The Structure of the Atom Atomic Theory Democritus














- Slides: 21

The Structure of the Atom

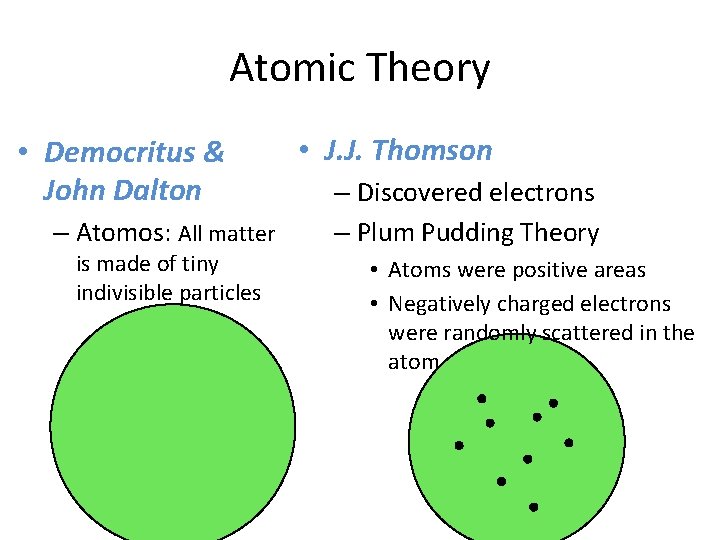
Atomic Theory • Democritus & John Dalton – Atomos: All matter is made of tiny indivisible particles • J. J. Thomson – Discovered electrons – Plum Pudding Theory • Atoms were positive areas • Negatively charged electrons were randomly scattered in the atom

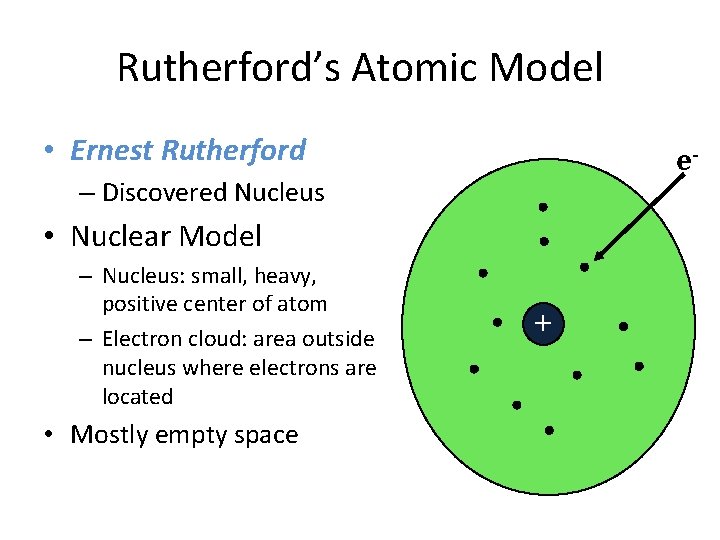
Rutherford’s Atomic Model • Ernest Rutherford e- – Discovered Nucleus • Nuclear Model – Nucleus: small, heavy, positive center of atom – Electron cloud: area outside nucleus where electrons are located • Mostly empty space +

Subatomic Particles • Electron – negatively charged particle, smallest, light, in electron cloud • Proton - positively charged pieces, heavy, in nucleus • Neutron - no charge, same mass as protons, in nucleus

Subatomic particles Name Relative Symbol Charge mass Electron e- -1 1/1840 Proton p+ +1 1 Neutron n 0 0 1 ***Relative Mass means compared to. *** A proton is 1840 times the mass of an electron.

How Atoms Differ

Counting the Pieces • Atomic Number = p+ – determines kind of atom (fingerprint) • Mass Number = p+ + n 0 • Number of e- = # of p+ – when it’s a neutral atom (no charge)

From periodic table • Using the example – 11 is the atomic number • There are 11 p+ in Sodium – Na is the element symbol – Sodium is the element name – 22. 99 is the avg. atomic mass • This is NOT the mass number!!! 11 Na Sodium 22. 99

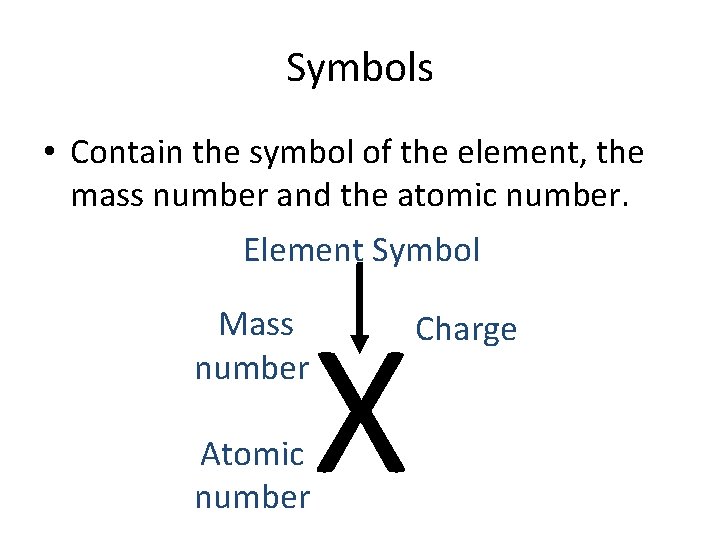
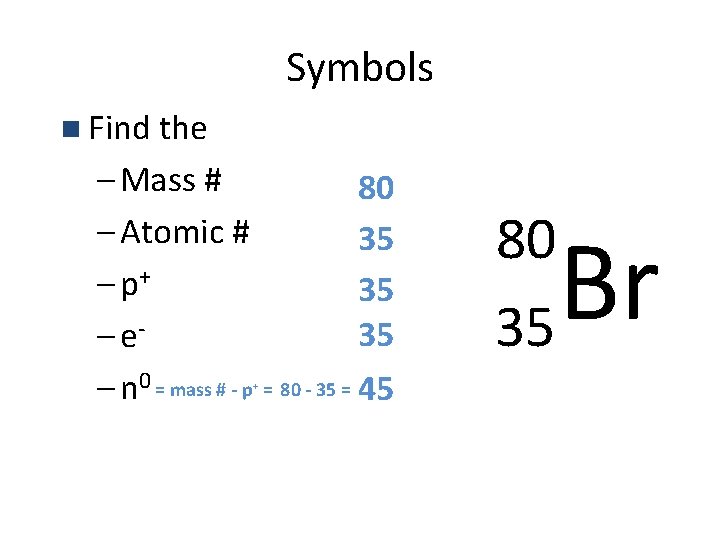
Symbols • Contain the symbol of the element, the mass number and the atomic number. Element Symbol Mass number Atomic number X Charge

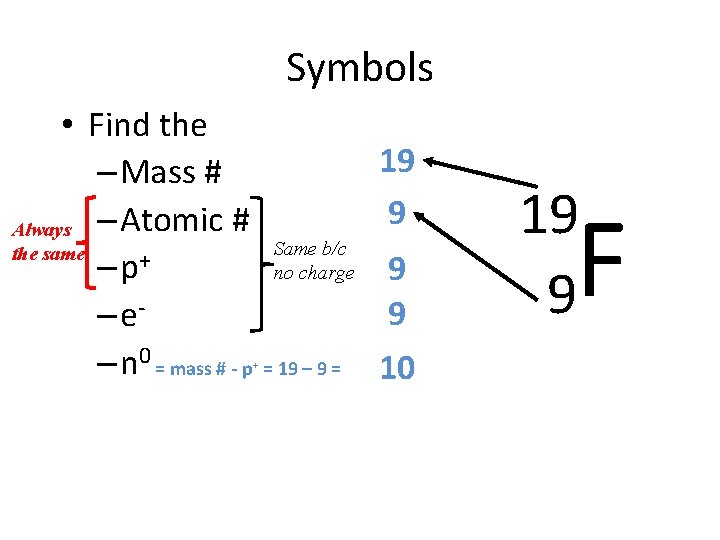
Symbols • Find the – Mass # Always – Atomic # the same – p+ – e– n 0 = mass # - p 19 9 Same b/c no charge + = 19 – 9 = 9 9 10 19 9 F

Symbols n Find the – Mass # – Atomic # – p+ – e– n 0 = mass # - p + 80 35 35 35 = 80 - 35 = 45 80 35 Br

Symbols n A neutral element has an atomic number of 34 and a mass number of 78. What is the – number of p+? 34 Same as atomic # – number of 78 n 0 - 34= ? 44 – number of e-? 34 Same as p – Complete symbol. + 78 34 Se

Symbols n A neutral element has 91 protons and 140 neutrons what is the 91 – Atomic #? Same as p 231 – Mass #? p + n = 91 + 140 – # of electrons? 91 Same as p – Complete symbol. + + 0 + 231 91 Pa

Isotopic Notation • Contain the symbol of the element, the mass number and the atomic number. All are neutral (no charge) Mass # Element Name-# • Example: Sodium-24

Isotopic Notation n Find the – Mass # 24 – Atomic # 11 – p+ 11 11 – e– n 0 = mass # - p = 24 – 11 = 13 + Sodium -24

Isotopic Notation n Find the – Mass # 56 – Atomic # 28 – p+ 28 28 – e– n 0 = mass # - p = 56 – 28 = 28 + Nickel -56

Charges • If Neutral: Atomic # = p+ = e- • Electrons tell you charge of the atom: • Lose/Gain e-: changes charge, creates ions • # e- < # p+: cation, lose e-, positive ions • # e- > # p+: anion, gain e-, negative ions

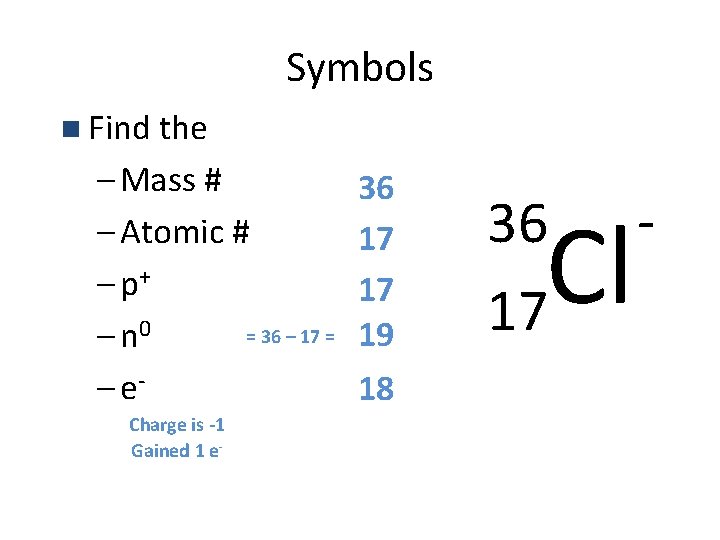
Symbols n Find the – Mass # – Atomic # – p+ = 36 – 17 = – n 0 – e. Charge is -1 Gained 1 e- 36 17 17 19 18 36 17 Cl -

Symbols n Find the – Mass # – Atomic # – p+ = 42 – 20 = – n 0 p - 2 = 20 - 2= – e+ Charge is +2 Lost 2 e- 42 20 20 22 18 42 20 +2 Ca

Symbols n An element has 76 electrons and 117 neutrons, and a charge of +2, what is the 78 – Atomic #? Charge is +2, lost 2 e , e + 2 = 76 + 2 = – Mass #? 195 p + n = 117 + 78 – # of protons? 78 same as atomic # – Complete symbol. - + 0 - 195 78 +2 Pt

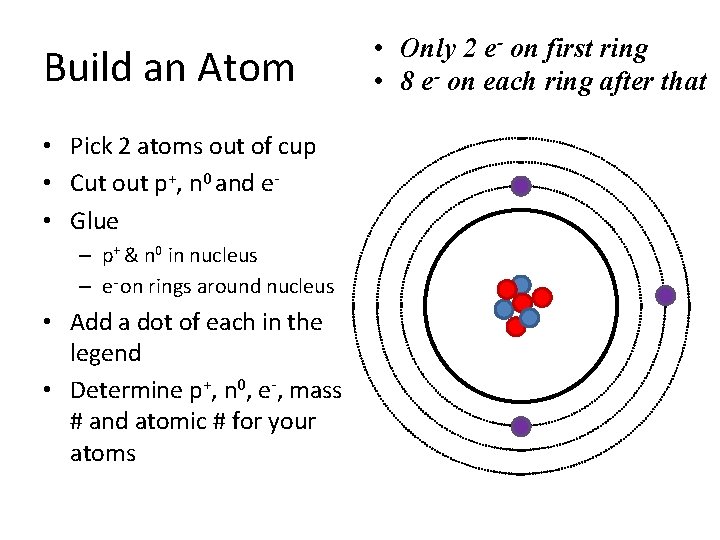
Build an Atom • Pick 2 atoms out of cup • Cut out p+, n 0 and e • Glue – p+ & n 0 in nucleus – e- on rings around nucleus • Add a dot of each in the legend • Determine p+, n 0, e-, mass # and atomic # for your atoms • Only 2 e- on first ring • 8 e- on each ring after that