known to exist): • • Be has a 1 s 22 s 2 electron configuration. Cl has a [Ne] 3 s 2 3 p 5 electronic configuration. Be has no unpaired electron available for bonding. We conclude that the atomic orbitals are not adequate to describe orbitals in molecules. • We know that the Cl-Be-Cl bond angle is 180 (VSEPR theory). • We also know that one electron from Be is shared with each one of the unpaired electrons from Cl. Prentice Hall © 2003 Chapter 9

Hybrid Orbitals sp Hybrid Orbitals • How could Be. Cl 2 occur then? • How does a covalent bond form between Be and l atoms? • How could a linear geometry be possible? Prentice Hall © 2003 Chapter 9
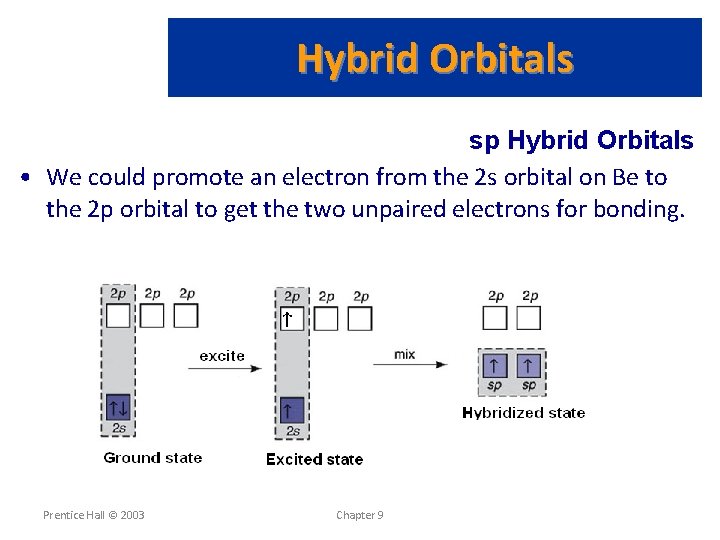
Hybrid Orbitals sp Hybrid Orbitals • We could promote an electron from the 2 s orbital on Be to the 2 p orbital to get the two unpaired electrons for bonding. Prentice Hall © 2003 Chapter 9

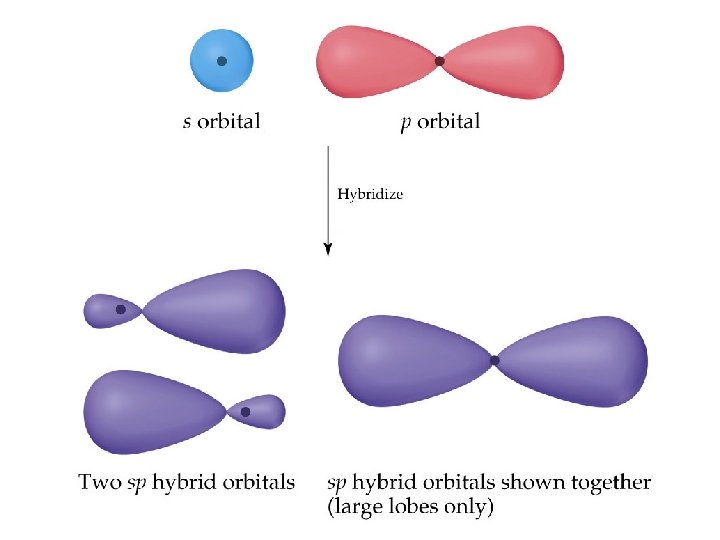
Hybrid Orbitals • We have solved the problem by allowing the 2 s and one 2 p orbital on Be to mix or form a hybrid orbital. • The hybrid orbital comes from an s and a p orbital and is called an sp hybrid orbital. Prentice Hall © 2003 Chapter 9

Hybrid Orbitals • How about the geometry? • How does an sp orbital look like? Prentice Hall © 2003 Chapter 9
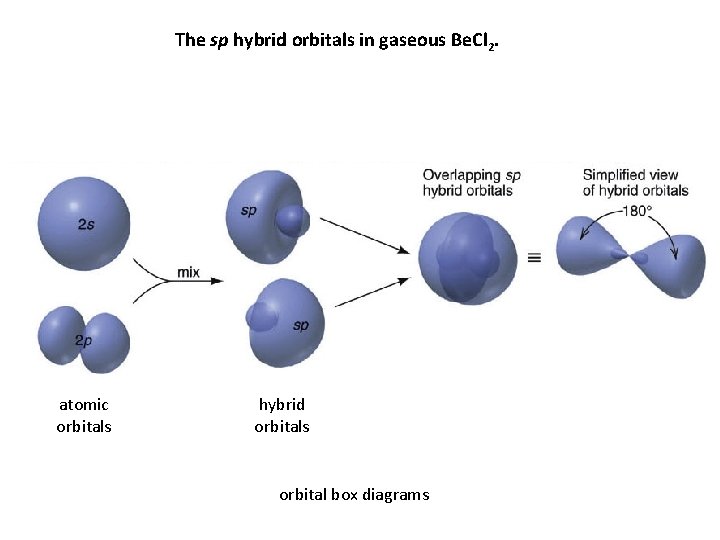
The sp hybrid orbitals in gaseous Be. Cl 2. atomic orbitals hybrid orbitals orbital box diagrams
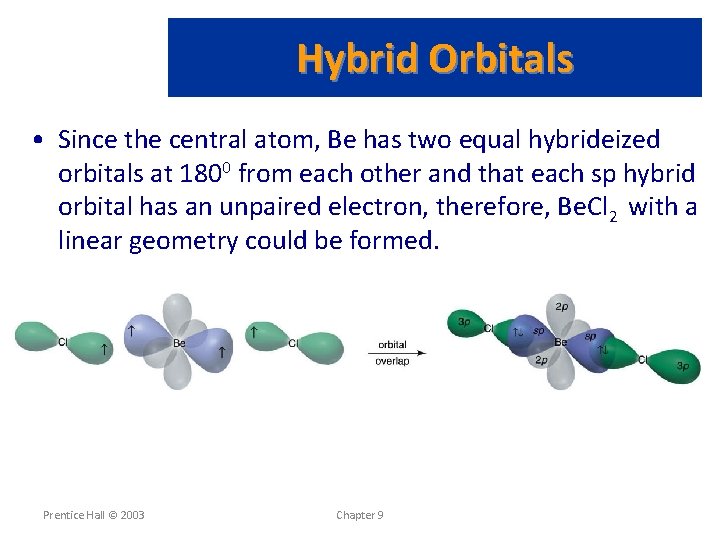
Hybrid Orbitals • Since the central atom, Be has two equal hybrideized orbitals at 1800 from each other and that each sp hybrid orbital has an unpaired electron, therefore, Be. Cl 2 with a linear geometry could be formed. Prentice Hall © 2003 Chapter 9

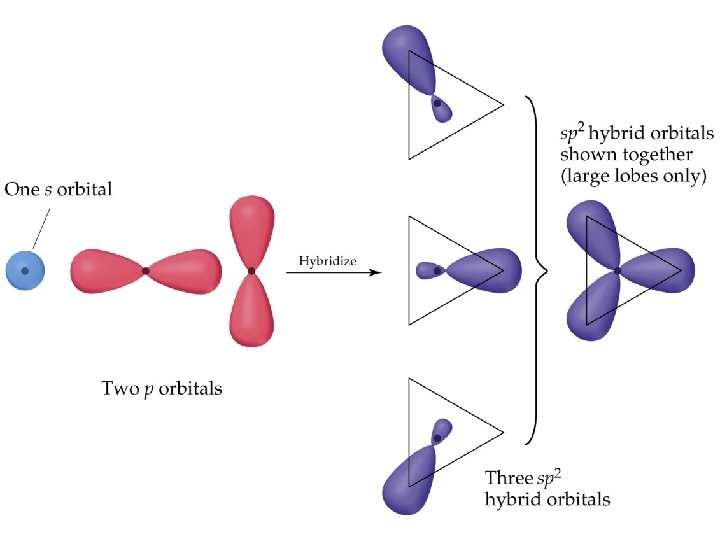
Hybrid Orbitals • • sp 2 Hybrid Orbitals Important: when we mix n atomic orbitals we must get n hybrid orbitals. sp 2 hybrid orbitals are formed with one s and two p orbitals. (Therefore, there is one unhybridized p orbital remaining. ) The large lobes of sp 2 hybrids lie in a trigonal plane. All molecules with trigonal planar electron pair geometries have sp 2 orbitals on the central atom. Prentice Hall © 2003 Chapter 9

Hybrid Orbitals • • sp 3 Hybrid Orbitals sp 3 Hybrid orbitals are formed from one s and three p orbitals. Therefore, there are four large lobes. Each lobe points towards the vertex of a tetrahedron. The angle between the large lobes is 109. 5. All molecules with tetrahedral electron pair geometries are sp 3 hybridized. Prentice Hall © 2003 Chapter 9
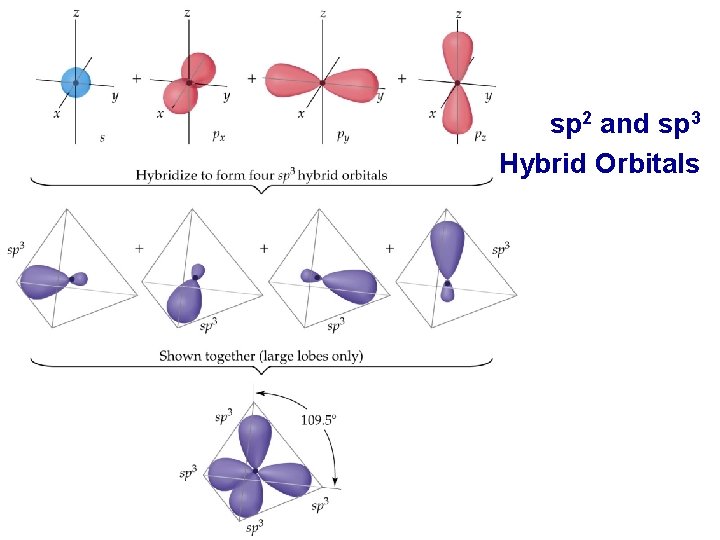
sp 2 and sp 3 Hybrid Orbitals
- Slides: 24