The Study of Noble Gas Noble Metal Halide

The Study of Noble Gas – Noble Metal Halide Interactions: Fourier Transform Microwave Spectroscopy of Xe. Cu. Cl Julie M. Michaud and Michael C. L. Gerry University of British Columbia, Vancouver, BC Canada June 2005 International Symposium on Molecular Spectroscopy

Xe. Cu. Cl Anticipated properties: § Short Xe-Cu bond length; § Small centrifugal distortion constant; § Large changes in nuclear quadrupole coupling constants (131 Xe, Cu and Cl) on bond formation; § Significant theoretical evidence of strong interactions between the Xe and Cu atoms in Xe. Cu. Cl. 2
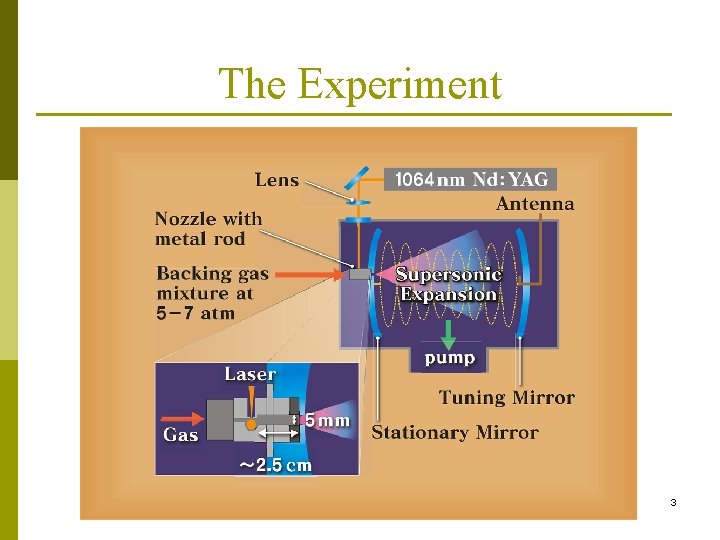
The Experiment 3
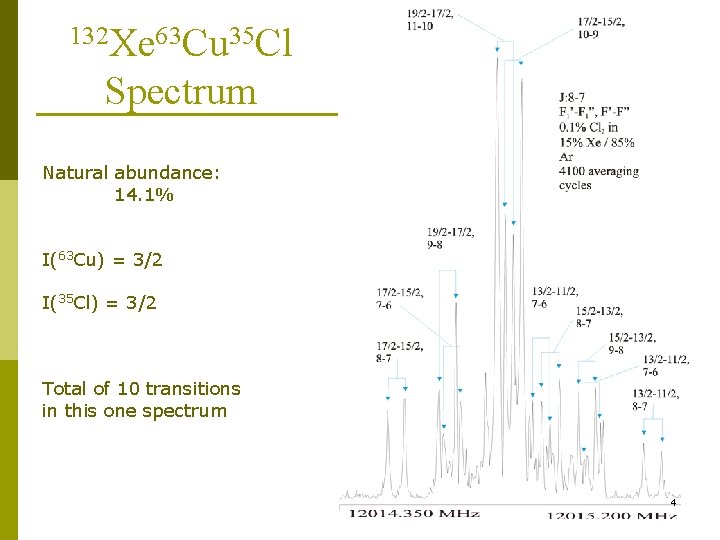
132 Xe 63 Cu 35 Cl Spectrum Natural abundance: 14. 1% I(63 Cu) = 3/2 I(35 Cl) = 3/2 Total of 10 transitions in this one spectrum 4
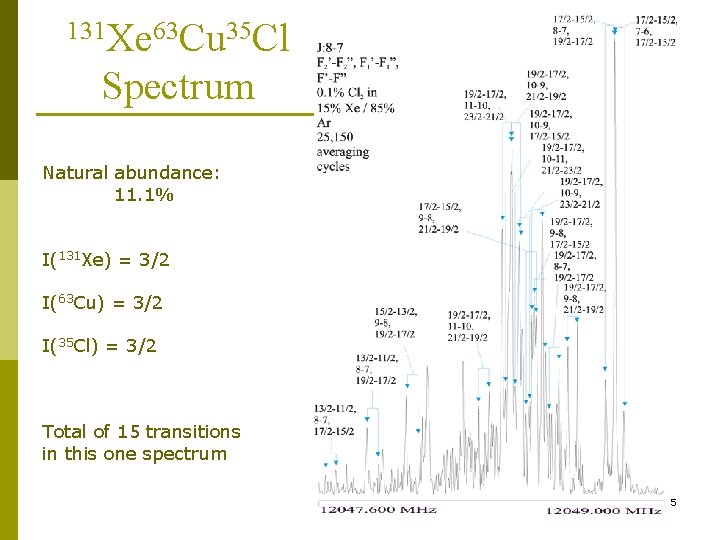
131 Xe 63 Cu 35 Cl Spectrum Natural abundance: 11. 1% I(131 Xe) = 3/2 I(63 Cu) = 3/2 I(35 Cl) = 3/2 Total of 15 transitions in this one spectrum 5
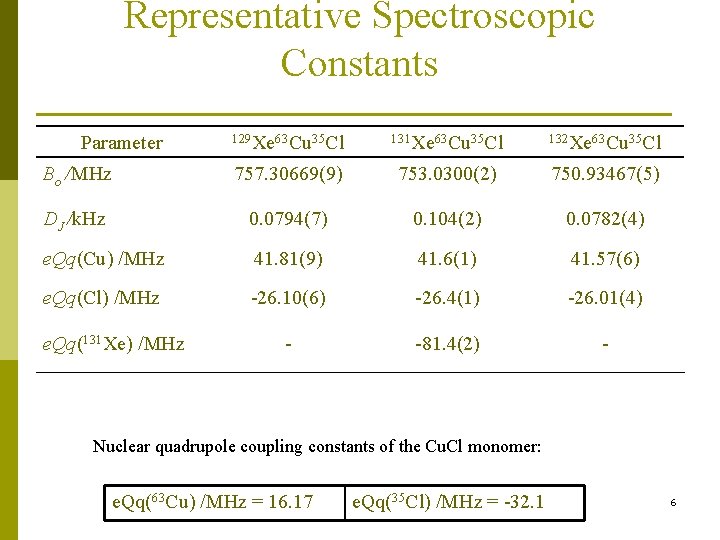
Representative Spectroscopic Constants Parameter 129 Xe 63 Cu 35 Cl 131 Xe 63 Cu 35 Cl 132 Xe 63 Cu 35 Cl Bo /MHz 757. 30669(9) 753. 0300(2) 750. 93467(5) DJ /k. Hz 0. 0794(7) 0. 104(2) 0. 0782(4) e. Qq(Cu) /MHz 41. 81(9) 41. 6(1) 41. 57(6) e. Qq(Cl) /MHz -26. 10(6) -26. 4(1) -26. 01(4) - -81. 4(2) - e. Qq(131 Xe) /MHz Nuclear quadrupole coupling constants of the Cu. Cl monomer: e. Qq(63 Cu) /MHz = 16. 17 e. Qq(35 Cl) /MHz = -32. 1 6

Xe-Cu bond lengths in Xe. Cu. Cl Experimental rm(2)(Xe-Cu) = 2. 4669(4) Å p MP 2 results r(Xe-Cu) = 2. 497 Å a p p van der Waals limit: (rvd. W(Xe)b + rion(Cu+)c) = 2. 78 Å p Covalent limit: (rcov(Xe)d + rcov(Cu(I))e) = 2. 36 Å Lovallo, C. C. ; Klobukowski, M. Chem. Phys. Lett. 2002, 368, 589. Pyykkö, P. Chem. Rev. 1997, 597. c Huheey, J. E. et al. Inorganic Chemistry, Principles of Structure and Reactivity, 4 th Ed. ; Harper-Collins: New York, 1993. d Bartlett, N et al. In Comprehensive Inorganic Chemistry; Bailar, J. C. et al. Eds. ; Pergamon: Oxford, 1973; 213 -330. e Pyykkö, P. Chem. Rev. 1988, 579. a b 7

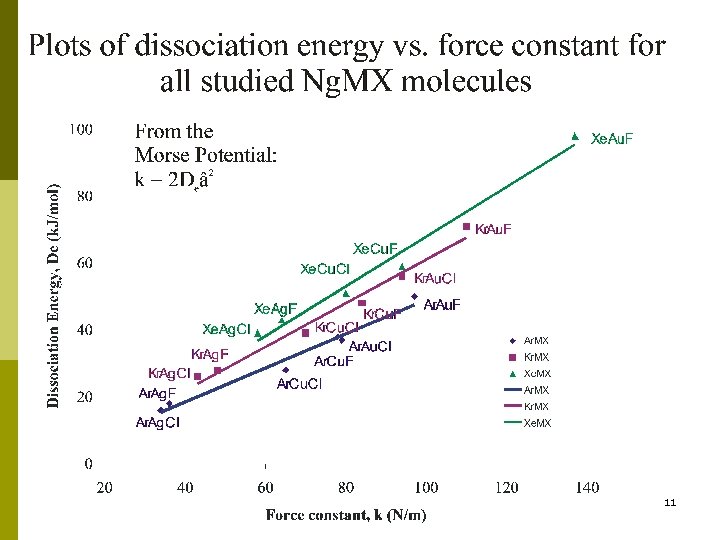
Centrifugal Distortion Constants Small centrifugal distortion constants Highly rigid molecules Xe. Cu. F DJ /k. Hz X 102 20 k(Ng-M) /Nm-1 94 Xe. Au. F 7 137 Ar. Ag. Cl 35 34 Ar-Na. Cla 900 0. 6 8 a Mizoguchi, A. ; Endo, Y. ; Ohshima, Y. J. Chem. Phys. 1998, 109, 10539.
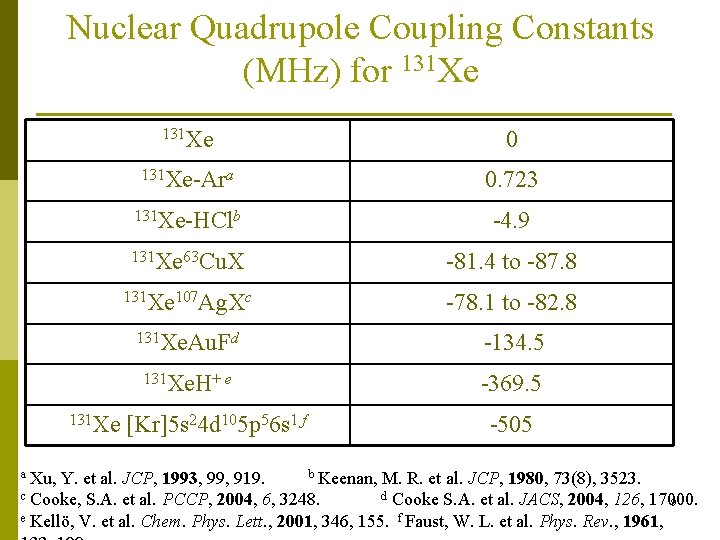
Nuclear Quadrupole Coupling Constants (MHz) for 131 Xe a Xu, 131 Xe 0 131 Xe-Ara 0. 723 131 Xe-HClb -4. 9 131 Xe 63 Cu. X -81. 4 to -87. 8 131 Xe 107 Ag. Xc -78. 1 to -82. 8 131 Xe. Au. Fd -134. 5 131 Xe. H+ e -369. 5 [Kr]5 s 24 d 105 p 56 s 1 f -505 b Keenan, M. R. et al. JCP, 1980, 73(8), 3523. Y. et al. JCP, 1993, 99, 919. c Cooke, S. A. et al. PCCP, 2004, 6, 3248. d Cooke S. A. et al. JACS, 2004, 126, 17000. 9 e Kellö, V. et al. Chem. Phys. Lett. , 2001, 346, 155. f Faust, W. L. et al. Phys. Rev. , 1961,

Ab initio calculations p. A large dissociation energy was calculated for Xe. Cu. Cl (55 k. J mol-1); p Charge-induced dipole induction energy for Xe. Cu. Cl: 21 k. J mol 1; p Mulliken and NBO populations show significant donation of electron density from the Xe to the Cu; p Donation of ~0. 1 -0. 2 of an elementary charge donated from Xe to Cu. 10
11
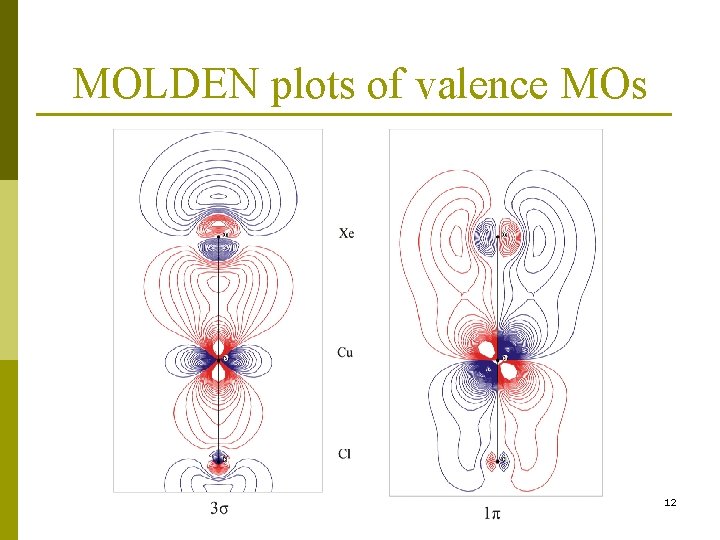
MOLDEN plots of valence MOs 12

Local Energy Densitiesa H(r) = G(r) + V(r) Values are calculated at the bond critical point of the maximum electron density (MED) path between bonded atoms. Negative H(r) implies … V(r) dominates … e- density accumulates at r. B … a covalent bond forms!! a Cremer, D. ; Kraka, E. Angew. Chem. 1984, 96, 612; see also Angew. Chem. Int. Ed. Engl. , 1984, 23, 627. 13
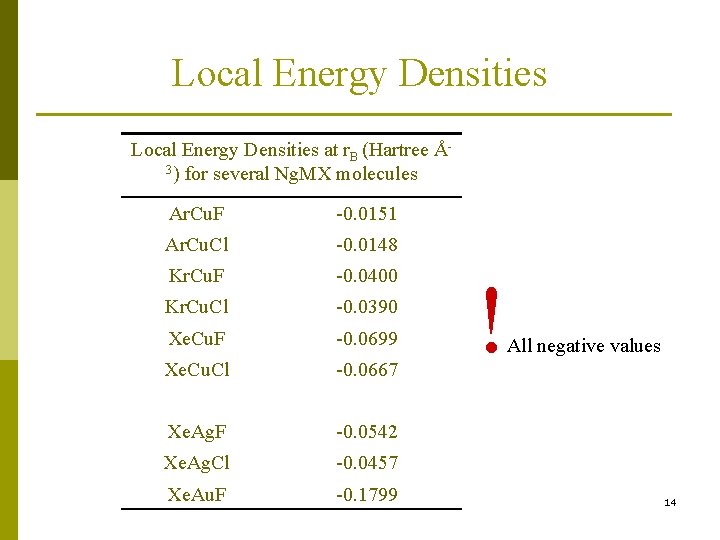
Local Energy Densities at r. B (Hartree Å3) for several Ng. MX molecules Ar. Cu. F -0. 0151 Ar. Cu. Cl -0. 0148 Kr. Cu. F -0. 0400 Kr. Cu. Cl -0. 0390 Xe. Cu. F -0. 0699 Xe. Cu. Cl -0. 0667 Xe. Ag. F -0. 0542 Xe. Ag. Cl -0. 0457 Xe. Au. F -0. 1799 ! All negative values 14

Conclusion First preparation and characterization Xe. Cu. Cl; p Very strong Xe-Cu interactions observed; p Strong Ng-M interactions similar to those expected from previous Ng. MX studies; p Xe. Cu. Cl shows convincing evidence of covalent Xe. Cu bonding. p 15

Acknowledgements Thank you to Mike Gerry, Steve Cooke and Christine Krumrey. This research has been supported by the Natural Sciences and Engineering Research Council (NSERC) of Canada. Thank you for your attention. 16
- Slides: 16