The Third Annual Medical Device Regulatory Reimbursement and

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Value-Based Pricing: The Good, The Bad, and The Ugly What Does Value Mean To Hospitals? March 27, 2008 Jeffrey C. Lerner, Ph. D. President and Chief Executive Officer ECRI Institute 5200 Butler Pike Plymouth Meeting, PA 19462

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress ECRI Institute The Discipline of Science. The Integrity of Independence. • • • The Institute is an independent nonprofit that researches the best approaches to improving patient care. It is designated as an Evidence-based Practice Center by the U. S. Agency for Healthcare Research(AHRQ), and is assigned to work on projects on behalf of CMS. Its assessments are also used by Medicaid programs and private insurers. ECRI Institute’s brand model laboratory-based evaluations of medical devices are used by thousands of hospitals and health agencies worldwide. The institute maintains offices in the U. K. , Malaysia and The United Arab Emirates. ECRI Institute is a Collaborating Center of the World Health Organization. Its more than 300 staff members include professionals from a great variety of scientific/clinical disciplines, as well as from the legal profession, health care planning and business.
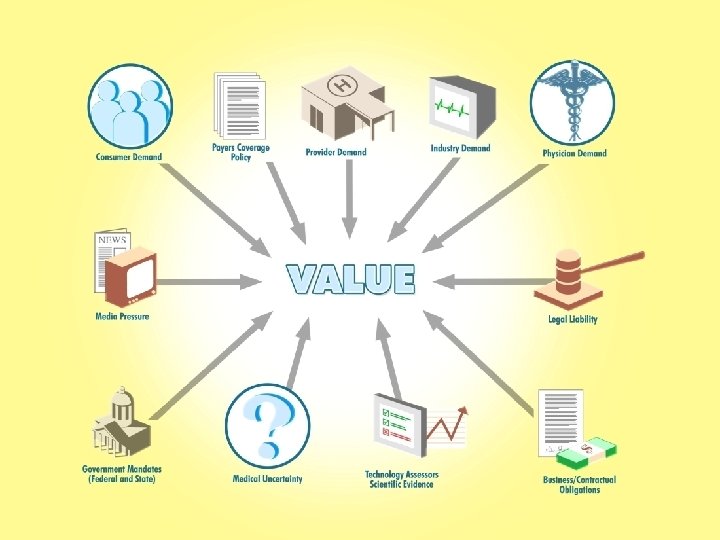
The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress
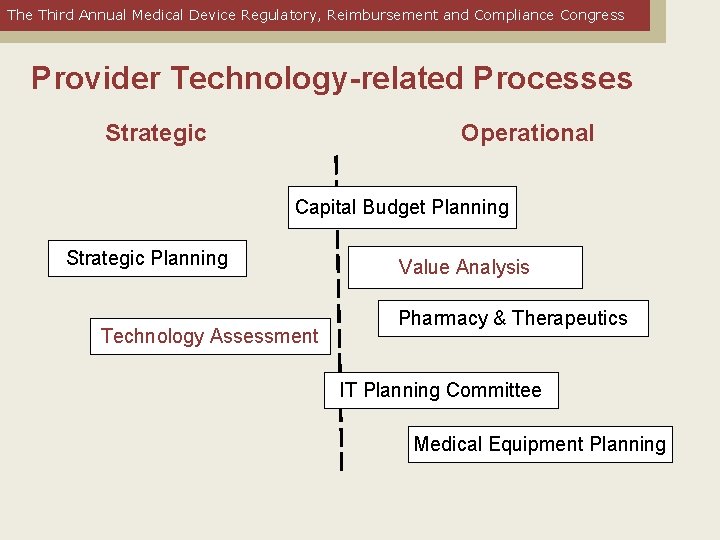
The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Provider Technology-related Processes Strategic Operational Capital Budget Planning Strategic Planning Technology Assessment Value Analysis Pharmacy & Therapeutics IT Planning Committee Medical Equipment Planning
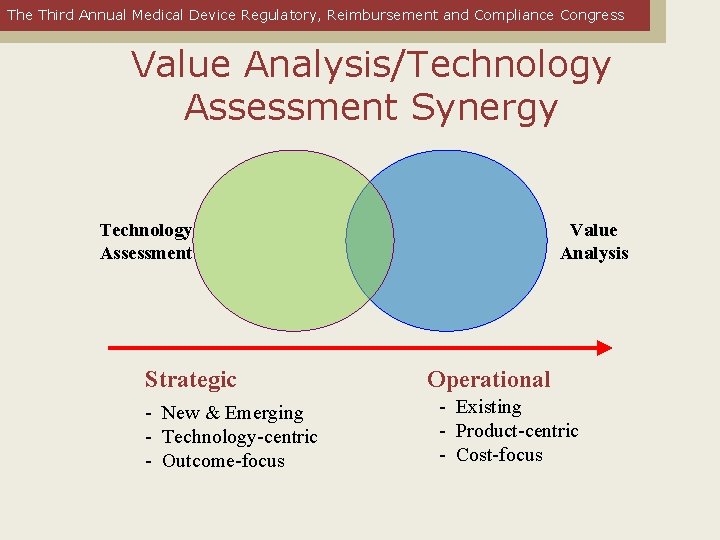
The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Value Analysis/Technology Assessment Synergy Technology Assessment Strategic - New & Emerging - Technology-centric - Outcome-focus Value Analysis Operational - Existing - Product-centric - Cost-focus

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress TA Case Study: Deep Brain Stimulation for Parkinson’s Disease Key discussion points: – – Quality of life evidence Medication therapy v DBS Total care requirements (e. g. , follow-up) Cost to transition from clinical trial to practice Result: – Recommended approval for a defined subset of Parkinson’s patients; not Essential Tremor patients
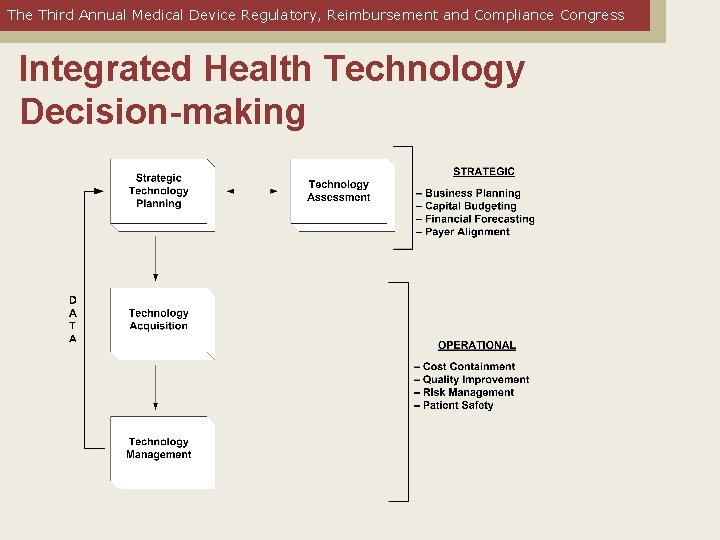
The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Integrated Health Technology Decision-making

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Some Best Practices for consideration: Build bridges between strategic & operational activities • Strategic technology planning Engage the empirical nature of physicians – Evidence-based; data driven Standardize criteria across assessment functions (e. g. , P&T, IT, TA, VAC) Ensure transparency of process & decisions Perform retrospective reviews of decisions – • •

The Third Annual Medical Device Regulatory, Reimbursement and Compliance Congress Some Best Practices for Consideration: Approve key technologies by indication; helps to limit (or begin to ID) “off-label” use 360° assessment considerations: • • – – – Patient safety Risk management Credentialing/Privileging Purchasing Contracting Reimbursement
- Slides: 9