The Synthesis of Carbon Nanotube on Activated Carbon

The Synthesis of Carbon Nanotube on Activated Carbon Prof. Dr. Sharifah Bee Abd Hamid, Imran Syakir Mohamad, Norli Abdullah, Ali Rinaldi Combinatorial Technology & Catalysis Research Centre (COMBICAT), Institute of Postgraduate Studies, University of Malaya, 50603 Kuala Lumpur, Malaysia.

Introduction • Why Activated Carbon? – Cheap and abundant resources – High thermal stability • Why Carbon-Carbon composites? – A new trend in catalysis; metal-free catalyst by exploiting the broad chemical functionality of carbon. – Carbon nanotube exhibit excellent activity and long term stability – However due to the compaction of CNTs bundle, it gives severe technical difficulties – Thus immobilizing CNTs on carbon supports (to avoid chemical discontunities) is a promissing solution
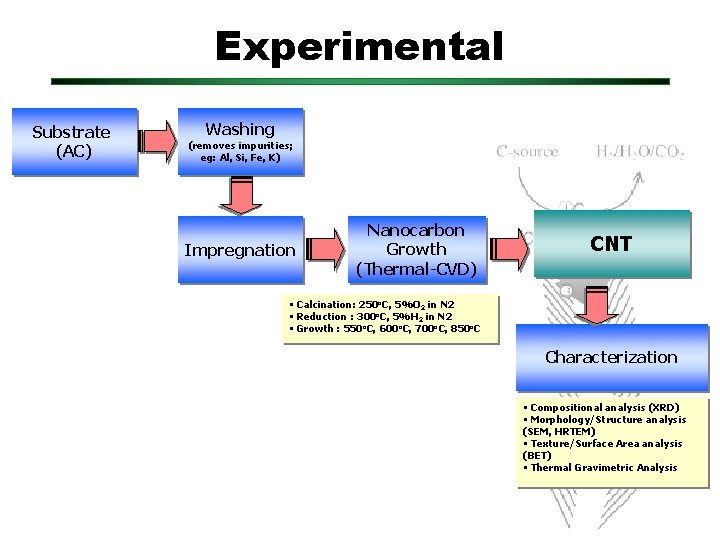
Experimental Substrate (AC) Washing (removes impurities; eg: Al, Si, Fe, K) Impregnation Nanocarbon Growth (Thermal-CVD) CNT • Calcination: 250 o. C, 5%O 2 in N 2 • Reduction : 300 o. C, 5%H 2 in N 2 • Growth : 550 o. C, 600 o. C, 700 o. C, 850 o. C Characterization • Compositional analysis (XRD) • Morphology/Structure analysis (SEM, HRTEM) • Texture/Surface Area analysis (BET) • Thermal Gravimetric Analysis
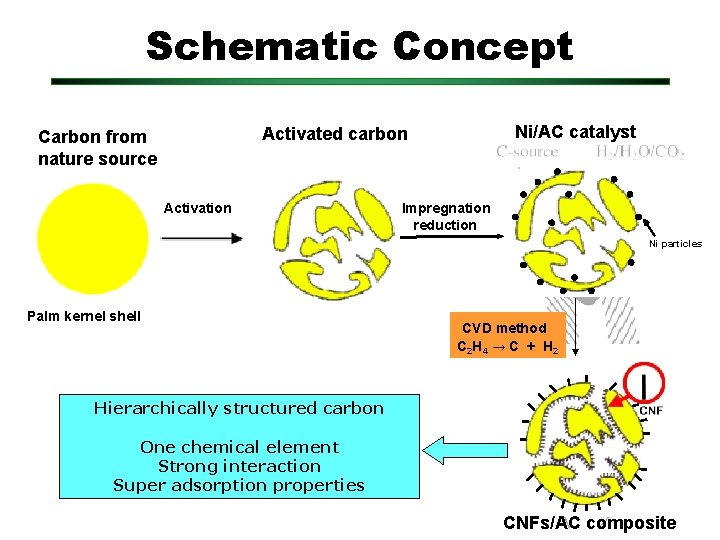
Schematic Concept Ni/AC catalyst Activated carbon Carbon from nature source Activation Impregnation reduction Ni particles Palm kernel shell CVD method C 2 H 4 → C + H 2 Hierarchically structured carbon One chemical element Strong interaction Super adsorption properties CNFs/AC composite
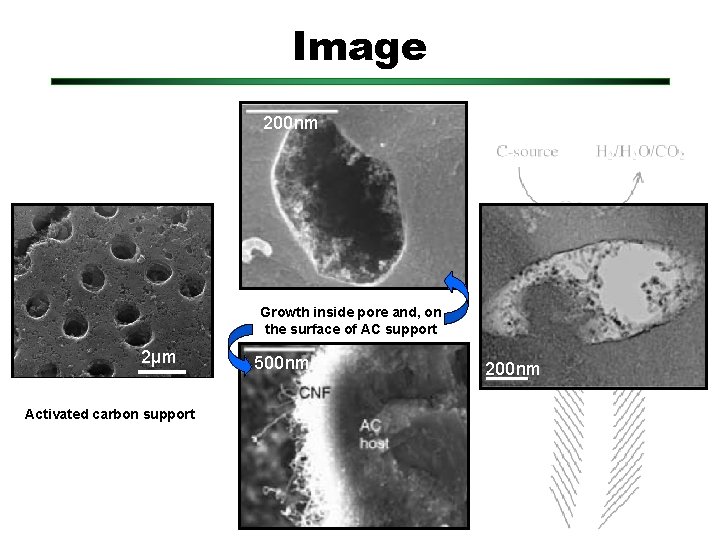
Image 200 nm Growth inside pore and, on the surface of AC support 2µm Activated carbon support 500 nm 200 nm

Result & Discussion • Ni particle on activated carbon support Ni catalyst Activated carbon support • The reduced-FFT d spacings measurements indicate the sample is fully reduced to Ni 0
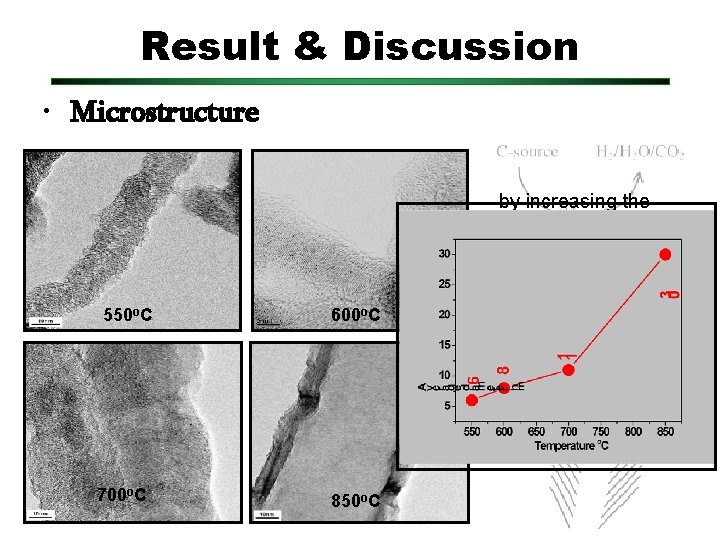
Result & Discussion • Microstructure by increasing the temperature growth; 550 o. C 600 o. C • Catalyst transform from solid to liquidlike behavior • different carbon diffusion in the catalyst • catalyst particle size also increase 700 o. C 850 o. C
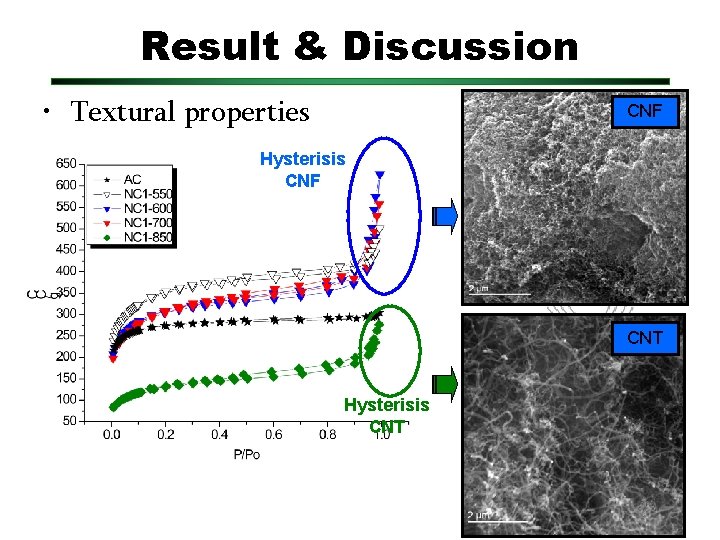
Result & Discussion • Textural properties CNF Hysterisis CNF CNT Hysterisis CNT
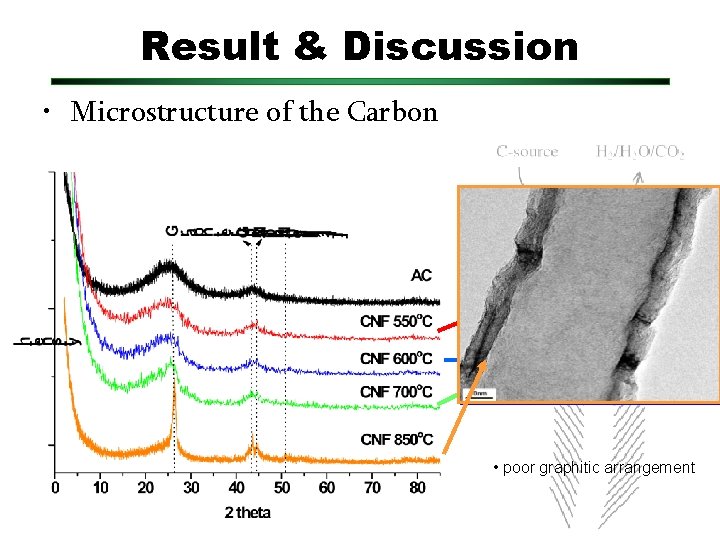
Result & Discussion • Microstructure of the Carbon • Increasing temperature, better graphitization • poor graphitic arrangement
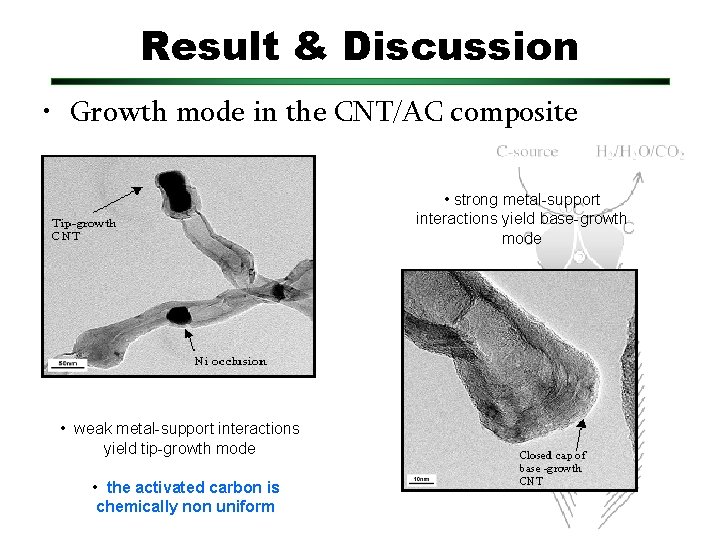
Result & Discussion • Growth mode in the CNT/AC composite • strong metal-support interactions yield base-growth mode • weak metal-support interactions yield tip-growth mode • the activated carbon is chemically non uniform
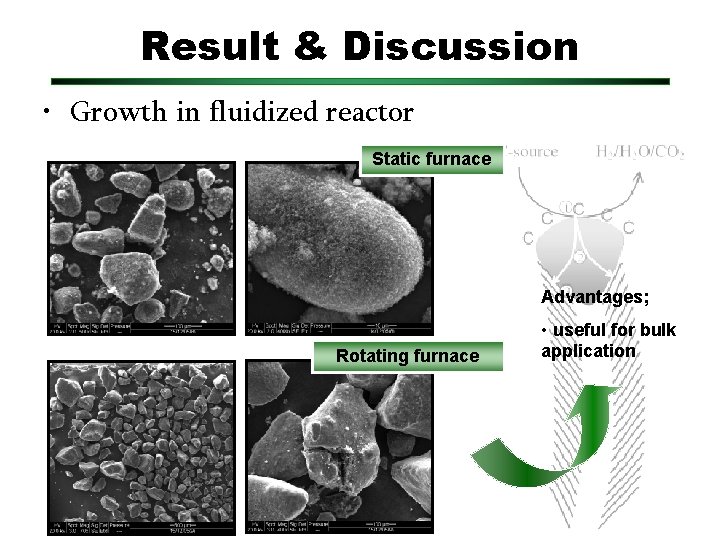
Result & Discussion • Growth in fluidized reactor Static furnace Advantages; Rotating furnace • useful for bulk application

Conclusion • Carbon nanotube has been successfully synthesized by Ni catalyst supported on activated carbon. • The CNF/CNT composition and morphology can be controlled by varying the temperature and fluidization of the catalyst support system during growth.

Carbon Team
- Slides: 13