Theoretical Analysis of the Hyperfine Structure of Na



![More Angular Momentum Coupling F= N+S+I Case 1 Case 2 F= [N+S] + I More Angular Momentum Coupling F= N+S+I Case 1 Case 2 F= [N+S] + I](https://slidetodoc.com/presentation_image_h2/4c14890e3dda99dcaba42e83b84ff7c6/image-15.jpg)




- Slides: 26

Theoretical Analysis of the Hyperfine Structure of Na. K Angela Wilkins Advisors : Dr. Hickman of Lehigh U. & Dr. Semak of UNC

Outline • • Molecular Spectroscopy Energy levels of Na. K Angular Momentum Coupling Conclusions

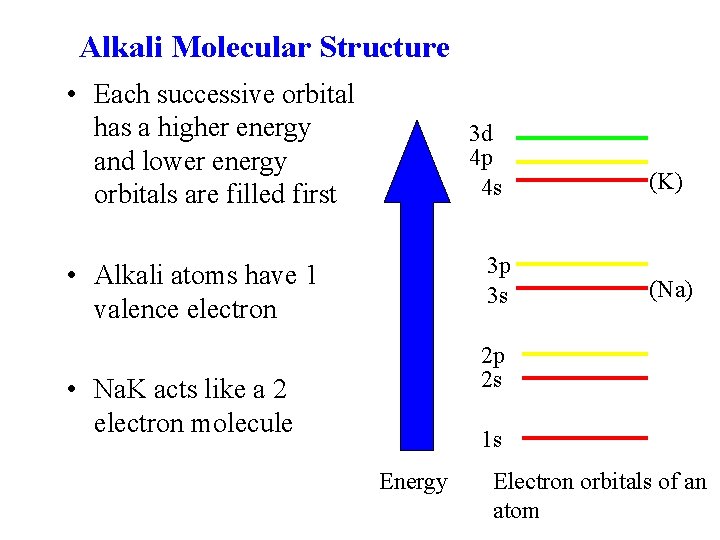
Alkali Molecular Structure • Each successive orbital has a higher energy and lower energy orbitals are filled first 3 d 4 p 4 s 3 p 3 s • Alkali atoms have 1 valence electron (K) (Na) 2 p 2 s • Na. K acts like a 2 electron molecule 1 s Energy Electron orbitals of an atom

Molecular Spectroscopy spectroscopy allows study of different energy levels Internuclear separation (R) Na K

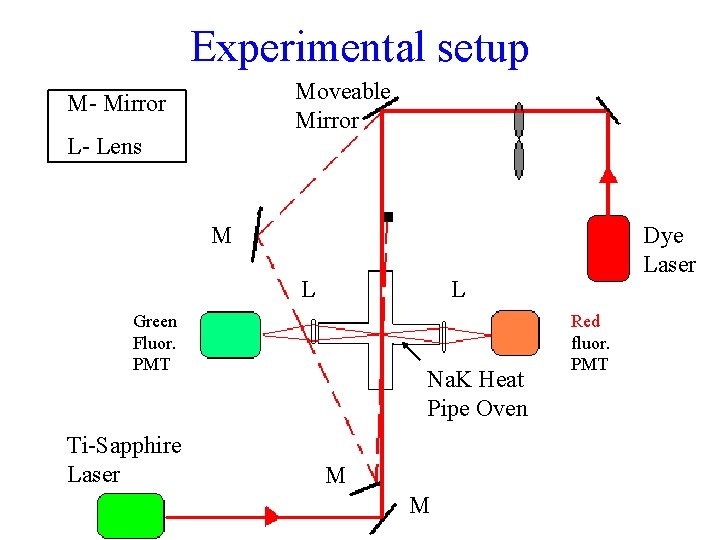
Experimental setup Moveable Mirror M- Mirror L- Lens M L L Green Fluor. PMT Ti-Sapphire Laser Dye Laser Na. K Heat Pipe Oven M M Red fluor. PMT

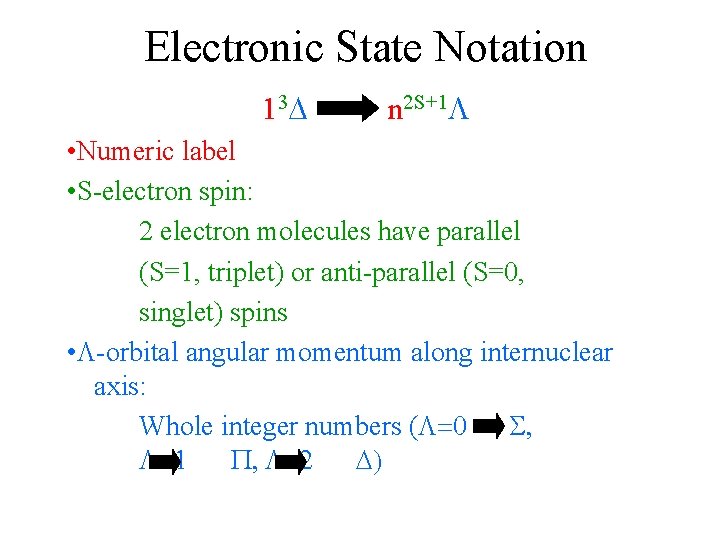
Electronic State Notation 13 n 2 S+1 • Numeric label • S-electron spin: 2 electron molecules have parallel (S=1, triplet) or anti-parallel (S=0, singlet) spins • -orbital angular momentum along internuclear axis: Whole integer numbers ( =0 S, =1 P, =2 )

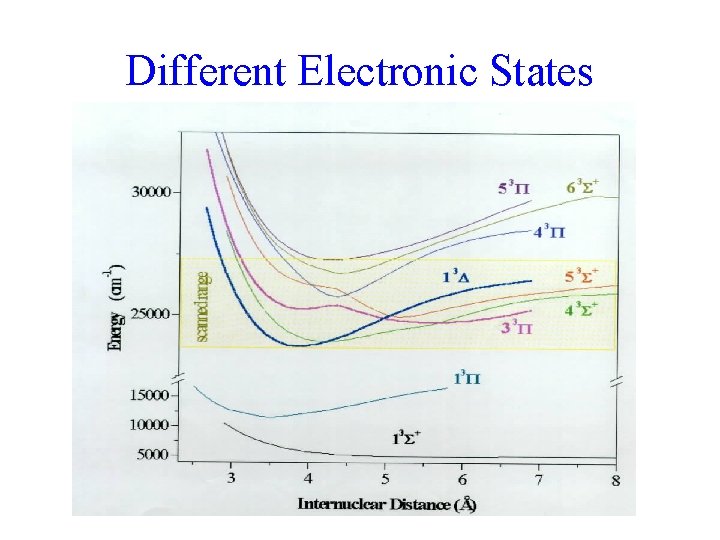
Different Electronic States

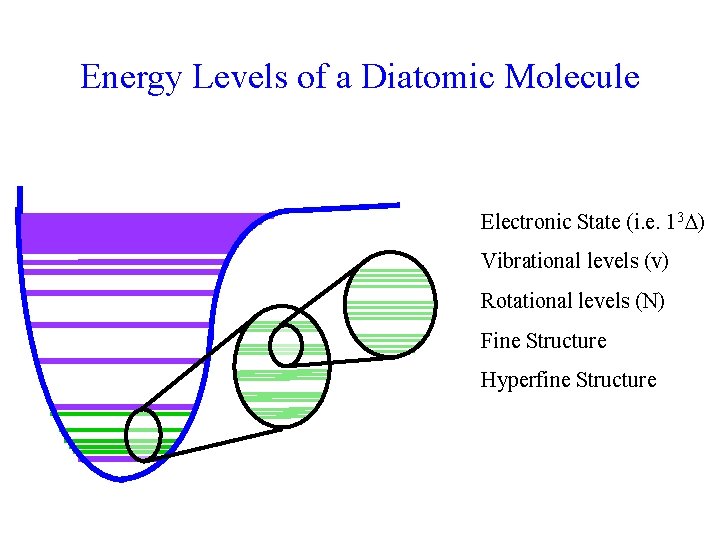
Energy Levels of a Diatomic Molecule Electronic State (i. e. 13 ) Vibrational levels (v) Rotational levels (N) Fine Structure Hyperfine Structure

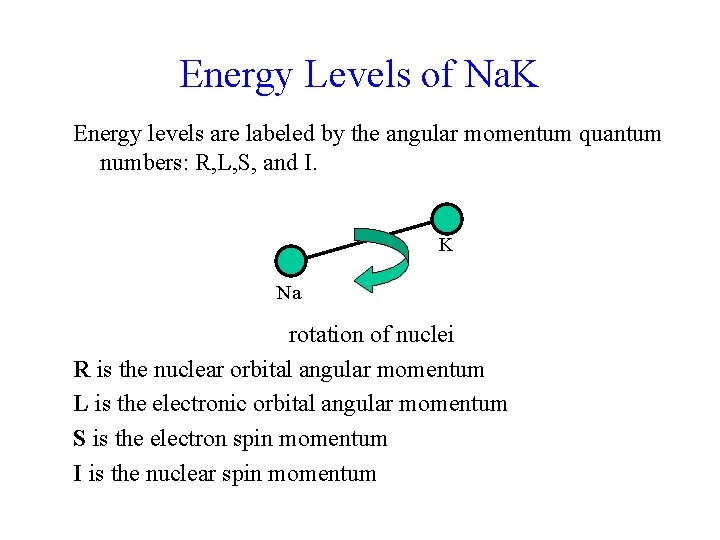
Energy Levels of Na. K Energy levels are labeled by the angular momentum quantum numbers: R, L, S, and I. K Na rotation of nuclei R is the nuclear orbital angular momentum L is the electronic orbital angular momentum S is the electron spin momentum I is the nuclear spin momentum

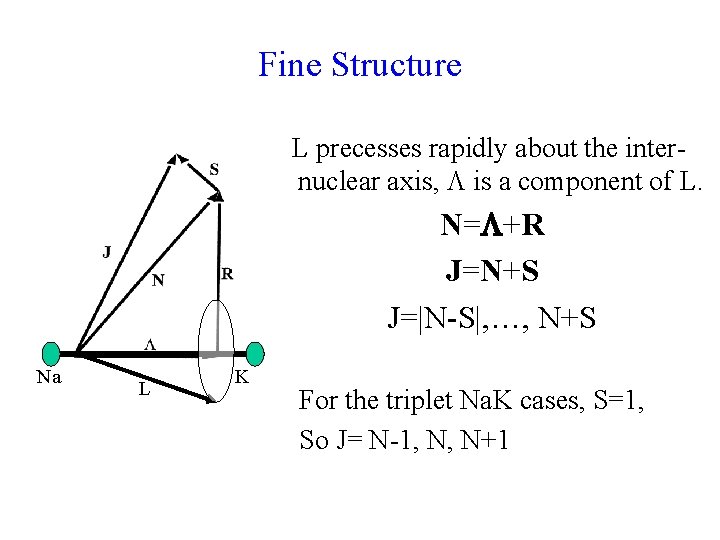
Fine Structure L precesses rapidly about the internuclear axis, is a component of L. N= +R J=N+S J=|N-S|, …, N+S Na L K For the triplet Na. K cases, S=1, So J= N-1, N, N+1

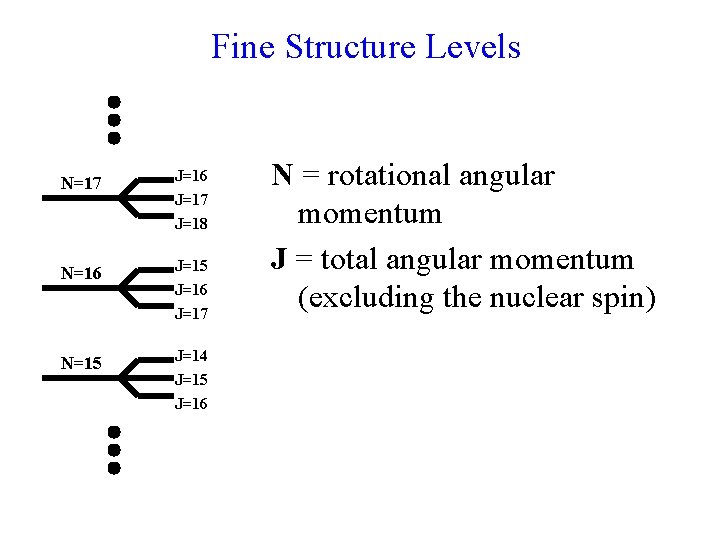
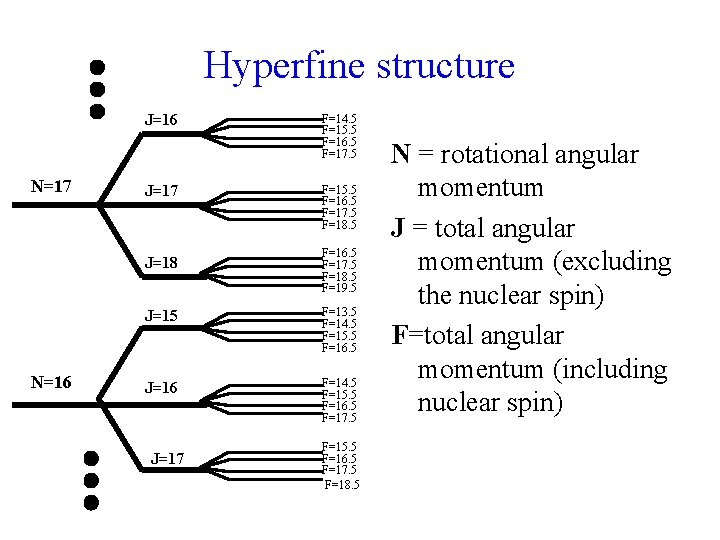
Fine Structure Levels N=17 J=16 J=17 J=18 N=16 J=15 N=15 J=14 J=15 J=16 J=17 N = rotational angular momentum J = total angular momentum (excluding the nuclear spin)

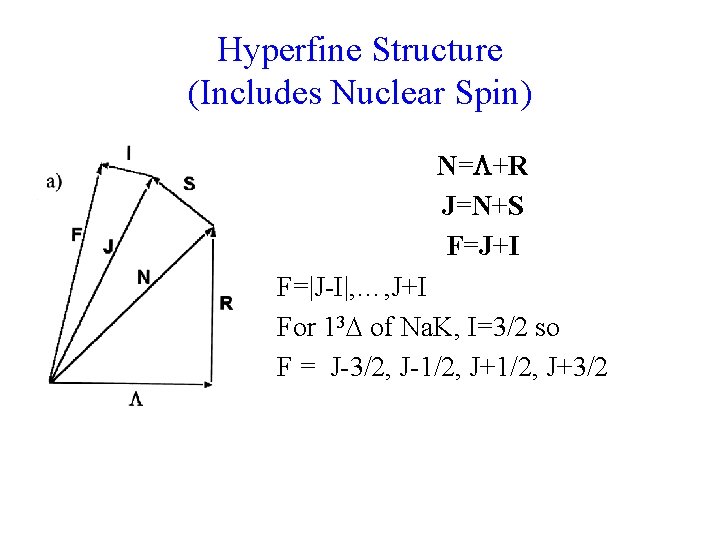
Hyperfine Structure (Includes Nuclear Spin) N= +R J=N+S F=J+I F=|J-I|, …, J+I For 13 of Na. K, I=3/2 so F = J-3/2, J-1/2, J+3/2

Hyperfine structure J=16 N=17 J=18 N=16 F=14. 5 F=15. 5 F=16. 5 F=17. 5 F=18. 5 F=19. 5 J=15 F=13. 5 F=14. 5 F=15. 5 F=16. 5 J=16 F=14. 5 F=15. 5 F=16. 5 F=17. 5 J=17 F=15. 5 F=16. 5 F=17. 5 F=18. 5 N = rotational angular momentum J = total angular momentum (excluding the nuclear spin) F=total angular momentum (including nuclear spin)

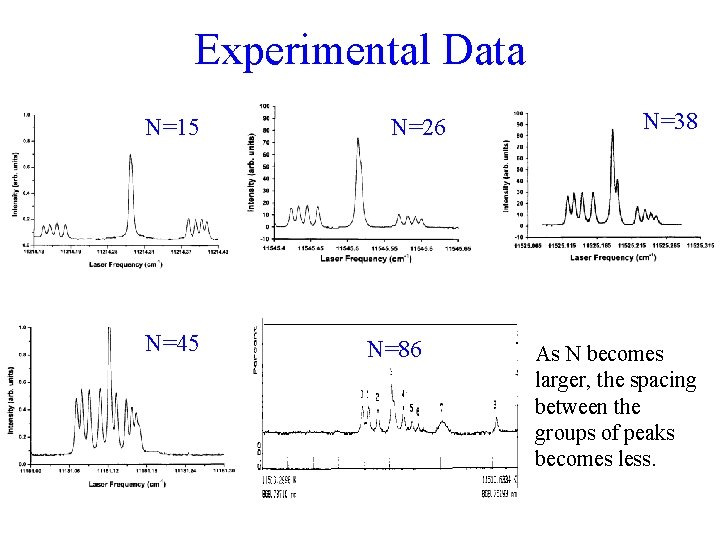
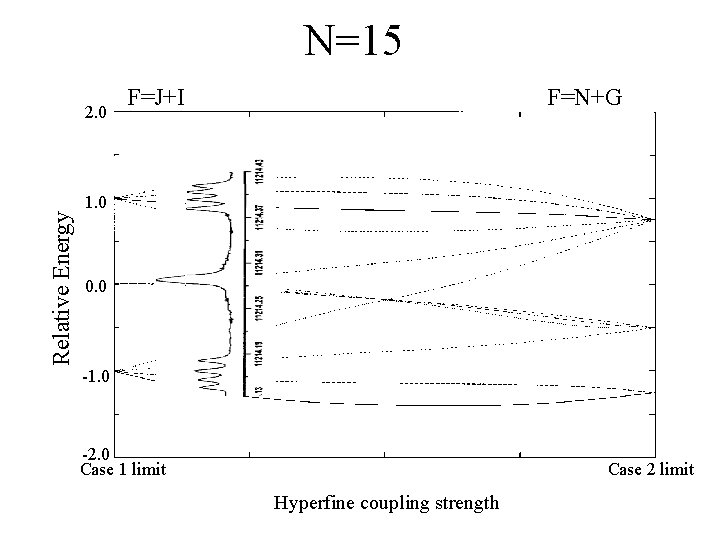
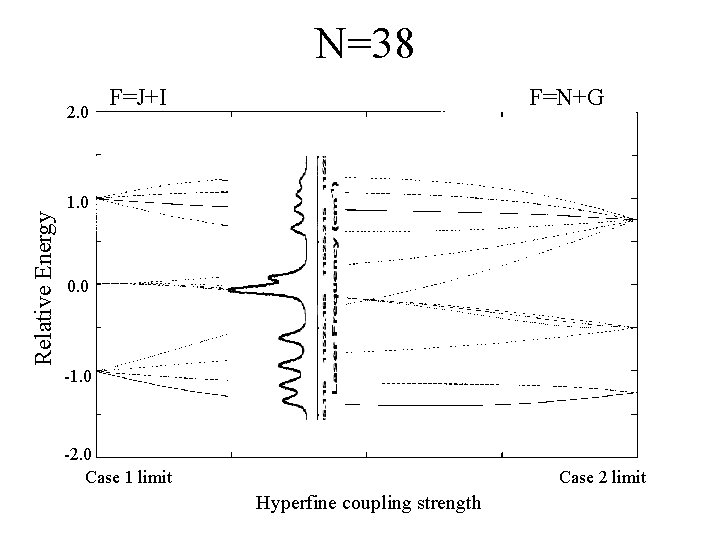
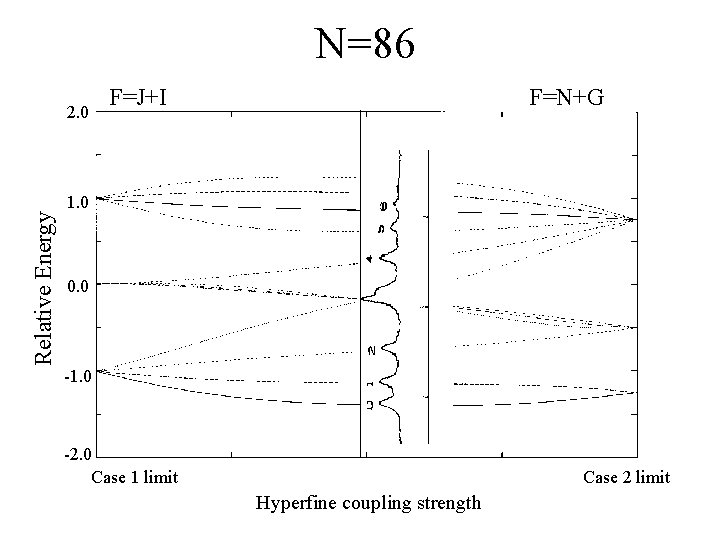
Experimental Data N=15 N=45 N=26 N=86 N=38 As N becomes larger, the spacing between the groups of peaks becomes less.
![More Angular Momentum Coupling F NSI Case 1 Case 2 F NS I More Angular Momentum Coupling F= N+S+I Case 1 Case 2 F= [N+S] + I](https://slidetodoc.com/presentation_image_h2/4c14890e3dda99dcaba42e83b84ff7c6/image-15.jpg)
More Angular Momentum Coupling F= N+S+I Case 1 Case 2 F= [N+S] + I F=N + [S+I] J=N+S G=S+I F=J+I F=N+G Recall: For 13 of Na. K, S=1 and I=3/2 G=|S-I|, …, S+I G=1/2, 3/2, 5/2

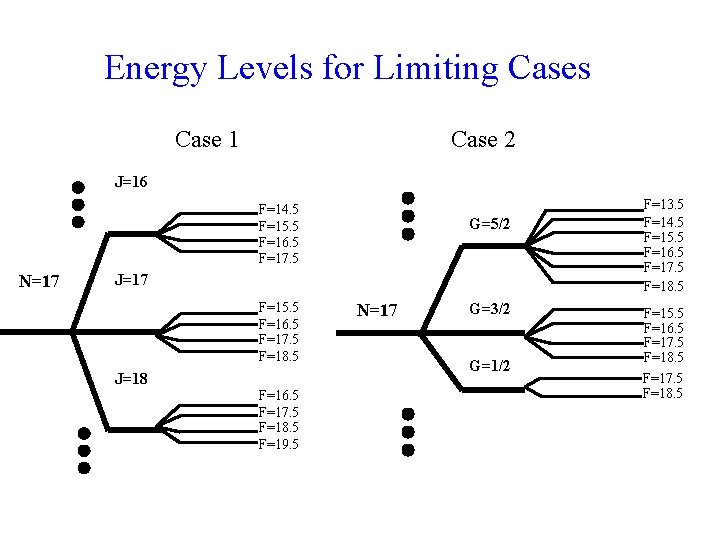
Energy Levels for Limiting Cases Case 1 Case 2 J=16 F=14. 5 F=15. 5 F=16. 5 F=17. 5 N=17 G=5/2 J=17 F=15. 5 F=16. 5 F=17. 5 F=18. 5 J=18 F=16. 5 F=17. 5 F=18. 5 F=19. 5 N=17 G=3/2 G=1/2 F=13. 5 F=14. 5 F=15. 5 F=16. 5 F=17. 5 F=18. 5

Model Hamiltonian for Na. K (3 ) H = Hspin-orbit + Hrotation + Hhyperfine + Hspin-rotation Hspin-orbit = Av. L S Hrotation= Bv [(N(N+1) - 2 ] - Dv [(N(N+1) - 2 ] 2 Hhyperfine= b. I S Hspin-rotation= R S The 12 energy levels are the eigenvalues of this Hamiltonian. We adjusted Av, b, and to fit the experimental energies. Case 1 Bv. N >> Av >> b Case 2 Bv. N >> b >>Av

Intermediate Case 2. 0 F=J+I F=N+G Relative Energy J=N-1 G=5/2 1. 0 0. 0 J=N G=3/2 J=N+1 -1. 0 G=1/2 -2. 0 Case 1 limit Case 2 limit Hyperfine coupling strength

N=15 Relative Energy 2. 0 F=J+I F=N+G 1. 0 0. 0 -1. 0 -2. 0 Case 1 limit Case 2 limit Hyperfine coupling strength

N=38 Relative Energy 2. 0 F=J+I F=N+G 1. 0 0. 0 -1. 0 -2. 0 Case 1 limit Case 2 limit Hyperfine coupling strength

N=86 Relative Energy 2. 0 F=J+I F=N+G 1. 0 0. 0 -1. 0 -2. 0 Case 1 limit Case 2 limit Hyperfine coupling strength

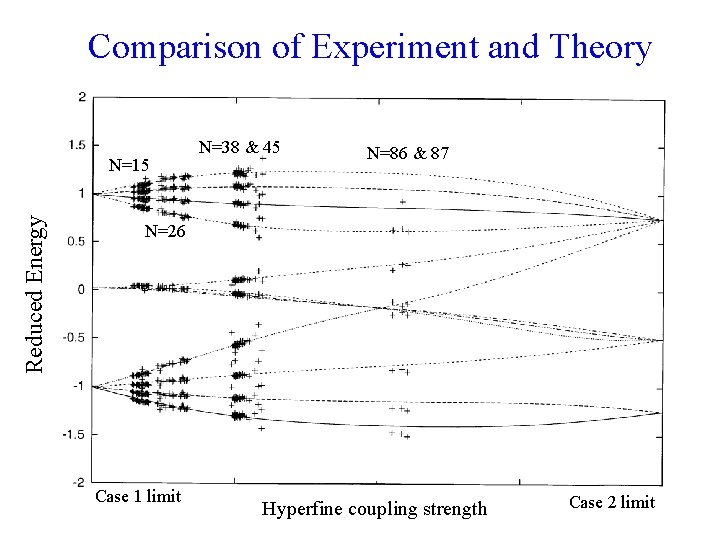
Comparison of Experiment and Theory Reduced Energy N=15 N=38 & 45 N=86 & 87 N=26 Case 1 limit Hyperfine coupling strength Case 2 limit

Conclusions • • • The intermediate angular momentum coupling cases explain data. The coupling scheme changes with N. Plan to work further and continue analysis on data at N values > 86 to check agreement with limiting cases and include other electronic states.

Acknowledgements • • • Dr A. Peet Hickman Dr Matthew Semak Dr. Huennekens Laurie Sibbach & Catherine Deibel NSF for funding

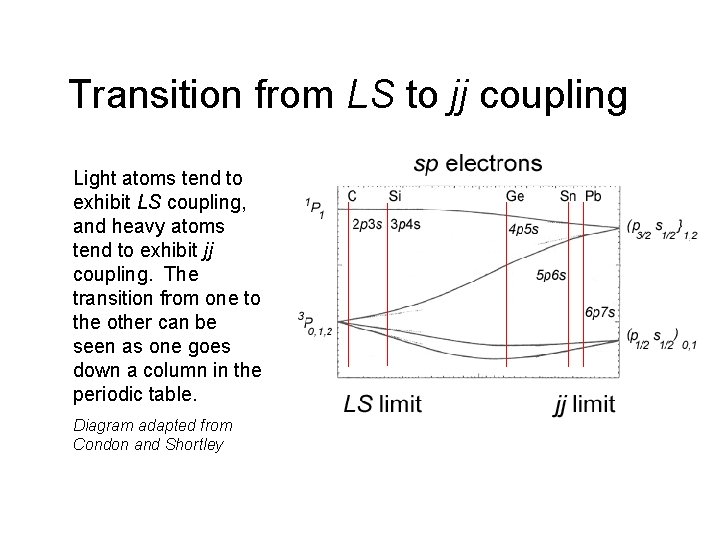
Transition from LS to jj coupling Light atoms tend to exhibit LS coupling, and heavy atoms tend to exhibit jj coupling. The transition from one to the other can be seen as one goes down a column in the periodic table. Diagram adapted from Condon and Shortley

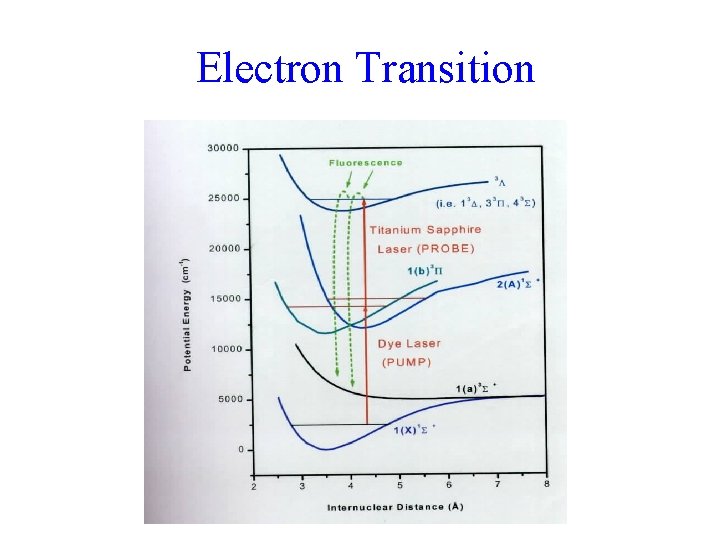
Electron Transition