The solar spectrum compared to a black body
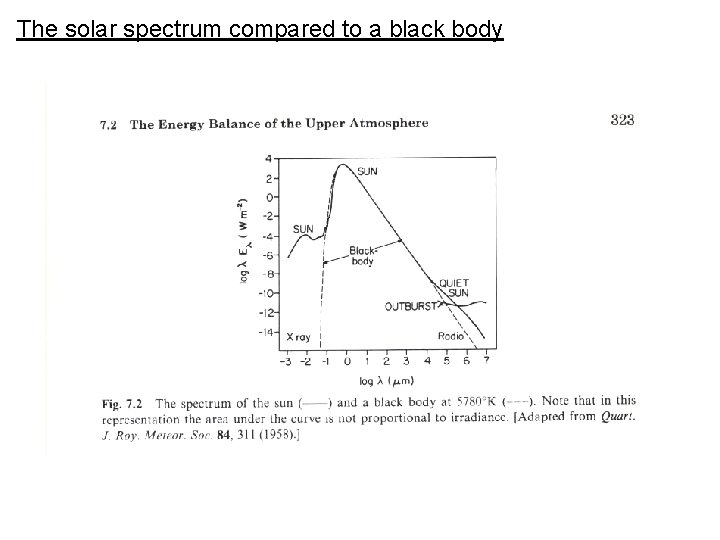
The solar spectrum compared to a black body
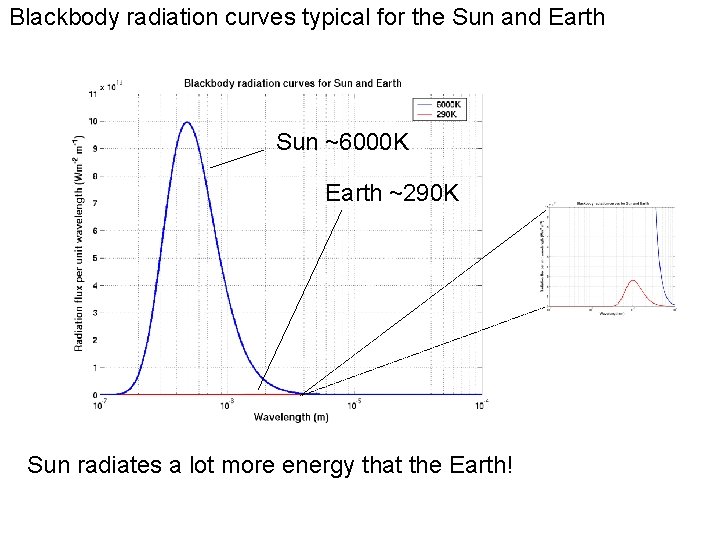
Blackbody radiation curves typical for the Sun and Earth Sun ~6000 K Earth ~290 K Sun radiates a lot more energy that the Earth!
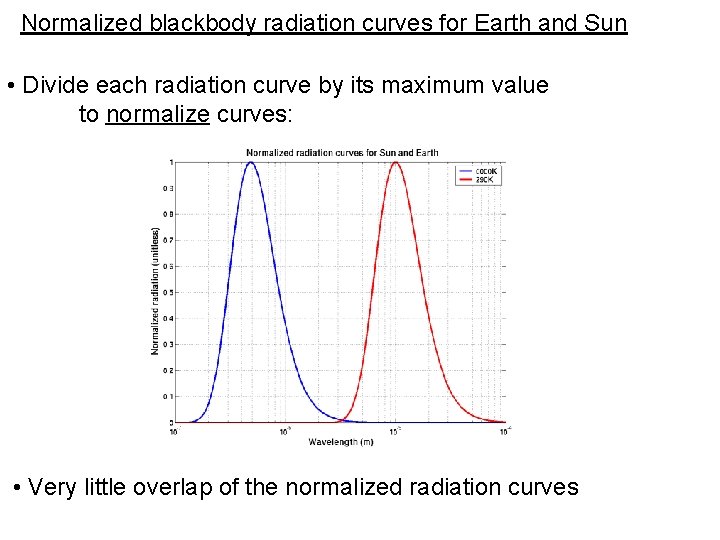
Normalized blackbody radiation curves for Earth and Sun • Divide each radiation curve by its maximum value to normalize curves: • Very little overlap of the normalized radiation curves
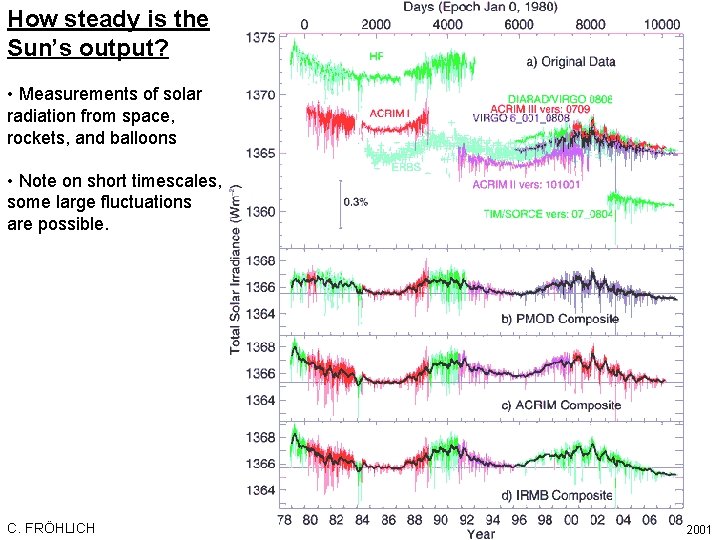
How steady is the Sun’s output? • Measurements of solar radiation from space, rockets, and balloons • Note on short timescales, some large fluctuations are possible. C. FRÖHLICH IPCC, 2001
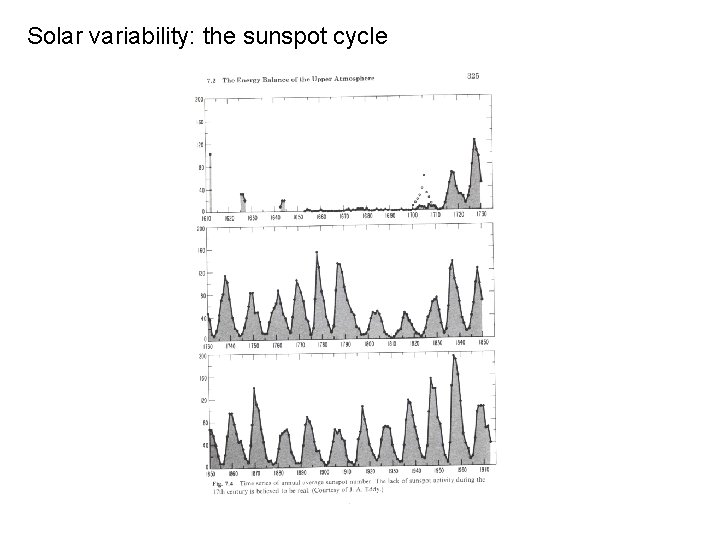
Solar variability: the sunspot cycle
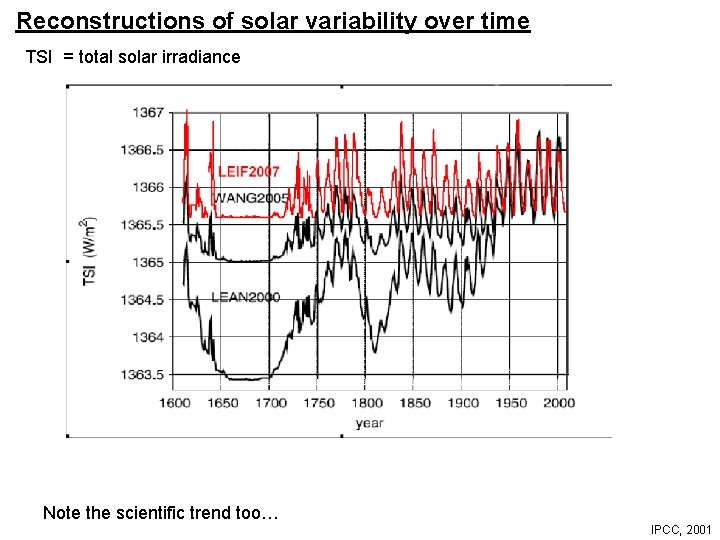
Reconstructions of solar variability over time TSI = total solar irradiance Note the scientific trend too… IPCC, 2001
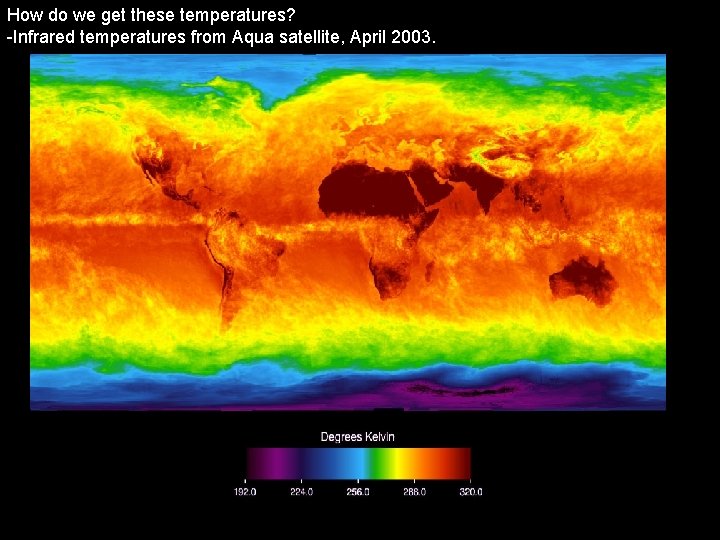
How do we get these temperatures? -Infrared temperatures from Aqua satellite, April 2003.
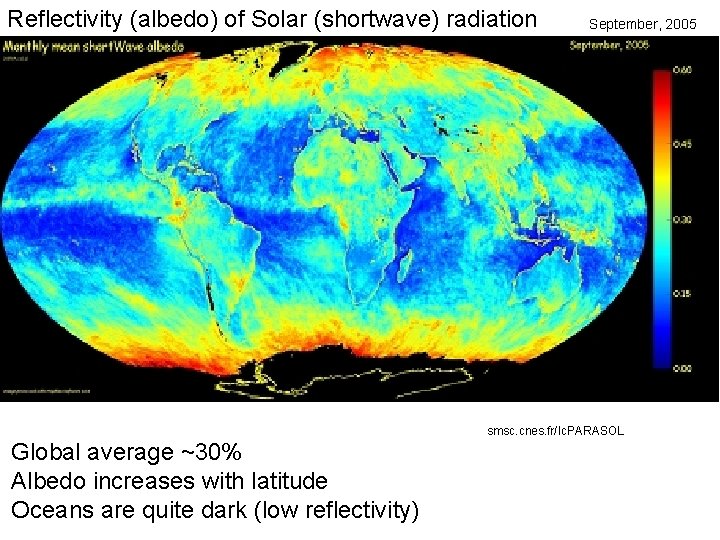
Reflectivity (albedo) of Solar (shortwave) radiation September, 2005 smsc. cnes. fr/Ic. PARASOL Global average ~30% Albedo increases with latitude Oceans are quite dark (low reflectivity)

Emissivity of infrared radiation at the surface cimss. ssec. wisc. edu/iremis/ Emissivity, e, is a measure of how well blackbody radiation is obeyed: F=es. T 4 Emissivity/absorptivity is close to 1. This implies a good approximation to black body in the infrared

Summary (important)At visible wavelengths, the Earth reflects about 30% of the incident radiation. At infrared wavelengths, most natural materials absorb almost Everything (~95 to 98%), so the Earth behaves quite closely as a true blackbody. -Go to calculation of black body temperature

Radiation and physical objects Any physical material (solid, liquid, gas) interacts with electromagnetic waves (radiation) in one of four different ways. TRANSMISSION: waves pass through the material ABSORPTION: some of the waves are absorbed (& heat) REFLECTION: some of the waves are reflected in the direction they came from. EMISSION: Every object (above absolute zero) emits radiation because it possesses thermal energy Less important: SCATTERING: waves are deflected (hence blue sky…)

Radiation and physical objects How a material interacts with radiation (transmission, absorption, emission, reflection) depends on what it is made of. For example: what’s the difference between the yellow light in these 3 pictures?

A key fact for Earth’s climate is that gases in the atmosphere absorb radiation. • Molecules absorb radiation at particular wavelengths, depending on amount of energy required to cause vibration or rotation of atomic bond. • Two essential things for the greenhouse effect: – The Earth’s atmosphere is mostly transparent to visible radiation (why not totally) – The Earth’s atmosphere is mostly opaque to infrared radiation.
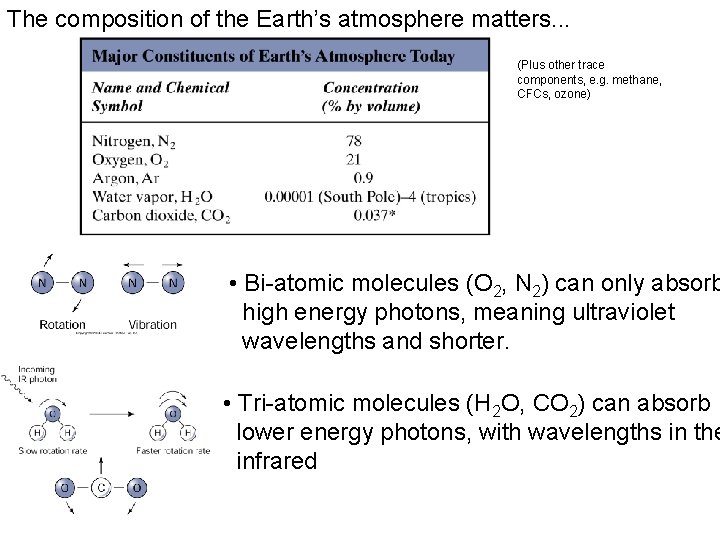
The composition of the Earth’s atmosphere matters. . . (Plus other trace components, e. g. methane, CFCs, ozone) • Bi-atomic molecules (O 2, N 2) can only absorb high energy photons, meaning ultraviolet wavelengths and shorter. • Tri-atomic molecules (H 2 O, CO 2) can absorb lower energy photons, with wavelengths in the infrared
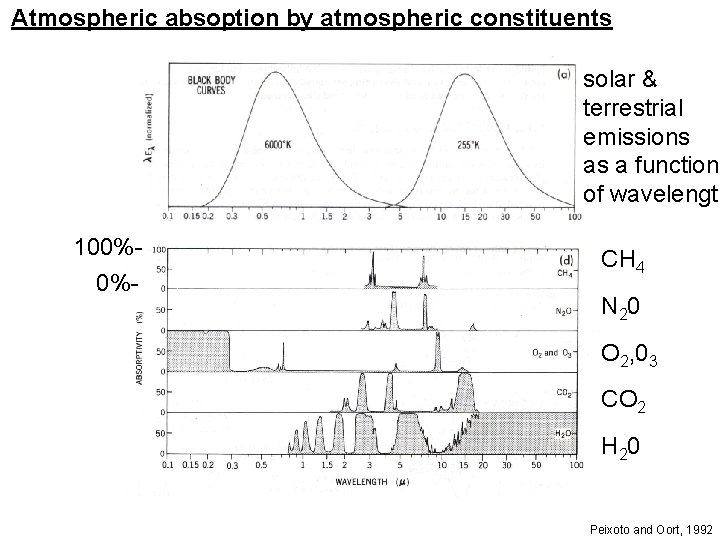
Atmospheric absoption by atmospheric constituents solar & terrestrial emissions as a function of wavelength 100%0%- CH 4 N 20 O 2, 03 CO 2 H 20 Peixoto and Oort, 1992

Key things from previous slide: - - Atmosphere mostly transparent to solar radiation (except in uv) - Atmosphere mostly opaque to terrestrial radiation (infrared) - Water vapor is the most important greenhouse gas (by far) - Carbon dioxide is a problem because of a ‘window’ in H 2 O absorption spectrum. This physics is very, very well known
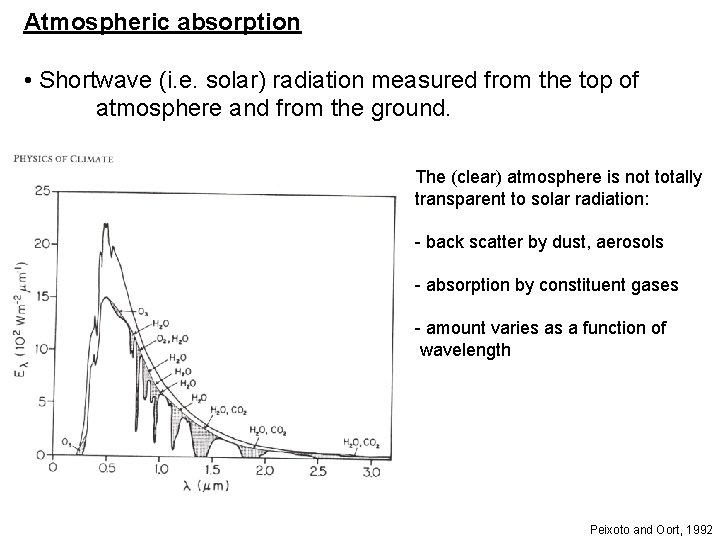
Atmospheric absorption • Shortwave (i. e. solar) radiation measured from the top of atmosphere and from the ground. The (clear) atmosphere is not totally transparent to solar radiation: - back scatter by dust, aerosols - absorption by constituent gases - amount varies as a function of wavelength Peixoto and Oort, 1992
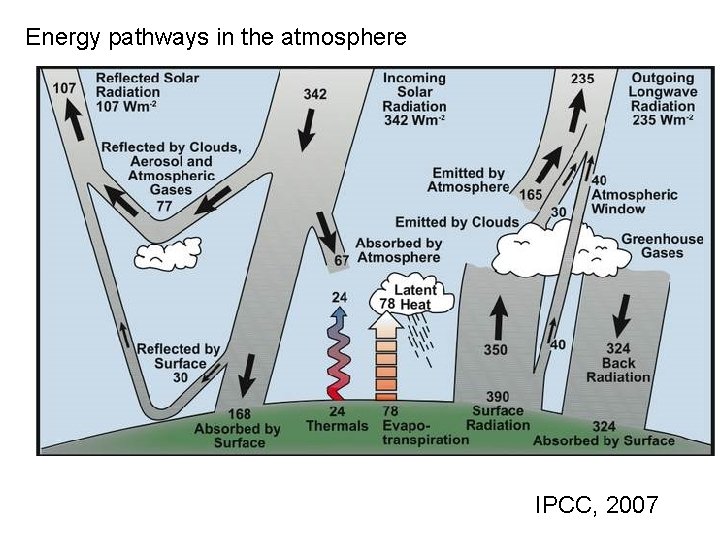
Energy pathways in the atmosphere IPCC, 2007
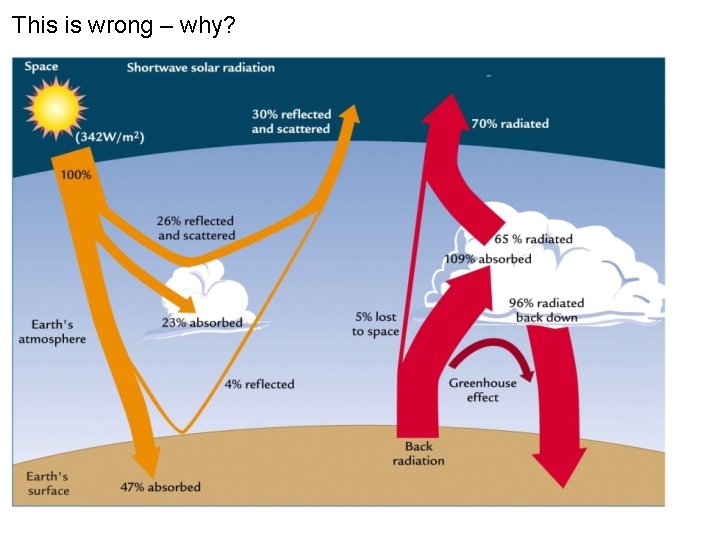
This is wrong – why?

Greenhouse effect summary • CO 2 and H 20 (and some other gasses) effectively absorb radiation at the same wavelengths that the Earth emits at. • Some of that radiation is then re-emitted back towards the ground keeping the surface warmer than it would otherwise be. Essential to remember: - CO 2 , H 20 in the atmosphere absorbs and re-emits infrared radiation - It does NOT (not, not) reflect radiation

- Slides: 22