The Surface Analysis Laboratory Cutting and Sputtering Getting

The Surface Analysis Laboratory Cutting and Sputtering: Getting to the Buried Interface John F Watts The Surface Analysis Laboratory Department of Mechanical Engineering Sciences 2 July 2014

The Problem! The Surface Analysis Laboratory
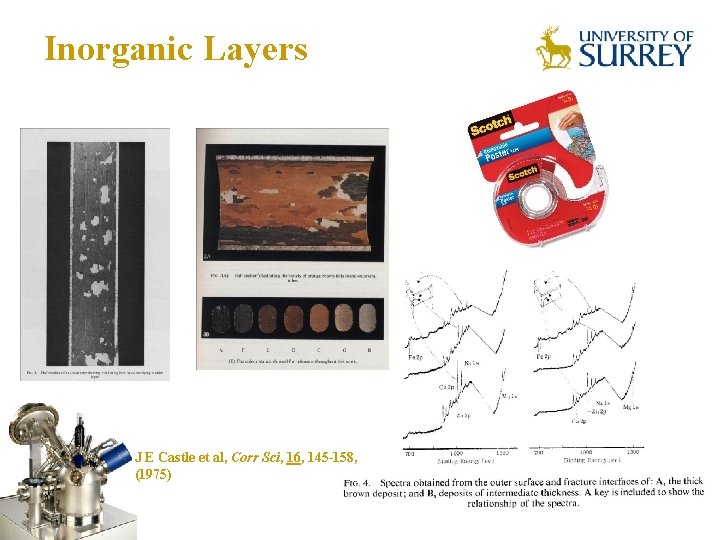
Inorganic Layers The Surface Analysis Laboratory J E Castle et al, Corr Sci, 16, 145 -158, (1975)
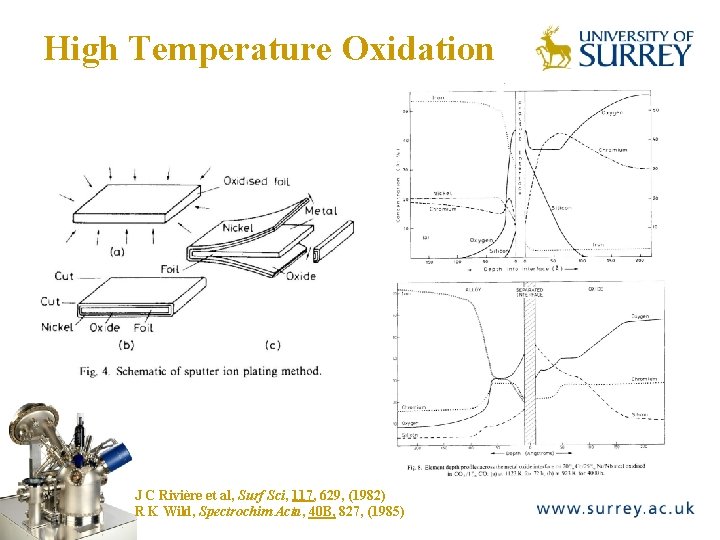
High Temperature Oxidation The Surface Analysis Laboratory J C Rivière et al, Surf Sci, 117, 629, (1982) R K Wild, Spectrochim Acta, 40 B, 827, (1985)
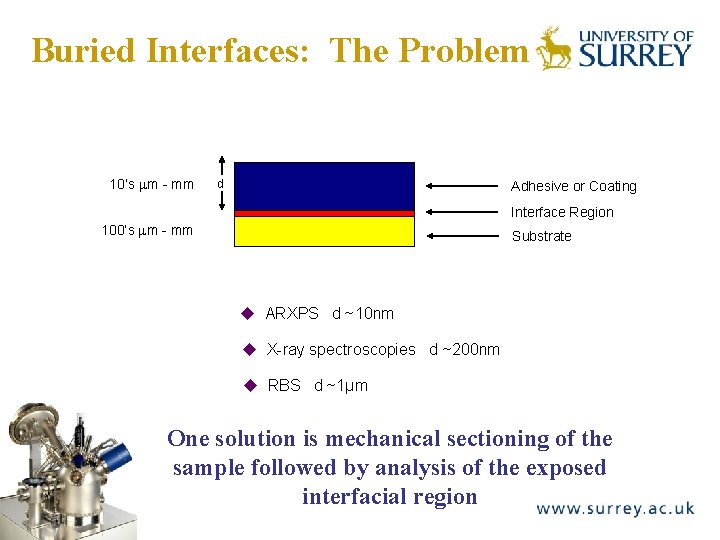
Buried Interfaces: The Problem The Surface Analysis Laboratory 10’s m - mm d Adhesive or Coating Interface Region 100’s m - mm Substrate ARXPS d ~10 nm X-ray spectroscopies d ~200 nm RBS d ~1μm One solution is mechanical sectioning of the sample followed by analysis of the exposed interfacial region
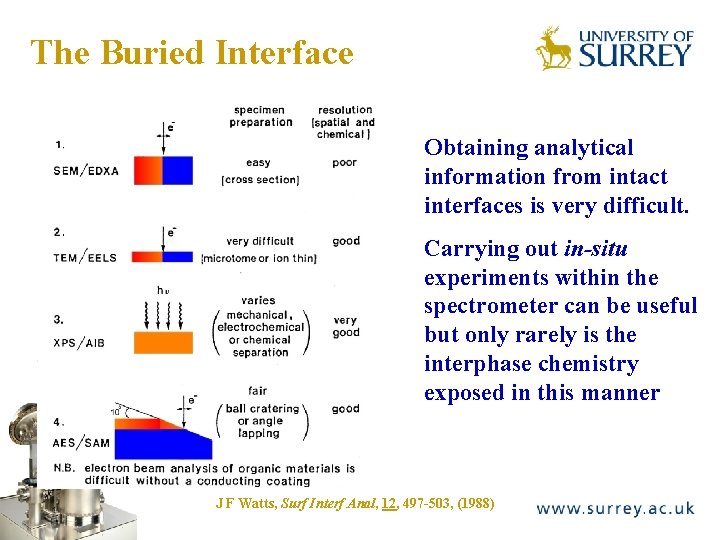
The Buried Interface The Surface Analysis Laboratory Obtaining analytical information from intact interfaces is very difficult. Carrying out in-situ experiments within the spectrometer can be useful but only rarely is the interphase chemistry exposed in this manner J F Watts, Surf Interf Anal, 12, 497 -503, (1988)

Oxide Stripping The Surface Analysis Laboratory Chemical removal of metal substrate, depth profiling of oxide in situ by ion sputtering. Interphase can then be analysed directly J F Watts, J E Castle, J Mat Sci, 18, 2987, (1983)
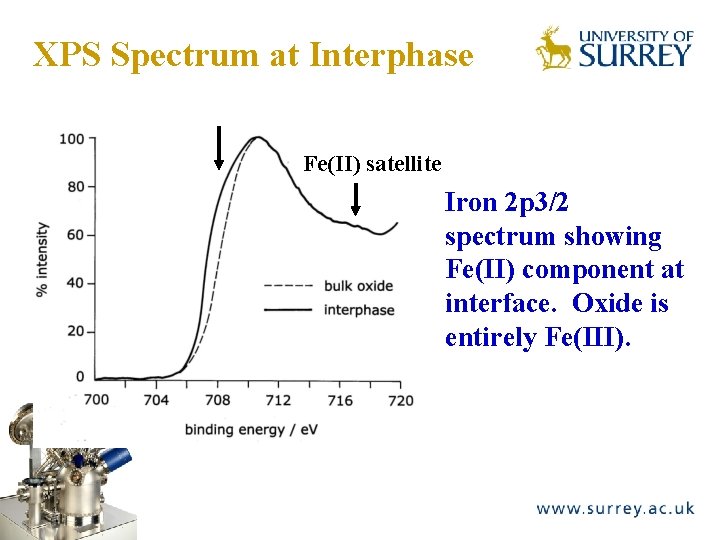
XPS Spectrum at Interphase The Surface Analysis Laboratory Fe(II) satellite Iron 2 p 3/2 spectrum showing Fe(II) component at interface. Oxide is entirely Fe(III).
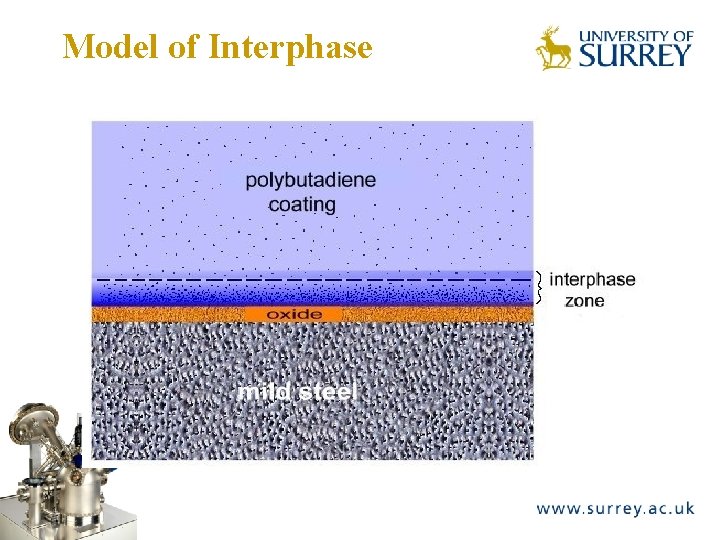
Model of Interphase The Surface Analysis Laboratory
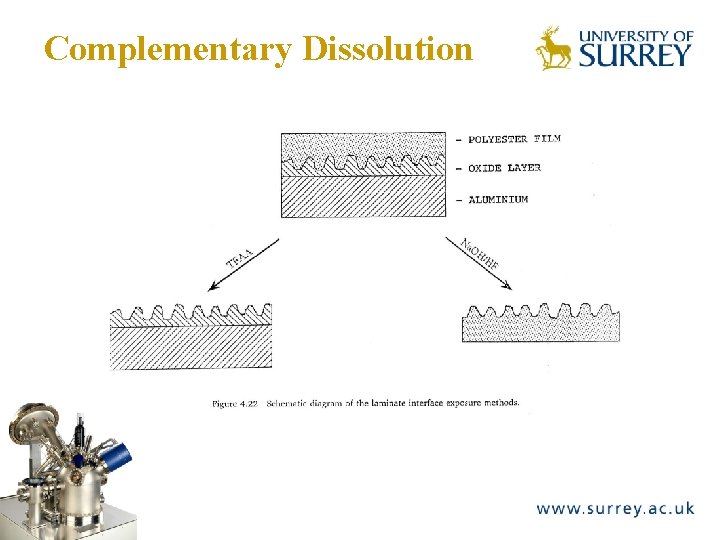
Complementary Dissolution The Surface Analysis Laboratory

Energy Filtered TEM The Surface Analysis Laboratory (a) (b) Energy-filtered (PEELS) TEM images of adhesively bonded aluminium showing the interpenetration of organic and oxide phase that is achieved when a primer is used (a). In the absence of a primer (b) the adhesive merely forms a interfacial boundary with the oxide. A J Kinloch, M Little, J F Watts, Acta Materialia, 48, 4543, (2000)

MICROM 355 S The Surface Analysis Laboratory

Ultra-Low Angle Microtomy The Surface Analysis Laboratory microtome blade sample angled sectioning block polyethylene Angle Sectioning Block 12 x 7 mm 3 + 25 mm = 0. 03 O + 50 mm = 0. 07 O + 100 mm = 0. 15 O + 200 mm = 0. 33 O
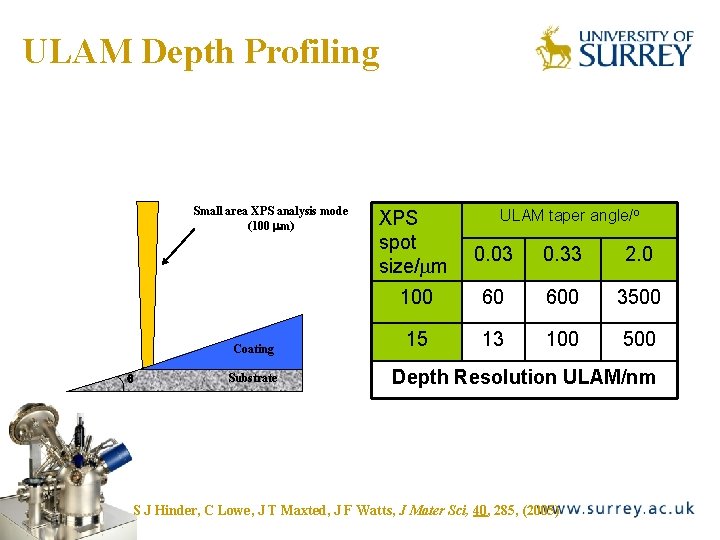
ULAM Depth Profiling The Surface Analysis Laboratory Small area XPS analysis mode (100 mm) Coating q Substrate XPS spot size/ m ULAM taper angle/o 0. 03 0. 33 2. 0 100 60 600 3500 15 13 100 500 Depth Resolution ULAM/nm S J Hinder, C Lowe, J T Maxted, J F Watts, J Mater Sci, 40, 285, (2005)
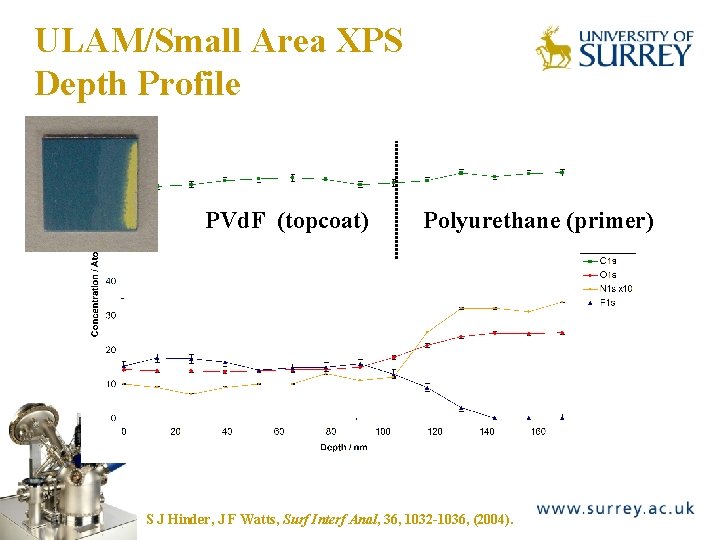
ULAM/Small Area XPS Depth Profile PVd. F (topcoat) The Surface Analysis Laboratory Polyurethane (primer) S J Hinder, J F Watts, Surf Interf Anal, 36, 1032 -1036, (2004).
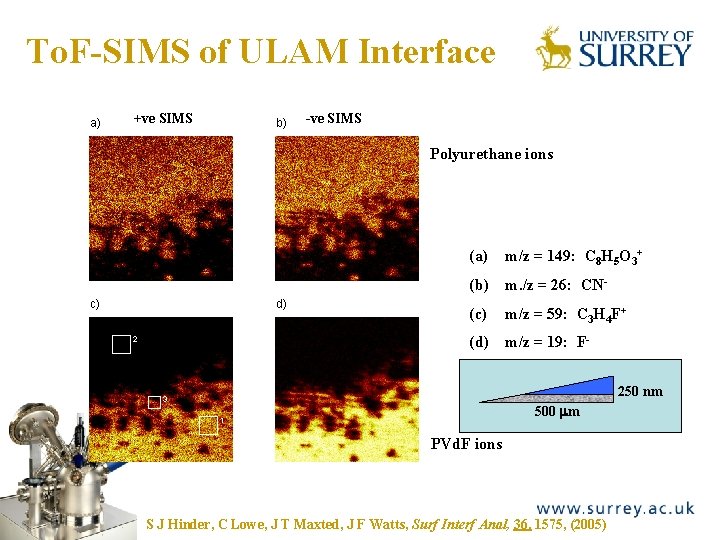
To. F-SIMS of ULAM Interface The Surface Analysis Laboratory a) +ve SIMS b) -ve SIMS Polyurethane ions (a) m/z = 149: C 8 H 5 O 3+ (b) m. /z = 26: CNd) c) (c) m/z = 59: C 3 H 4 F+ (d) m/z = 19: F- 2 250 nm 3 500 mm 1 PVd. F ions S J Hinder, C Lowe, J T Maxted, J F Watts, Surf Interf Anal, 36, 1575, (2005)
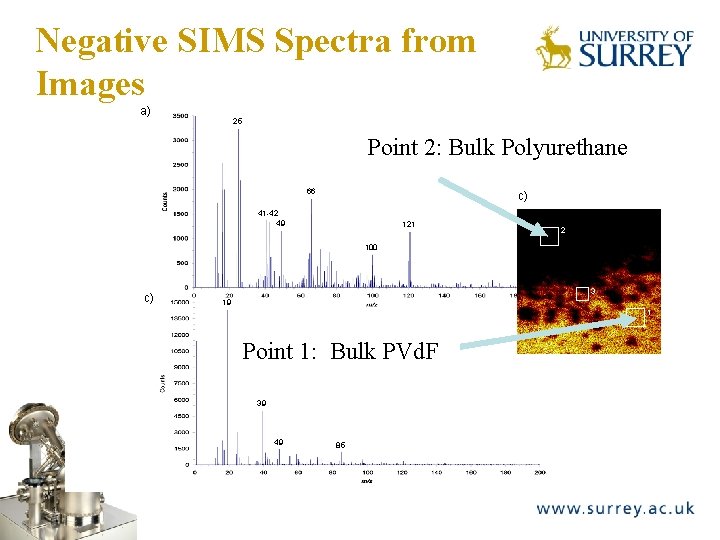
Negative SIMS Spectra from Images a) The Surface Analysis Laboratory 25 Point 2: Bulk Polyurethane 66 c) 41 -42 49 121 2 100 c) 3 19 1 Point 1: Bulk PVd. F 39 49 85
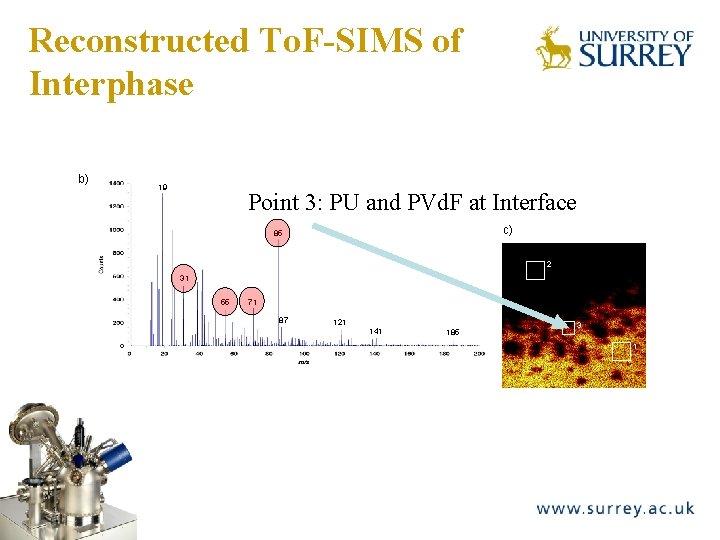
Reconstructed To. F-SIMS of Interphase b) 19 The Surface Analysis Laboratory Point 3: PU and PVd. F at Interface c) 85 2 31 55 71 87 121 141 185 3 1
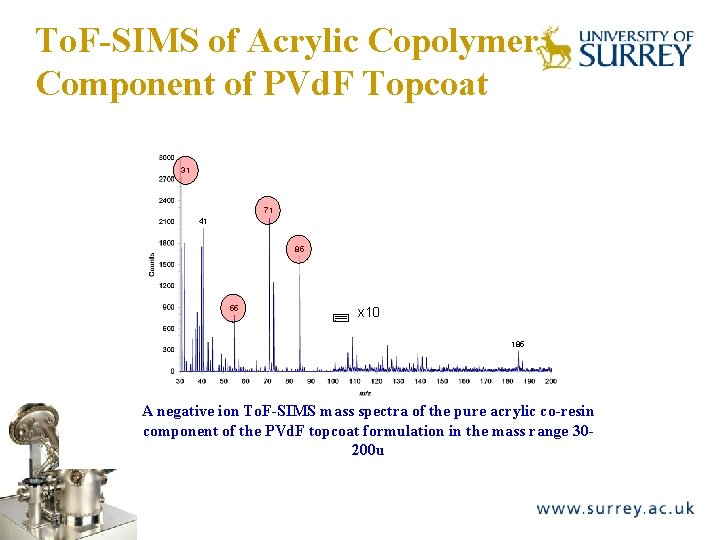
To. F-SIMS of Acrylic Copolymer Component of PVd. F Topcoat The Surface Analysis Laboratory 31 71 41 85 55 x 10 185 A negative ion To. F-SIMS mass spectra of the pure acrylic co-resin component of the PVd. F topcoat formulation in the mass range 30200 u
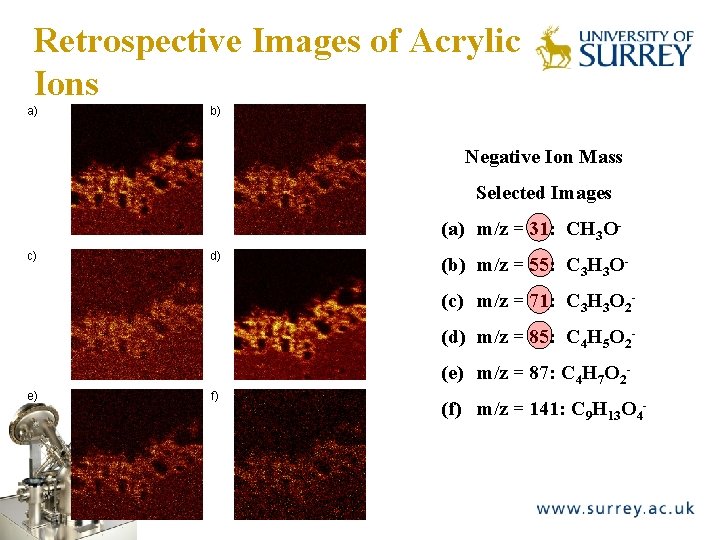
Retrospective Images of Acrylic Ions a) The Surface Analysis Laboratory b) Negative Ion Mass Selected Images (a) m/z = 31: CH 3 Oc) d) (b) m/z = 55: C 3 H 3 O(c) m/z = 71: C 3 H 3 O 2(d) m/z = 85: C 4 H 5 O 2(e) m/z = 87: C 4 H 7 O 2 - e) f) (f) m/z = 141: C 9 H 13 O 4 -
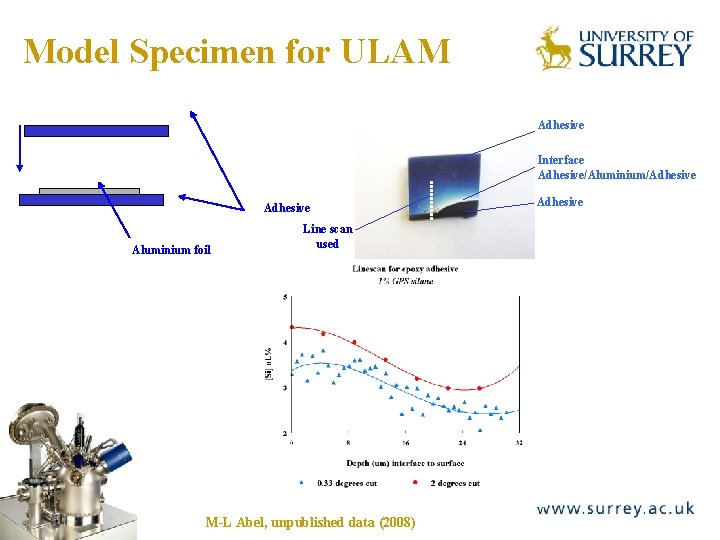
Model Specimen for ULAM The Surface Analysis Laboratory Adhesive Interface Adhesive/Aluminium/Adhesive Aluminium foil Line scan used M-L Abel, unpublished data (2008) Adhesive

Polyamide Powder Coating + Aminosilane addition The Surface Analysis Laboratory 100 mm thick thermoplastic polyamide powder coating with aminosilane added to the powder stock prior to spray coating ULAM is carried out on the intact outer surface to provide profile of air/coating interface and delaminated coating interfacial failure surface to provide steel/coating profile M Guichenuy, J F Watts, M-L Abel, M Audenaert, Surf Interf Anal, 38, 168 -171, (2006).
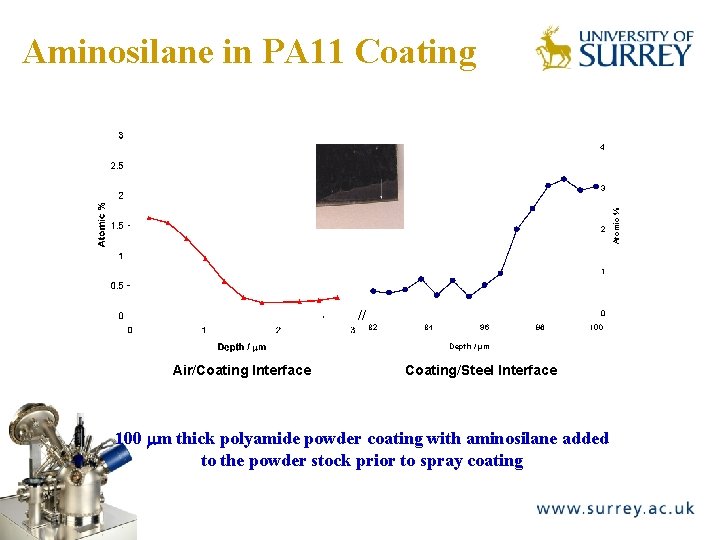
Aminosilane in PA 11 Coating The Surface Analysis Laboratory 4 2 1 // 0 Depth / μm Air/Coating Interface Coating/Steel Interface 100 mm thick polyamide powder coating with aminosilane added to the powder stock prior to spray coating Atomic % 3

Thin Film Solution 10’s m - mm d The Surface Analysis Laboratory Adhesive or Coating Interface Region 100’s m - mm Substrate Deposit a very thin layer of organic phase This may be from the plateau region of an adsorption isotherm Prepare specimen at monolayer coverage (i. e. plateau region) for XPS or To. F-SIMS analysis It is then possible to probe interface chemistry directly
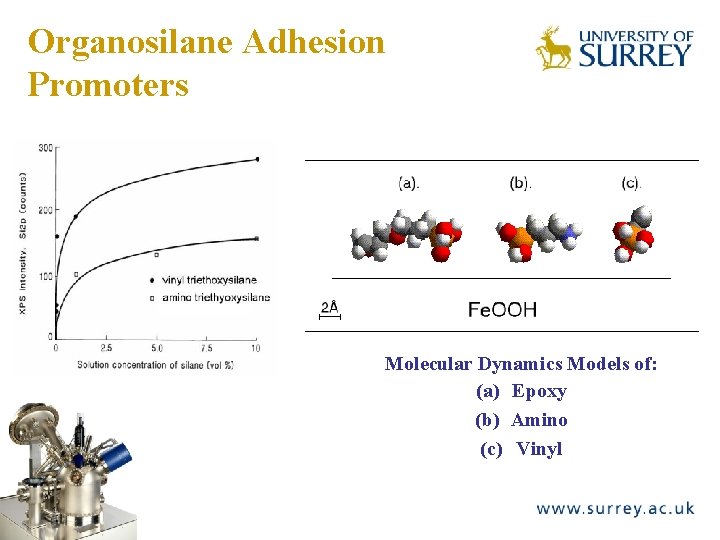
Organosilane Adhesion Promoters The Surface Analysis Laboratory Molecular Dynamics Models of: (a) Epoxy (b) Amino (c) Vinyl

To. F-SIMS to Identify Specific Interactions The Surface Analysis Laboratory The intense Si. OAl+ peak is indicative of a covalent bond between the aluminium oxide and the organosilane adhesion promoter

Conclusions § The Surface Analysis Laboratory
- Slides: 27