The solvent system The only solvent for sugars








- Slides: 10

The solvent system • The only solvent for sugars is water. • Hence, the only chromatographic solvents which are useful with these compounds contain water. • When watery solvent are used, it is necessary to remove most ions from the material being examined.

• Monosaccharides, however, often have to be released from various combinations in drugs by hydrolysis with acid; thus the problem is particularly obtaining neutral solutions without leaving soluble salts.

§Neutralization can be done in several ways: 1. Addition of excess barium carbonate to convert sulfuric acid to insoluble barium sulfate. 2. Passing the solution through a suitable OH exchanger so that the Cl- or SO 4 -- ions are replaced by OH-. 3. Shaking the acid solution with an organic solution of an amine whose salt are soluble in the organic solvent.

Identification of the monosaccharides produced by hydrolysis of acacia 1. Dissolve 2 g powdered acacia gum in 20 ml 20% v/v H 2 SO 4, and place the solution in boiling w. b for 15 minutes. 2. Transfer the solution to a large evaporating dish, and whilst heating gently and carefully over a flame to avoid charring, gradually add excess barium carbonate. 3. Add a little water if necessary and filter. The filtrate is a neutral solution containing sugars and should b examined by paper chromatography against suitable reference sugars

Materials needed for paper chromatography of sugars: 1. Filter paper: whatman No. 1 2. Solvent systems: • n- butanol-acetic acid-water(4: 2: 1) or(4: 1: 5) • prepared by shaking its components and is left to equilibrate.

• Spray reagent: 1. 2 -5% aniline phthalate in n-butanol saturated with water. 2. 0. 5% benzidine in glacial acetic acid.

• Reference sugars: • Arabinose, dextrose, galactose, rhamnose and xylose. • Arabinose and xylose give red colour with aniline phthalate spray reagent. • Galactose, glucose and rhamnose give yellow spots with the same reagent.

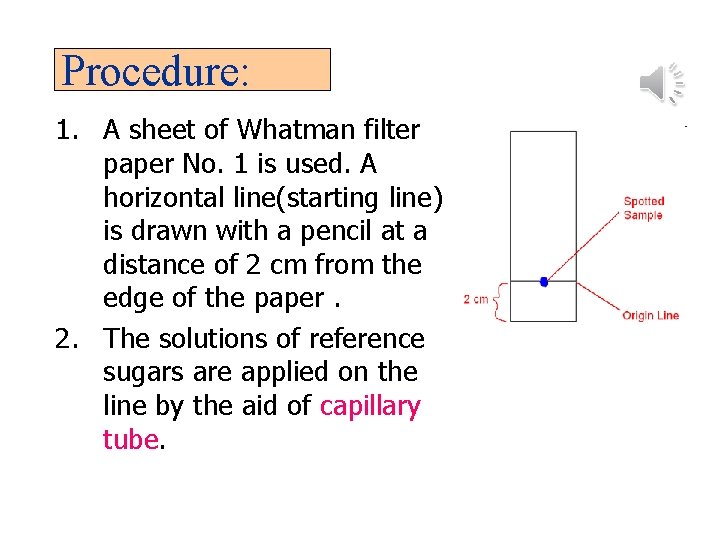
Procedure: 1. A sheet of Whatman filter paper No. 1 is used. A horizontal line(starting line) is drawn with a pencil at a distance of 2 cm from the edge of the paper. 2. The solutions of reference sugars are applied on the line by the aid of capillary tube.

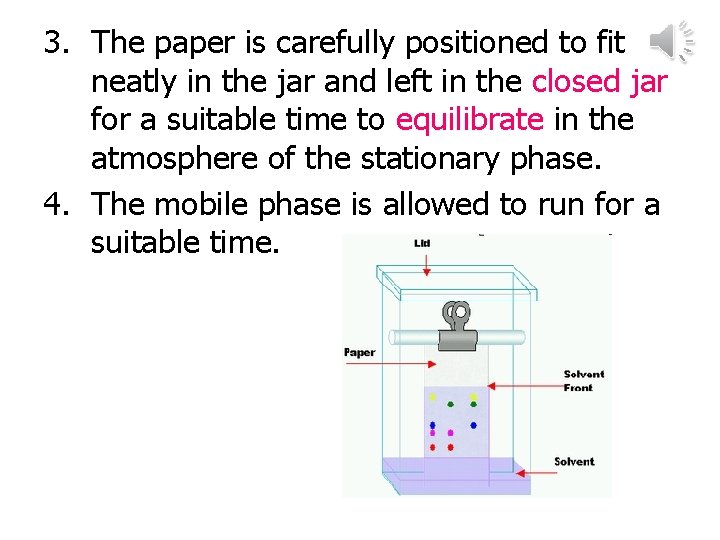
3. The paper is carefully positioned to fit neatly in the jar and left in the closed jar for a suitable time to equilibrate in the atmosphere of the stationary phase. 4. The mobile phase is allowed to run for a suitable time.

5. Carefully remove the paper, mark the front line with a pencil, left to dry. 6. The spots are revealed by the spray reagent, their color recorded and their Rf values calculated.