The Third Annual Medical Device Regulatory Reimbursement and

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Gift Disclosure and Registration Tom Tremble, Adva. Med Julie Cohen, Adva. Med Vickie Mc. Cormick, St. Jude Medical Debjit Ghosh, Huron Consulting Group, LLC March 27, 2008

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Overview of State Legislative Activity Presented by Tom Tremble Adva. Med

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Overview of State Legislative Activity • Background • Current Proposals • What’s Driving Them • Outlook

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Overview of State Legislative Activity § Background Enacted Laws § Reporting § § § Registration/certification Minnesota (1993) California (2004) Vermont (2005) Nevada (2007) Maine (2004) District of Columbia (2004) West Virginia (2004)

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Overview of State Legislative Activity • Bans – Minnesota—prohibits gifts of $50 or more • Exceptions: – Samples, conferences, honoraria, and faculty. – Device companies that distribute drugs as an incidental part of business • Separate MN provision requiring annual report of payments totaling $100 or more to individual practitioners • Reporting Provisions – DC, ME, and VT require reporting of gifts over $25. 00

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Registration Requirements • California SB 1765 Signed by Governor 9/29/04 • Requires a pharmaceutical company to adopt a comprehensive compliance program in accord with the OIG guidelines • Companies are to include provisions for complying with the Ph. RMA Code • CCP to include limits on gifts or incentives provided to medical or health professionals • CCP to include a specific dollar limit for gifts to individual physicians • Exempts samples, conference support, and scholarships • Defines “dangerous drug” as drugs and device(s) that may be dispensed only by prescription

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Registration Requirements • Nevada – Requires drug and device manufacturers and wholesalers to adopt a marketing code of conduct that addresses: • Basis for interactions, informational presentations, thirdparty educational meetings, use of consultants, scholarships, and educational items • By June 1 every year, device firms are to notify the NV Board of Pharmacy of their code of conduct and should indicate if they are adhering to the Adva. Med Code.

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Overview of State Legislative Activity • Recent and Current Activity ▪ District of Columbia Safe RX Act of 2007 ▪ Massachusetts S 2535 ▪ Washington ▪ New Jersey Attorney General Task Force on Physician Compensation ▪ New Hampshire HB 1518 ▪ Tennessee HB 3602/HB 2677 Who and What’s Driving It ▪ Consumers Union, AARP, NLARx, Ortho Outlook

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Discussion of Physician Payment Sunshine Legislation Presented by Julie Cohen Adva. Med

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Environment on Capitol Hill • Democratic Majority in Congress made pharmaceutical pricing, influence, safety and transparency a top priority issue • Medical devices are increasingly included in scrutiny of industry practices. • Congressional Budget Office hearings/reports/statements about the influence of medical technology in driving overall health care cost increases • Oversight hearings on FDA’s ability to carry out its mission • Passage of 2007 Ethics/Lobbying Disclosure Legislation

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Recent Congressional Activity on Gift Disclosure • June 2007 – Senate Aging Committee hearing on Drug Industry Payments to Physicians – chaired by Senator Herb Kohl (D-WI) • July 2007 – Rep. Peter Defazio (D-OR) introduced H. R. 3023, Drug and Medical Device Company Gift Disclosure Act • September 2007 – Senators Grassley (R-IA) and Kohl introduced S. 2029, Physician Payments Sunshine Act • Efforts by Grassley-Kohl to include “Sunshine” Act in Medicare legislation in Fall 2007 • February 27, 2008 – Senate Aging Committee hearing on Device Industry Payments to Physicians

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Grassley-Kohl legislation (S. 2029) • Requires all manufacturers of drugs, devices, and medical supplies, with annual gross revenues of more than $100 million, to disclose any payment above $25, quarterly and in an annual summary report • Must disclose all payments, including compensation, food, entertainment, gifts, trips or travel, discounted products, continuing medical education, product rebates, consulting fees or any other economic benefit as defined by the Secretary. Product rebates and discounts specifically included • Covered health care professionals include physicians or an entity a physician is employed by, has tenure with or an ownership interest • Exceptions include free drug samples for patients, payments for clinical trials • Penalties for non-compliance: minimum $10, 000 up to $100, 000 per violation

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Adva. Med’s Key Advocacy Points • Collaboration with health care professionals is essential to invent and improve medical devices, and to ensure health care professionals are trained to use devices safely and effectively • Adva. Med has embraced a strong Code of Ethics to preserve collaborations with health care professionals • With some key changes to the legislation – to ensure a fair and level playing field, provide clear and meaningful information to patients, and to preserve relationships that benefit patients and innovation – Adva. Med could support the legislation.

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Understanding Collaborative Relationships Between the Medical Device Industry and Physicians • Physicians are key partners in the development and improvement of medical technology - Innovation requires ongoing collaboration to improve technology for patients - Partners in invention, development, clinical testing, education/training, providing feedback for improvements • Physician training is a critical component in the safe and effective use of medical devices - Physician technique/skills are critical with medical devices. - Hands-on training is essential. • Physician collaboration with the device industry has led to ground -breaking advances in patient care

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Adva. Med Has Embraced a Strong Code of Ethics to Preserve Collaboration with Health Care Professionals • The medical device industry recognizes that strong ethical standards is critical to our ability to continue collaborative relationships • Our Code of Ethics, effective January 1, 2004, distinguishes bona fide interactions that benefit patients from those that may influence medical decisions inappropriately • We have aggressively promoted adoption of the code with members and health care professionals • An independent 2006 survey found nearly 100% compliance among members surveyed • The Code addresses training and education, consulting, support of thirdparty educational conferences, sales and promotion, gifts, grants and charitable contributions • Adva. Med member companies take the Code further with dedicated resources and additional compliance procedures

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Adva. Med’s Position on S. 2029 • Adva. Med supports appropriate disclosure of relationships between medical technology companies and physicians • We could support the legislation if key changes are made to ensure a fair and level playing field, provide clear and meaningful information to patients, and to preserve relationships that benefit patients and innovation – – Preemption: create one uniform, strong Federal standard Alternative threshold: companies that make more than $250, 000 in annual reportable payments to physicians should disclose – Physician-owned entities should be included – Context of relationships should be provided to patients

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Additional Legislative Changes Sought by Adva. Med • Instead of reporting “transfers of value, ” legislation should enumerate specific types of payments to be disclosed • Delayed disclosure of product development agreements/clinical trials to protect proprietary information • Exclude reporting of discounts, rebates, and other pricing information • Exclude reporting of warranties, technical support, reimbursement information, and device samples/models for provider and patient demonstration (continued)

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Additional Legislative Changes Sought by Adva. Med • Require reporting for education and training to be limited to the value of meals, travel, and compensation only – rather than imputing a value to the training itself • Provide two years before implementation to allow companies to develop or refine tracking systems • Require annual reporting instead of quarterly reporting • Cap penalties • Allow reporting of payments in dollar ranges or bands

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Legislative Prospects for 2008 • Sponsors are considering revisions to the legislation • New House bill expected to be introduced that more closely aligns with S. 2029 • Medicare legislation expected in first half of 2008 – sponsors are wellpositioned and will likely seek to attach S. 2029 • Adva. Med doing outreach to Hill offices to advocate for key recommendations to improve the legislation

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Discussion of Aggregate Spend Tracking Challenges – Key Operational Issues Presented by Vickie Mc. Cormick St. Jude Medical Djebit Ghosh Huron Consulting Group, LLC
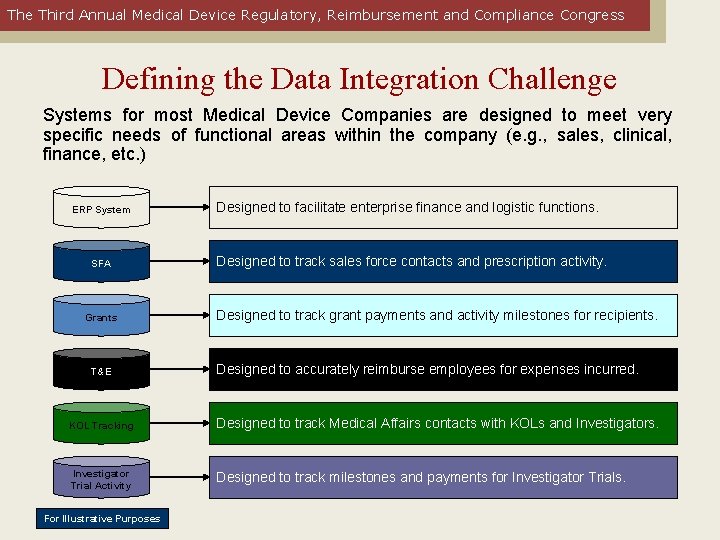
The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Defining the Data Integration Challenge Systems for most Medical Device Companies are designed to meet very specific needs of functional areas within the company (e. g. , sales, clinical, finance, etc. ) ERP System SFA Grants T&E Designed to facilitate enterprise finance and logistic functions. Designed to track sales force contacts and prescription activity. Designed to track grant payments and activity milestones for recipients. Designed to accurately reimburse employees for expenses incurred. KOL Tracking Designed to track Medical Affairs contacts with KOLs and Investigators. Investigator Trial Activity Designed to track milestones and payments for Investigator Trials. For Illustrative Purposes

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Changing the Paradigm for Data Tracking Measuring General Effects Accuracy and Completeness Key Points for Consideration • Tracking spending on healthcare professionals (“HCP”) requires accurate and complete data integration across key legacy systems • The need to integrate data at the transactional level across enterprise systems has not been a previous focus of medical device IT departments • Data across multiple systems cannot be accurately and completely integrated without the ability to identify transactions at the HCP level • Every transaction must be identifiable and attributable to an individual HCP for aggregation, tracking, and reporting (aggregate spend)

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Tracking and Disclosure is a “Bottom-Up” Process • The transactional data must be in a form that can be used for “aggregate spend tracking” purposes – If a record for a “trackable” expense does not have adequate and identifiable information (e. g. value, nature, purpose, recipient, identifier, etc. ), identifying the details of the transaction and the individual later in the process becomes significantly more difficult – Rather than receiving an accurate record of the HCP from the source system or the individual responsible for the expense or activity, a “downstream” system or process will have the burden of identifying the transaction • Solving the data integration challenge requires a data capture approach that will be sufficiently flexible, scalable, and adaptable to integrate transactional data across multiple source systems – Although an Enterprise Level Customer Master Strategy is generally the most effective approach, it is not always feasible – The cost of changing enterprise systems or adding functionality may be insurmountable – Disruption to company operations can range from being minimal with “buy-in” from functional areas or excessive if functional areas do not cooperate
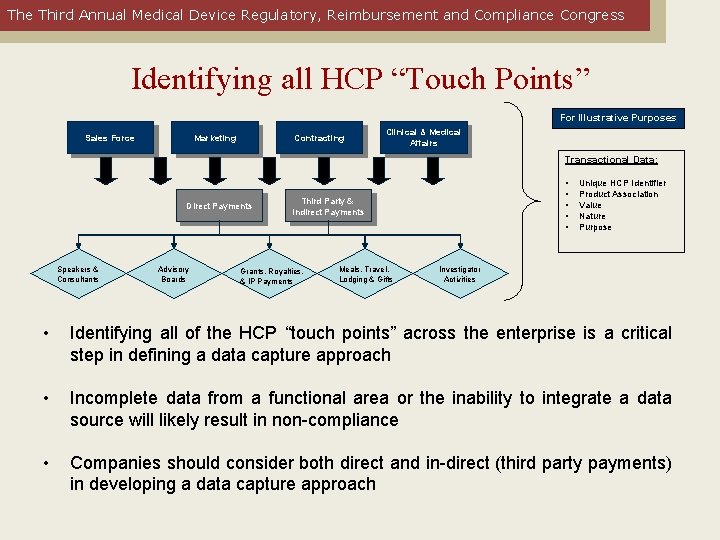
The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Identifying all HCP “Touch Points” For Illustrative Purposes Sales Force Marketing Commercial & Commercial Contracting & Managed Care Clinical&&Medical Affairs Transactional Data: Direct Payments Speakers & Consultants Advisory Boards • • • Third Party & Indirect. Payments Grants, Royalties, Grants & IP Payments Meals, Meals Travel, & Lodging Gifts& Gifts Unique HCP Identifier Product Association Value Nature Purpose Investigator Activities • Identifying all of the HCP “touch points” across the enterprise is a critical step in defining a data capture approach • Incomplete data from a functional area or the inability to integrate a data source will likely result in non-compliance • Companies should consider both direct and in-direct (third party payments) in developing a data capture approach
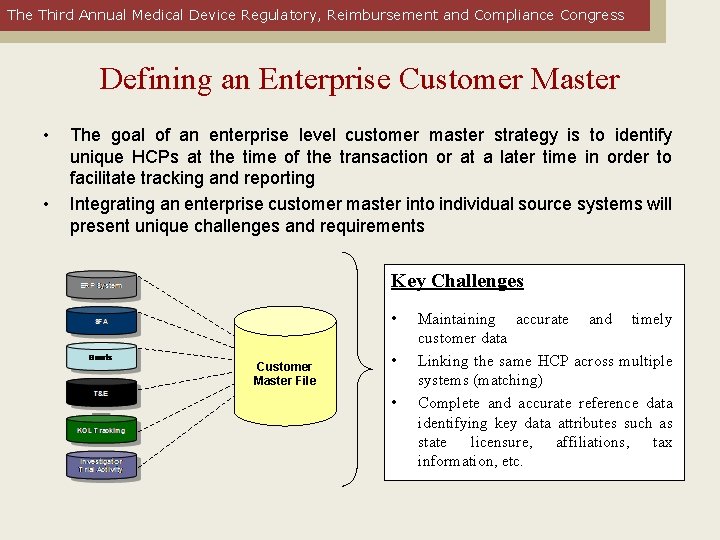
The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Defining an Enterprise Customer Master • • The goal of an enterprise level customer master strategy is to identify unique HCPs at the time of the transaction or at a later time in order to facilitate tracking and reporting Integrating an enterprise customer master into individual source systems will present unique challenges and requirements Key Challenges • Customer Master File • • Maintaining accurate and timely customer data Linking the same HCP across multiple systems (matching) Complete and accurate reference data identifying key data attributes such as state licensure, affiliations, tax information, etc.

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Key Considerations in Developing a ECM (1) • • Development and maintenance of a customer master file – Multiple practices – Institutional affiliations – Multiple addresses – Multiple Tax IDs – Name Changes – Multiple State License Numbers Managing multiple HCP types – Physicians – Mid-Levels (NP, PA, RPh, etc. ) – Hospitals and ambulatory surgical centers – Academic institutions

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Key Considerations in Developing a ECM (2) • • Managing history – Identifying data elements can change from year to year. – History should be maintained for audit and tracking purposes. – Develop “cross-walks” for multiple affiliations or duplicate records Use of reference data – Third party vendors – National Provider ID (HIPAA mandated)

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Interfacing an ECM with Enterprise Systems • • • ERP System – Typically accounts payable functions – Accurate name, address, and tax ID SFA System – “Call plan” synchronization – “Cross-walk” to sales data Grants System – Customer master data for institutions – “Back-end” linkage to HCP T&E System – Would require “current” data from customer master for HCP selection – Transactional level allocations KOL Tracking – Typically used only by Medical Affairs/MSL force – Tracking of fee-for-service activities Investigator Study Tracking – Part of an overall “aggregate spend tracking” strategy For Illustrative Purposes

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Challenges in Implementing Aggregate Spend Capture Processes

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Changing Systems and Business Processes • Effective data capture at the “front end” is the only reasonable strategy to improve accuracy and completeness of aggregate spend data – However, integrating a customer master strategy into existing legacy systems and business processes is a significant challenge • Existing processes and systems may not be able to support enhancements to core transaction sets that have been designed to meet very specific functional requirements: – ERP – T&E – KOL Contact Management Portals – Grants Management Systems – Investigator Study Management Systems • Organizations should assess their exposure and risk tolerance in assessing the need to change or enhance core systems or business processes – Pareto’s principle of “ 80/20” may apply in assessing exposure and risk
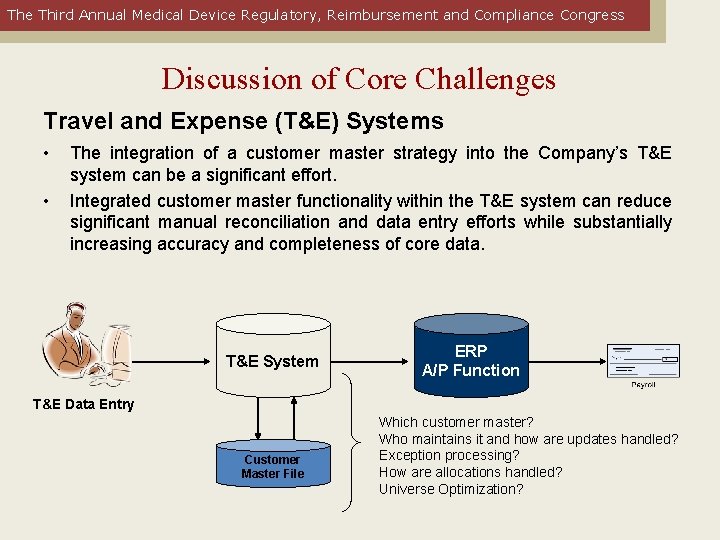
The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Discussion of Core Challenges Travel and Expense (T&E) Systems • • The integration of a customer master strategy into the Company’s T&E system can be a significant effort. Integrated customer master functionality within the T&E system can reduce significant manual reconciliation and data entry efforts while substantially increasing accuracy and completeness of core data. T&E System ERP A/P Function T&E Data Entry Customer Master File Which customer master? Who maintains it and how are updates handled? Exception processing? How are allocations handled? Universe Optimization?
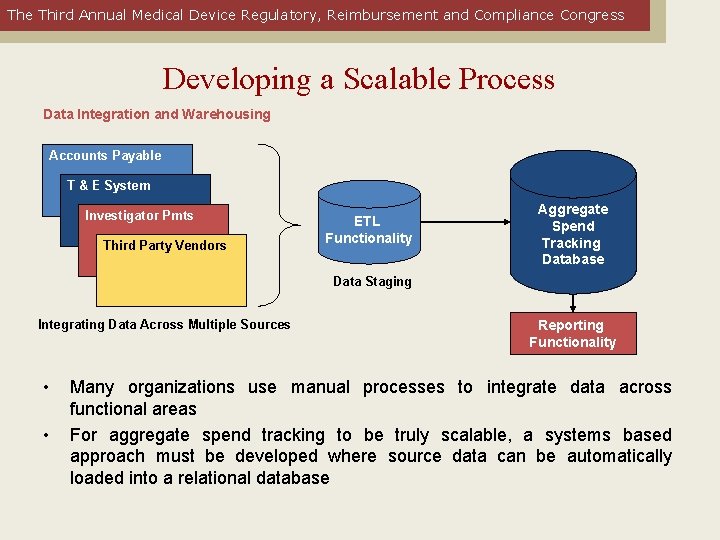
The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Developing a Scalable Process Data Integration and Warehousing Accounts Payable T & E System Investigator Pmts Third Party Vendors ETL Functionality Aggregate Spend Tracking Database Data Staging Integrating Data Across Multiple Sources • • Reporting Functionality Many organizations use manual processes to integrate data across functional areas For aggregate spend tracking to be truly scalable, a systems based approach must be developed where source data can be automatically loaded into a relational database
- Slides: 32