The Significance of Our Gut Microbiome and Potential

The Significance of Our Gut Microbiome and Potential Roles of Probiotics Lincoln Westfall, DO Family Physician in Sunnyside, WA (soon in Chelan, WA) AAPCE San Diego 11/1/2019

Objectives • Recognize the significance of a balanced gut microbiome • Identify factors that can disrupt healthy gut flora • Identify conditions affected by disruption of healthy gut flora • Offer ways to mitigate disruption of a healthy microbiome • Explore possible future studies to pursue further application of probiotics and other manipulation of bowel flora

Microbiome / Microbiota • “Ecological community of commensal, symbiotic And pathogenic microorganisms within a body Space or other environment. ” Lederberg & Mc. Cray 2001 • "All animals and plants establish symbiotic relationships with microorganisms. “ Rosenberg & Rosenberg Microbiol Rev 2008 32(5): 723 -35

The intestinal microbiome • Composed of microbes that reside in the gut and may be altered by diet, lifestyle, exposure to toxins, and antibiotic use. • There is a relationship between disease, health, the immune system, and changes in the microbiota. • Probiotics have an important role in the maintenance of immunologic equilibrium in the GI tract through direct interaction with immune cells. • The microbiome diversity is likely important in health maintenance, and it is likely that broad-spectrum probiotics may increase the effectiveness of treatment.

In the human body • 1013 human cells • 1014 microbial cells • 1012 bacteriophages/g of feces • 1011 bacteria/g of feces • Gut microbes add an average of 600, 000 genes to each human

Gut microbiota • >3 million microbial genes in our gut microbiota – 150 times more genes than in the human genome. • Microbiota, in total, can weigh up to 2 kg. • More than 1, 000 different known bacterial species can be found in human gut microbiota, but only 150 to 170 predominate in any given subject. • One third of our gut microbiota is common to most people, while two thirds are specific to each one of us. gutmicrobiotaforhealth. com
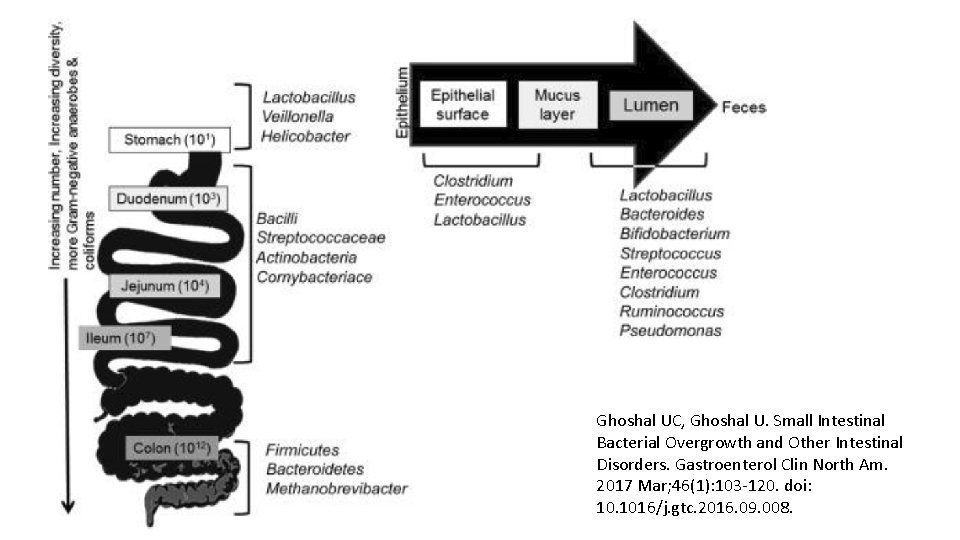
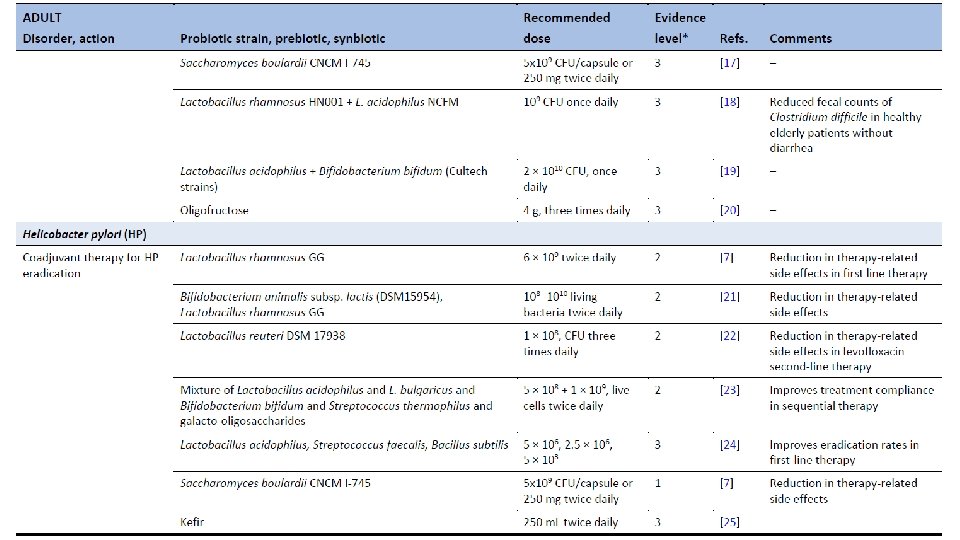
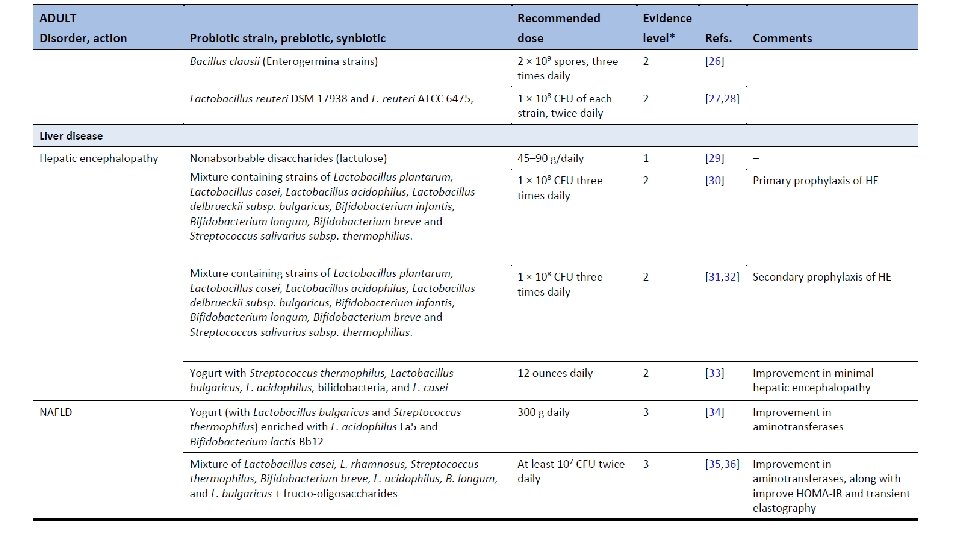
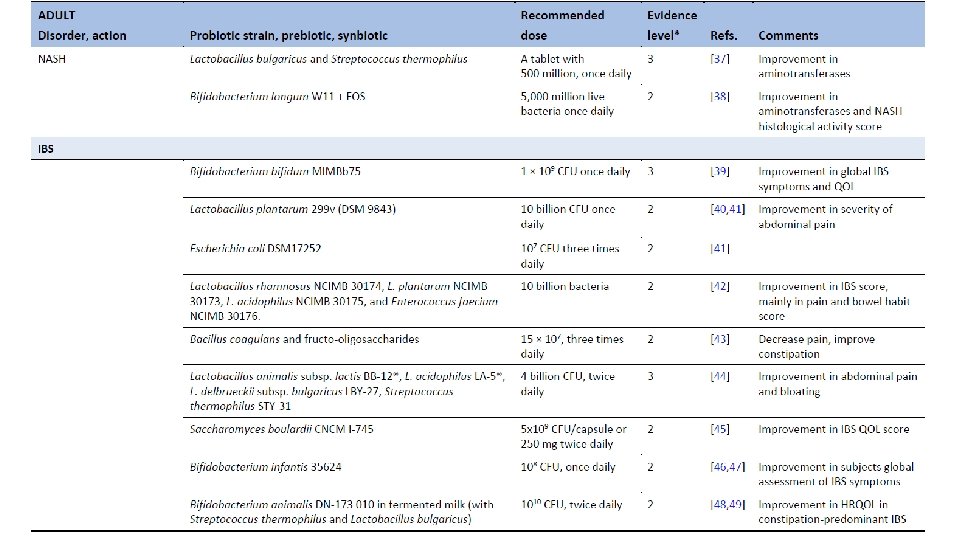
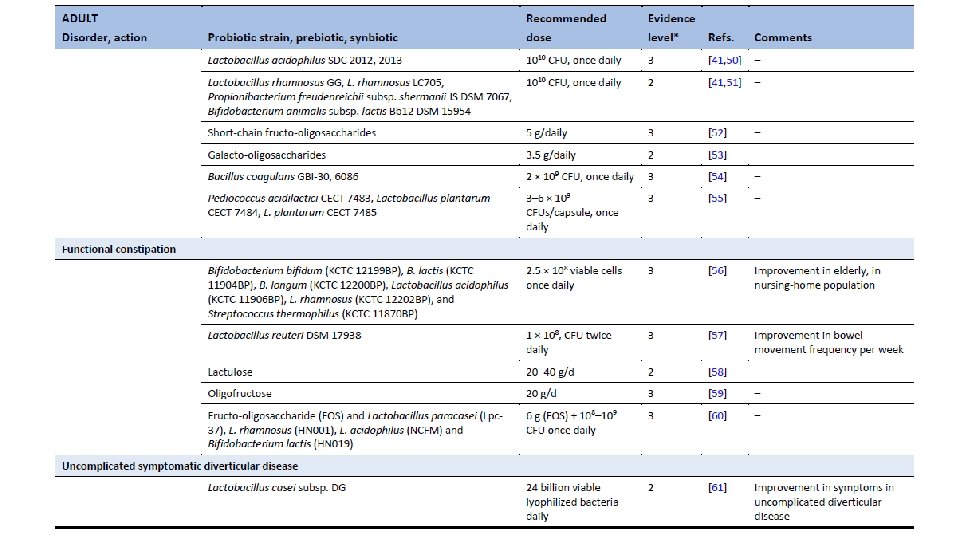
Ghoshal UC, Ghoshal U. Small Intestinal Bacterial Overgrowth and Other Intestinal Disorders. Gastroenterol Clin North Am. 2017 Mar; 46(1): 103 -120. doi: 10. 1016/j. gtc. 2016. 09. 008.
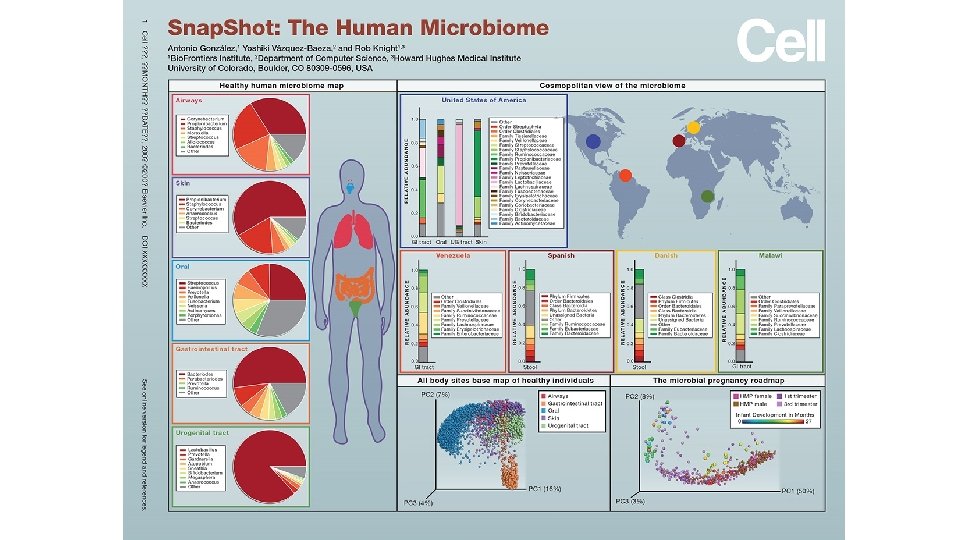
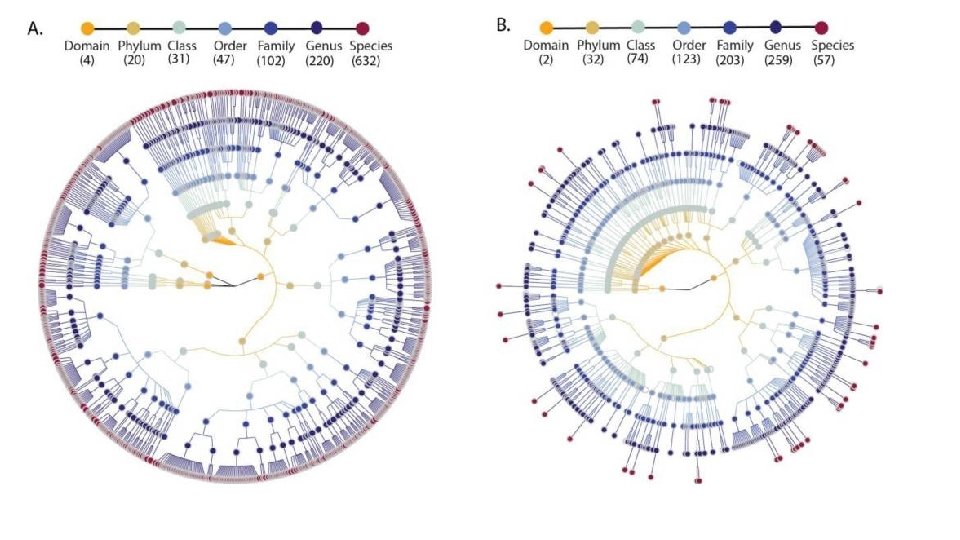
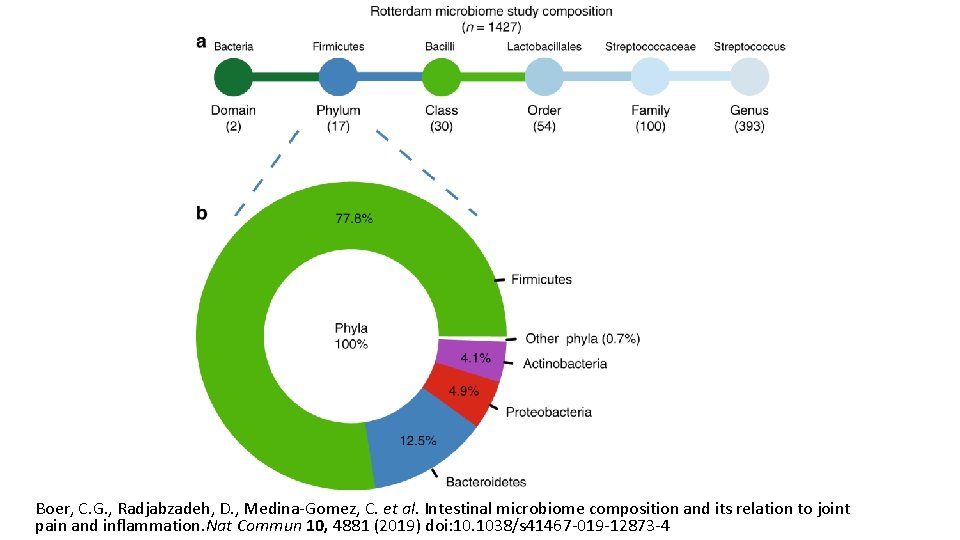
Boer, C. G. , Radjabzadeh, D. , Medina-Gomez, C. et al. Intestinal microbiome composition and its relation to joint pain and inflammation. Nat Commun 10, 4881 (2019) doi: 10. 1038/s 41467 -019 -12873 -4
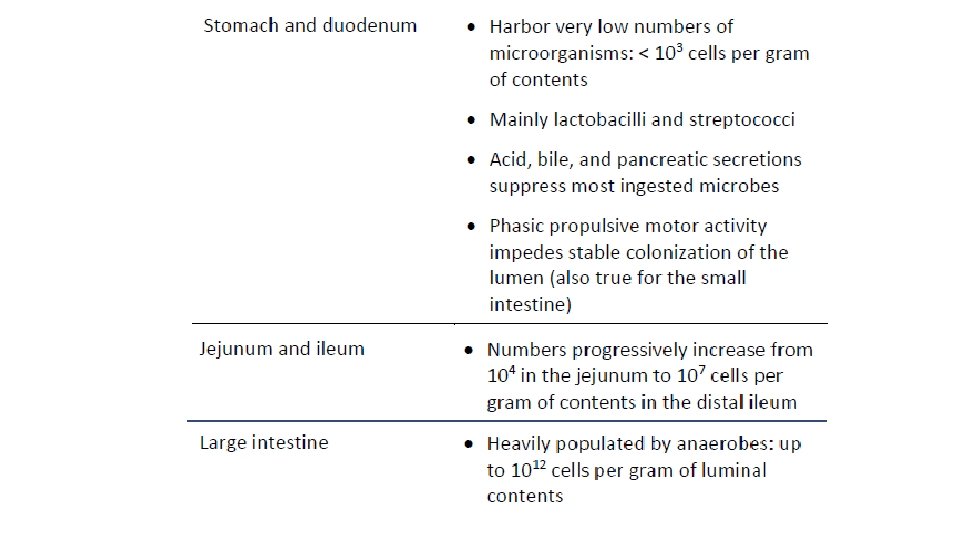
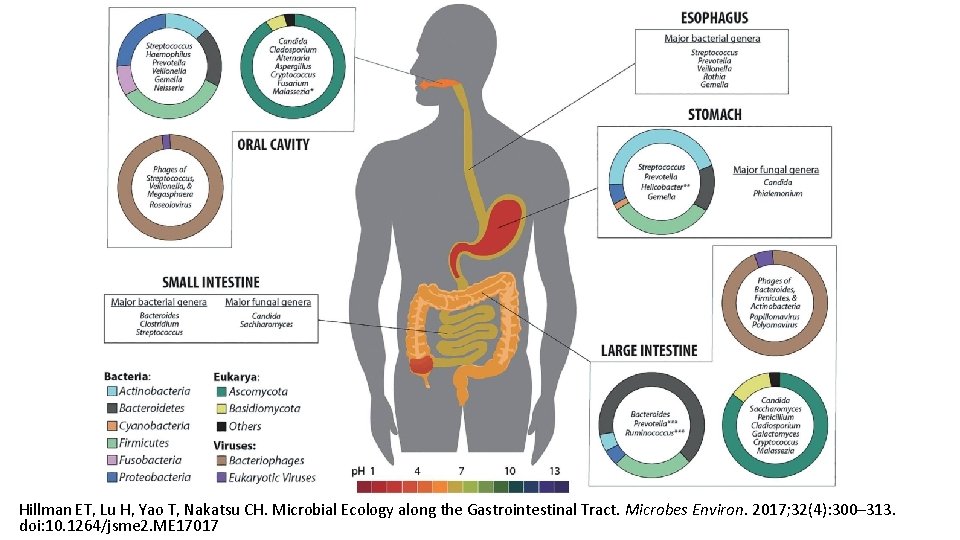
Hillman ET, Lu H, Yao T, Nakatsu CH. Microbial Ecology along the Gastrointestinal Tract. Microbes Environ. 2017; 32(4): 300– 313. doi: 10. 1264/jsme 2. ME 17017

Function of Gut Microbiota • Metabolism of nondigestible carbohydrates, which include large polysaccharides, such as resistant starches, cellulose, hemicellulose, pectins, and gums; some oligosaccharides that escape digestion; unabsorbed sugars and alcohols from the diet; and host-derived mucins. • Produce antimicrobial compounds and compete for nutrients and sites of attachment in the gut lining, thereby preventing colonization by pathogens. • Prevention of allergy (ie, a disproportionate reaction of the immune system to nonharmful antigens). Allergic infants and young children have been found to have a different composition of intestinal bacteria than those who do not develop allergies. • The gut–brain axis is a communication system that integrates neural, hormonal, and immunological signaling between the gut and the brain, offering the intestinal microbiota and its metabolites a potential route through which to access the brain.

Important metabolites produced by the microbiota • Folate • Indoles • Secondary bile acids • Trimethylamine-N-oxide (TMAO) • Short-chain fatty acids (SCFAs) (i. e. butyrate, propionate and acetate). • Neurotransmitters like serotonin and gamma amino butyric acid • 90% of the body's serotonin is made in the digestive tract!

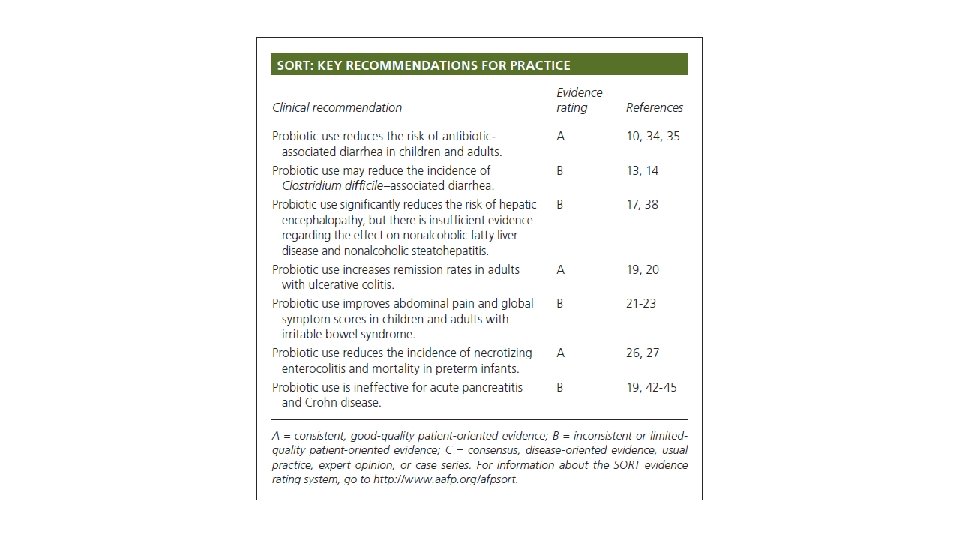
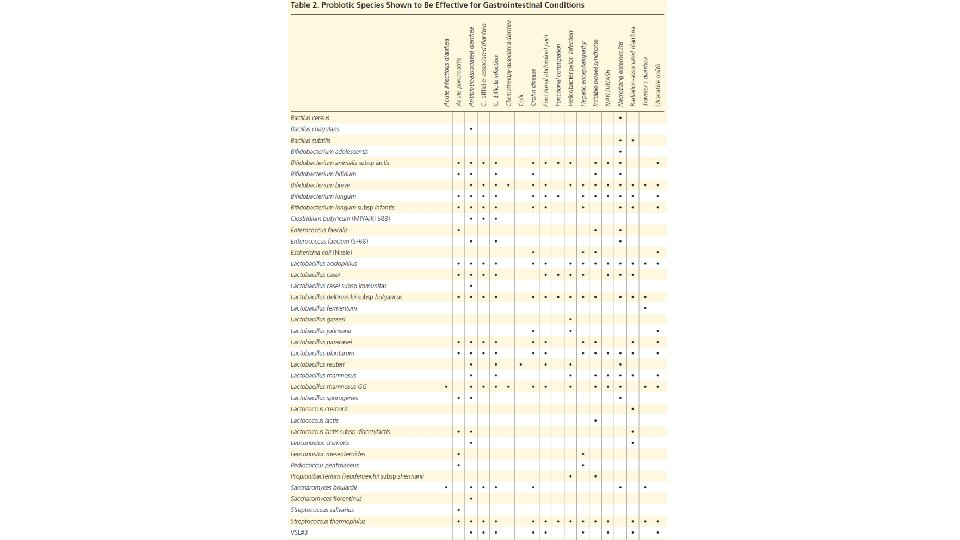
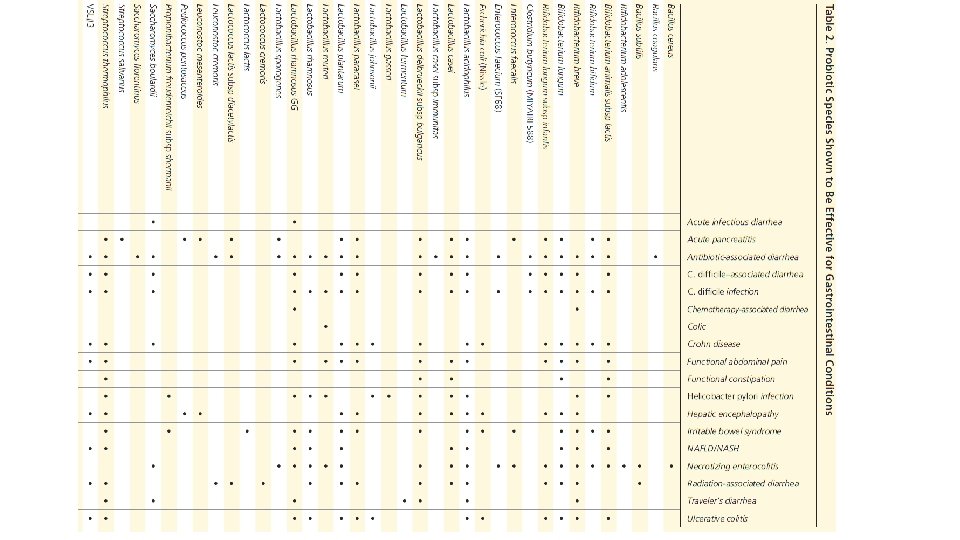
Proposed Mechanisms of Probiotics • Block the adhesion of pathogenic bacteria to the intestinal epithelium; produce inhibitory agents • Enhance the intestinal immune response • Maintain normal levels of short-chain fatty acids • Modulate immune system function, such as suppression of intestinal proinflammatory cytokines • Repair intestinal permeability • Suppress the growth of pathogenic bacteria by directly binding to gram-negative bacteria • Upregulate intestinal electrolyte absorption Probiotics for Gastrointestinal Conditions: A Summary of the Evidence. Thad Wilkins, Jacqueline Sequoia. Am Fam Physician. 2017 Aug 1; 96(3): 170– 178.
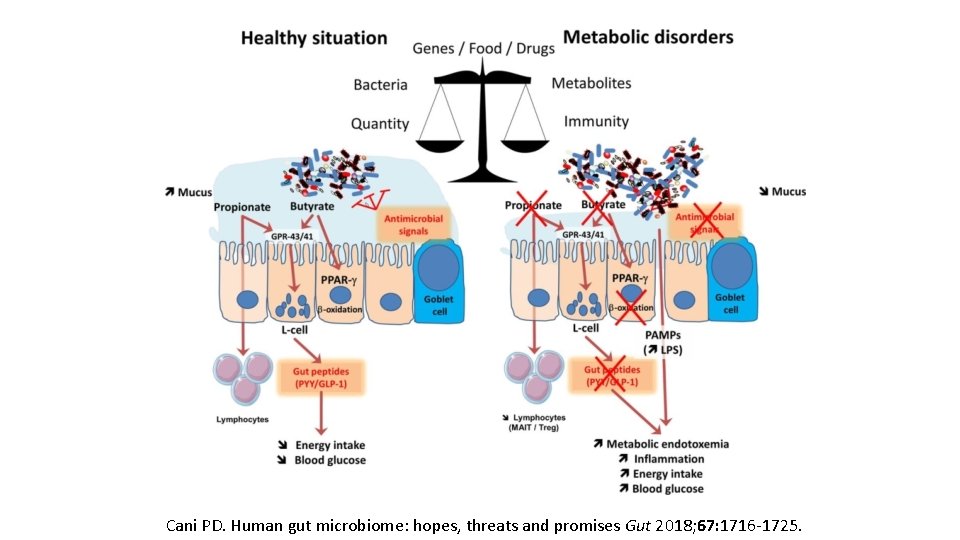
Cani PD. Human gut microbiome: hopes, threats and promises Gut 2018; 67: 1716 -1725.
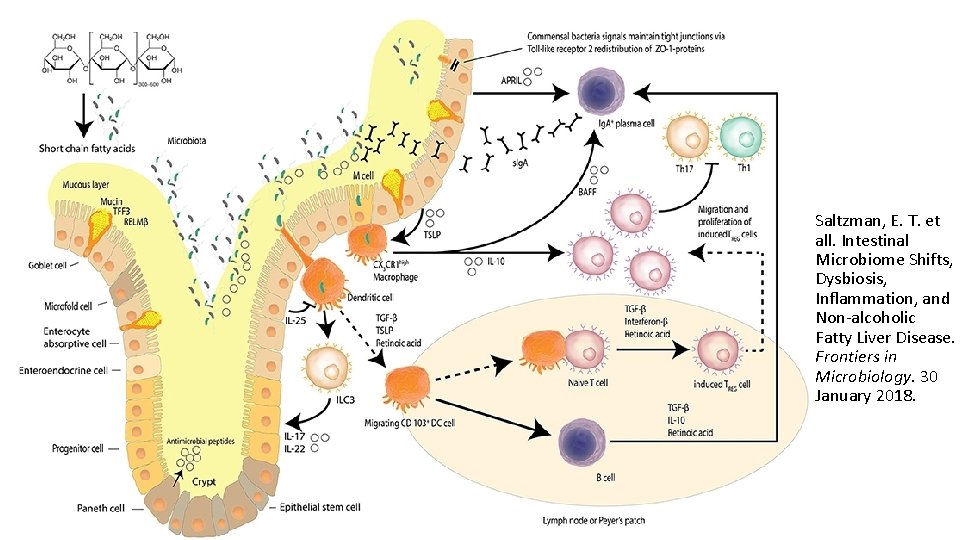
• Saltzman, E. T. et all. Intestinal Microbiome Shifts, Dysbiosis, Inflammation, and Non-alcoholic Fatty Liver Disease. Frontiers in Microbiology. 30 January 2018.

Dysbiosis • Stress has been shown to influence the integrity of the gut epithelium and to alter peristalsis, secretions, and mucin production • IBS patients have shown a 2 -fold increase in the ratio of Firmicutes to Bacteroidetes • IBD patients have shown greatly reduced diversity of Firmicutes (producers of anti-inflammatory SCFA) • Diverting ileostomy resolves IBD. • Differences in microbiota between obese and lean people. • Decreased diversity of Bacteroidetes at 12 months of age in atopic-eczema patients. Bull MJ, Plummer NT. Part 1: The Human Gut Microbiome in Health and Disease. Integr Med (Encinitas). 2014; 13(6): 17– 22.
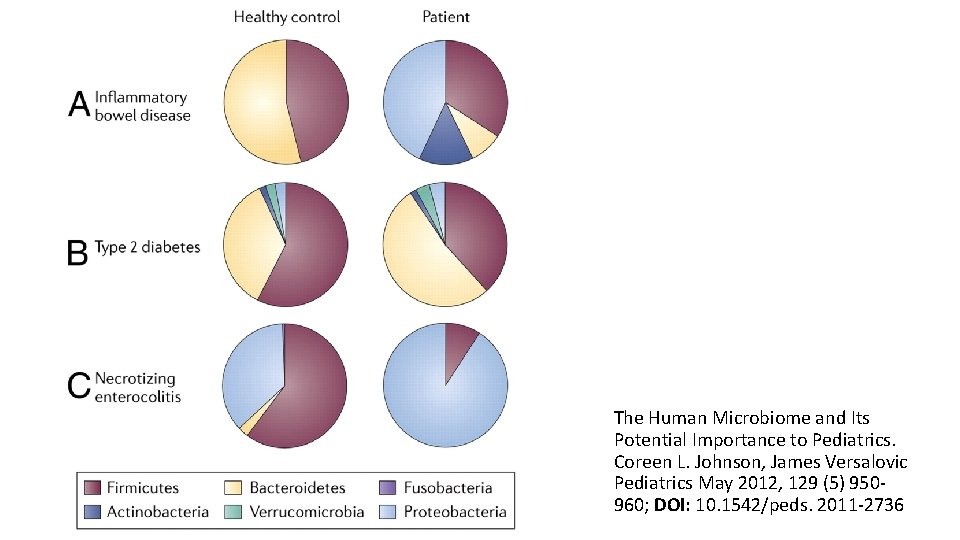
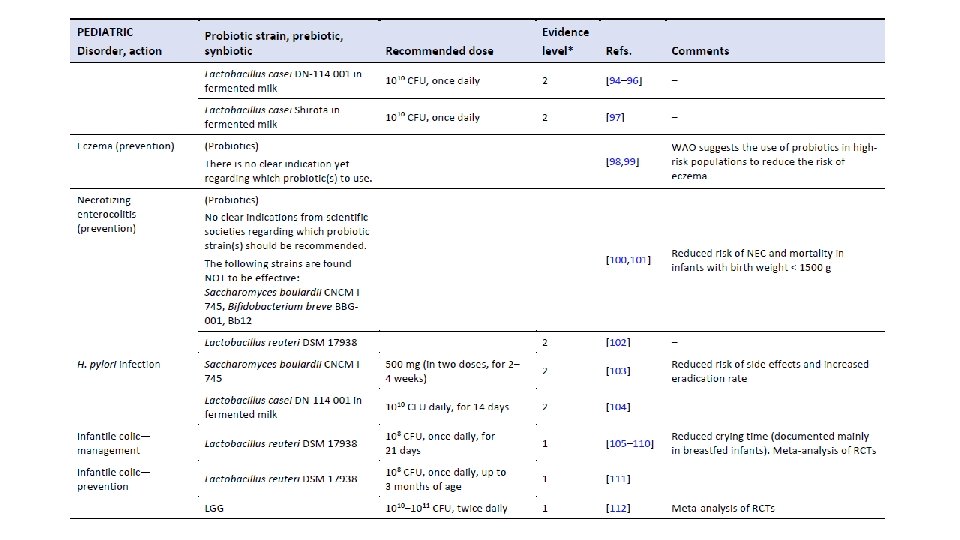
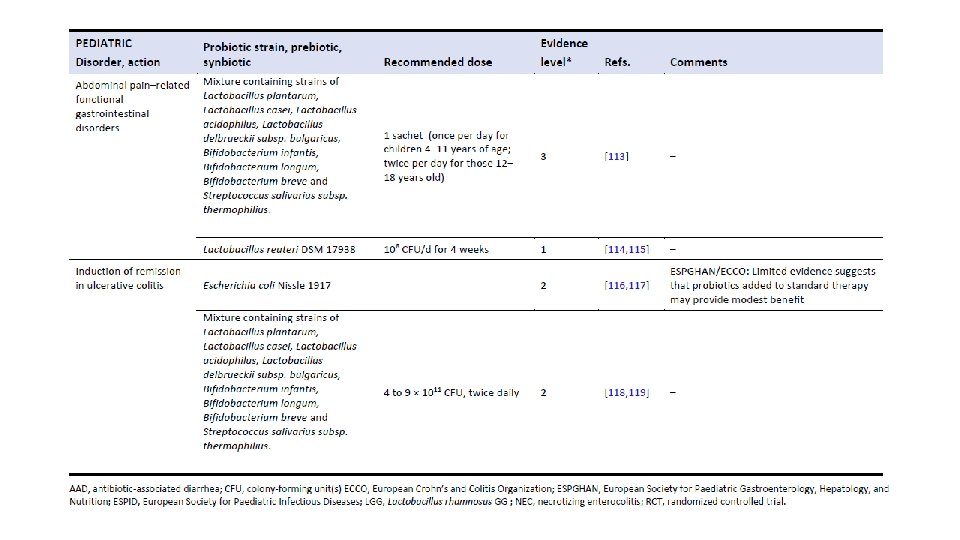
The Human Microbiome and Its Potential Importance to Pediatrics. Coreen L. Johnson, James Versalovic Pediatrics May 2012, 129 (5) 950960; DOI: 10. 1542/peds. 2011 -2736
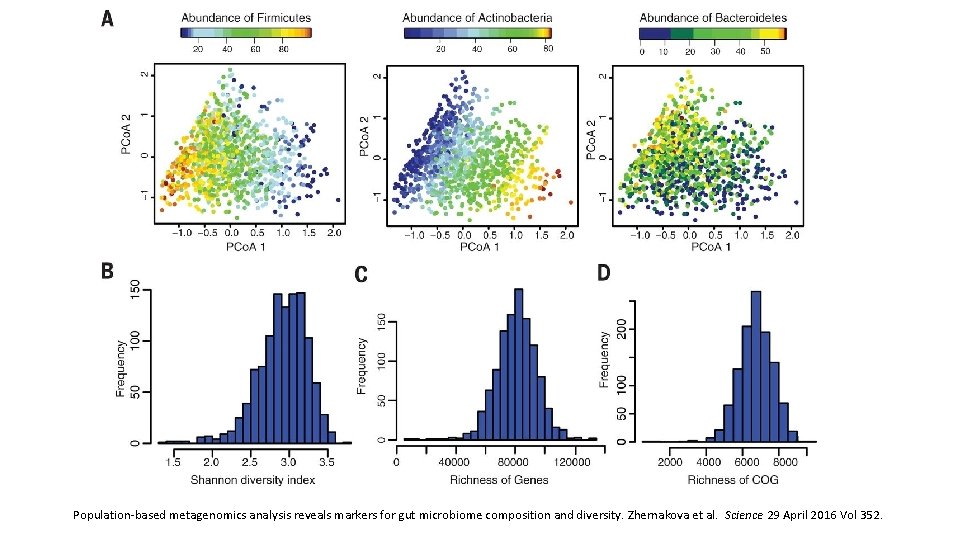
Population-based metagenomics analysis reveals markers for gut microbiome composition and diversity. Zhernakova et al. Science 29 April 2016 Vol 352.
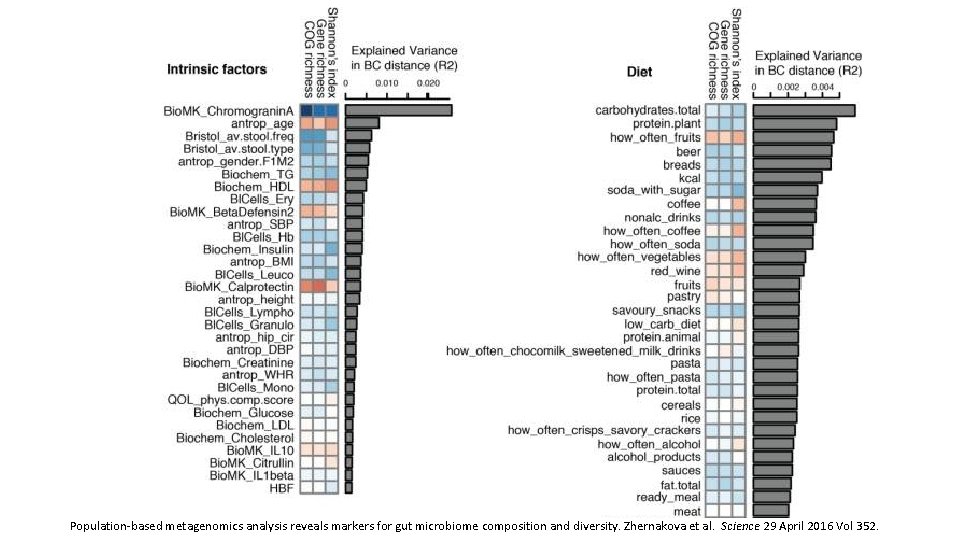
Population-based metagenomics analysis reveals markers for gut microbiome composition and diversity. Zhernakova et al. Science 29 April 2016 Vol 352.
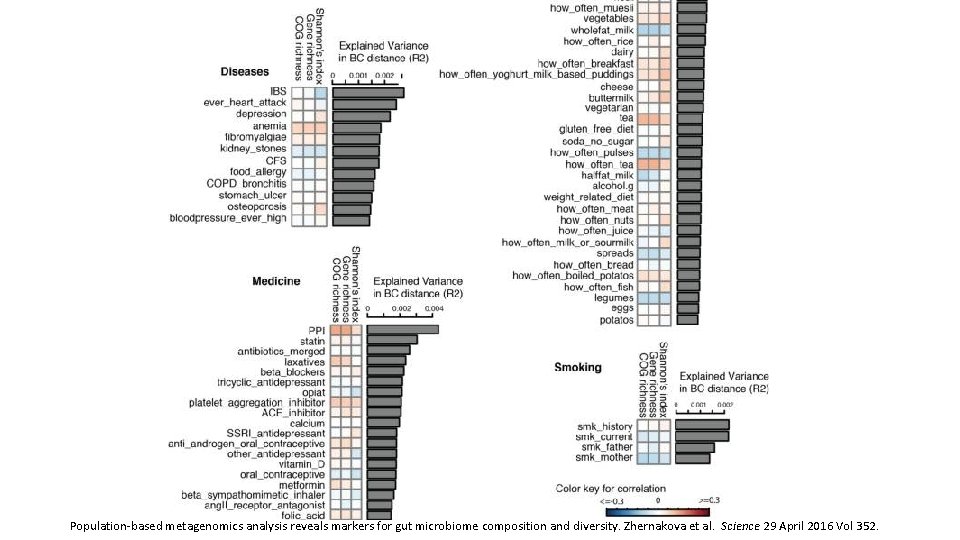
Population-based metagenomics analysis reveals markers for gut microbiome composition and diversity. Zhernakova et al. Science 29 April 2016 Vol 352.

Dietary markers for gut microbiome diversity • Drinking buttermilk (sour milk with a low fat content) was associated with high diversity, while drinking highfat (whole) milk (3. 5% fat content) was associated with lower diversity (table S 6). Two of the species most strongly associated with drinking buttermilk are Leuconostoc mesenteroides (q=9. 1× 10− 46) and Lactococcus lactis (q=2. 5× 10− 8), both used as a starter culture for industrial fermentation (table S 11). The abundance of dairy-fermentation-related bacteria increased with increasing dairy consumption, indicating potential for the use of probiotic drinks to augment and alter the gut microbiome composition. • Consumption of alcohol-containing products, coffee, tea, and sugar-sweetened drinks were also correlated with microbial composition. Consumption of sugar-sweetened soda had a negative effect on microbial diversity (adjusted P=5× 10− 4), whereas consumption of coffee, tea and red wine, which all have a high polyphenol content, was associated with increased diversity (19– 21). Red wine consumption correlated with F. prausnitzii abundance, which has anti-inflammatory properties, correlates negatively with inflammatory bowel disease (22), and shows higher abundance in high-richness microbiota (23). • Apart from the negative associations between sugar-sweetened soda and bacterial diversity, other features of a Western-style diet, such as higher intake of total energy, snacking, and high-fat (whole) milk, were also associated with lower microbiota diversity (Fig. 3). A higher amount of carbohydrates in the diet was associated with lower microbiome diversity. Total carbohydrate intake was positively associated with Bifidobacteria, but negatively with Lactobacillus, Streptococcus, and Roseburiaspecies. A low carbohydrate diet consistently showed opposite directions for these species. Population-based metagenomics analysis reveals markers for gut microbiome composition and diversity. Zhernakova et al. Science 29 April 2016 Vol 352.

Medication effects on gut microbiome • As expected, the use of antibiotics was significantly associated with microbiome composition, in particular with strong and significant decreases in two species from the genus Bifidobacterium (Actinobacteriaphylum). • PPI users were found to have profound changes in 33 bacterial pathways. The most significant positive correlation of PPIs was observed with the pathway of 2, 3 -butanediol biosynthesis, and PPI use was correlated with calprotectin levels, particularly for bacteria typical of the oral microbiome. Levels of calprotectin were positively correlated with age and metabolic phenotypes (body mass index (BMI), diabetes, use of statins and metformin, HBAc 1, and systolic blood pressure), but negatively correlated with the consumption of vegetables, plant proteins, chocolate, and breads. • Multivariate analysis correcting for all factors revealed 14 species (table S 13) and 114 bacterial metabolic pathways (table S 14) exclusively associated with calprotectin, suggesting calprotectin is robustly associated with gut microbiome. • In 15 metformin users, we observed an increased abundance of Escherichia coli (E. coli) and a positive correlation with specific pathways, including the degradation and utilization of D-glucarate and D-galactarate and pyruvate fermentation pathways. SCFA levels were consistently higher in metformin-users, especially for propionate. Population-based metagenomics analysis reveals markers for gut microbiome composition and diversity. Zhernakova et al. Science 29 April 2016 Vol 352.

Correlation between disease and dysbiosis • IBS was reported by 9. 9% of participants (n=112, table S 1) and was associated with changes in the gut microbiome and a lower microbial diversity (adjusted P=0. 05). • Species from the Eggerthella and Coprobacillus genera were positively associated with medication and food allergies, respectively. • Individuals who had suffered a heart attack (n=10) in the past had a significantly lower abundance of Eubacterium eligens bacterium. • Higher BMI was associated with lower level of two species from the family Rikenellaceae, Alistipes finegoldii, and Alistipes senegalensis, while blood lipids were associated with other two species, Alistipes shahii and Alistipes putredinis. • Correlations with the number of different species were also found for other bacteria including Roseburia hominis, Coprococcus catus, and Barnesiella intestinihominis and unclassified species from genus Anaerotruncus that also showed correlation both with fruit, vegetable, and nut consumption and with intrinsic phenotypes like HDL, triglycerides and quality of life. Based on this data, it would be interesting to explore the potential to modulate disease-associated species through medication or diet, although we still need to address the causality and underlying mechanism. Population-based metagenomics analysis reveals markers for gut microbiome composition and diversity. Zhernakova et al. Science 29 April 2016 Vol 352.



Prebiotics - You are what your microbiome eats! • non-digestible by host enzymes • fermented in the GI tract by anaerobic endogenous colon bacteria • Selective in stimulation of intestinal flora Patrick Hanaway, Treating Dysbiosis: Weed, Seed, and Feed, IFM GI AFP, Oct 2018.
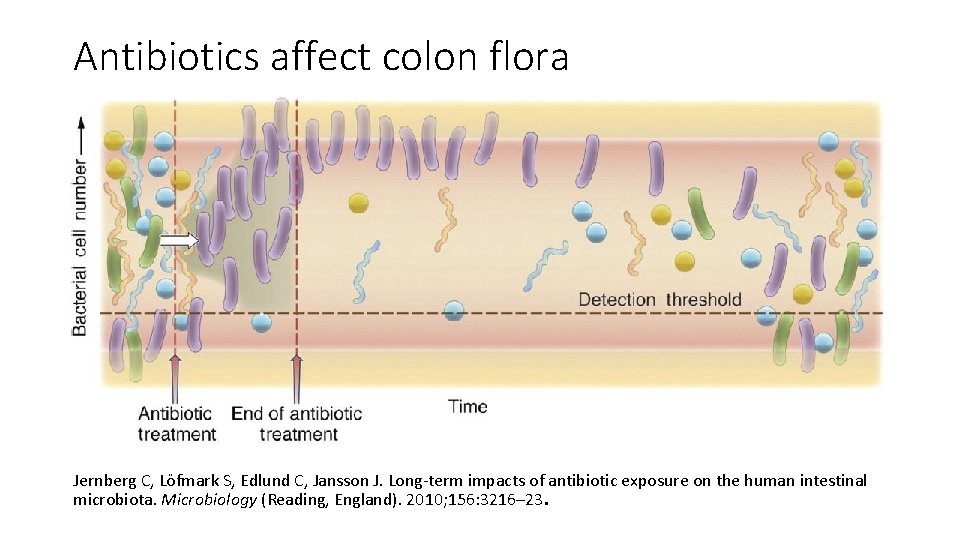
Antibiotics affect colon flora Jernberg C, Löfmark S, Edlund C, Jansson J. Long-term impacts of antibiotic exposure on the human intestinal microbiota. Microbiology (Reading, England). 2010; 156: 3216– 23.
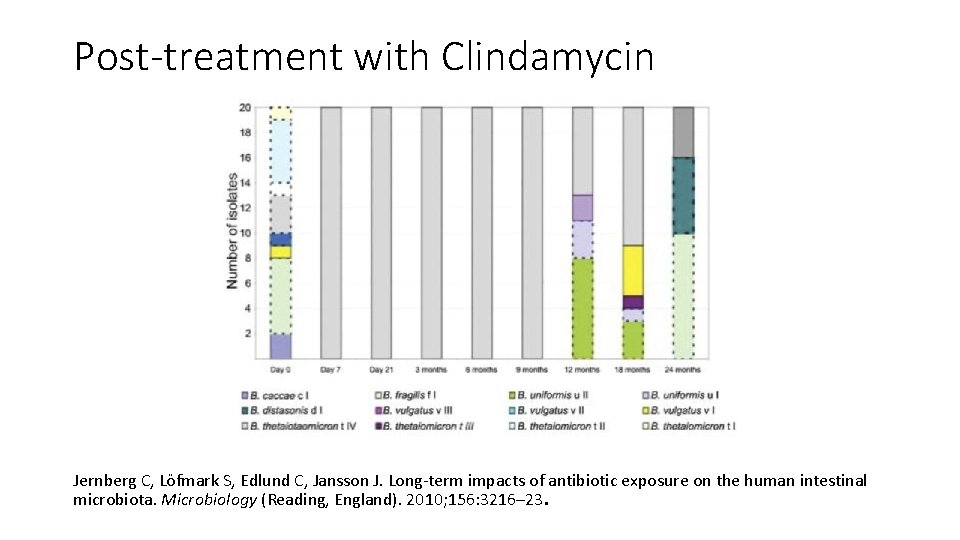
Post-treatment with Clindamycin Jernberg C, Löfmark S, Edlund C, Jansson J. Long-term impacts of antibiotic exposure on the human intestinal microbiota. Microbiology (Reading, England). 2010; 156: 3216– 23.

Association of Antibiotics in Infancy With Early Childhood Obesity • CONCLUSIONS AND RELEVANCE Repeated exposure to broad-spectrum antibiotics at ages 0 to 23 months is associated with early childhood obesity. Because common childhood infections were the most frequent diagnoses co-occurring with broad-spectrum antibiotic prescription, narrowing antibiotic selection is potentially a modifiable risk factor for childhood obesity Bailey LC et al. Association of antibiotics in infancy with early childhood obesity. JAMA Pediatr. 2014 Nov; 168(11): 1063 -9. doi: 10. 1001/jamapediatrics. 2014. 1539.

Yeast colonization • Candida colonization of GI tract is associated with GI illnesses and perpetual inflammation. • Pro-inflammatory cytokine IL-17 elevated in IBD and in Candida colonization. Kumamoto, C. A. (2011). Inflammation and gastrointestinal Candida colonization. Current Opinion in Microbiology, 14(4), 386– 391. http: //doi. org/10. 1016/j. mib. 2011. 07. 015

Yeast eradication • Yeast thrives on sugar. Just stop feeding it! • Probiotics: Saccharomyces boulardii, Lactobacilli, Bifidobacter • Enzymes: amylase, peptidase, lipase. • Oregano, thyme, garlic, goldenseal • Nystatin, fluconazole, amphotericin B, ketoconazole, terbinafine, itraconazole. Consider parasite testing and eradication. Patrick Hanaway, Treating Dysbiosis: Weed, Seed, and Feed, IFM GI AFP, Oct 2018.
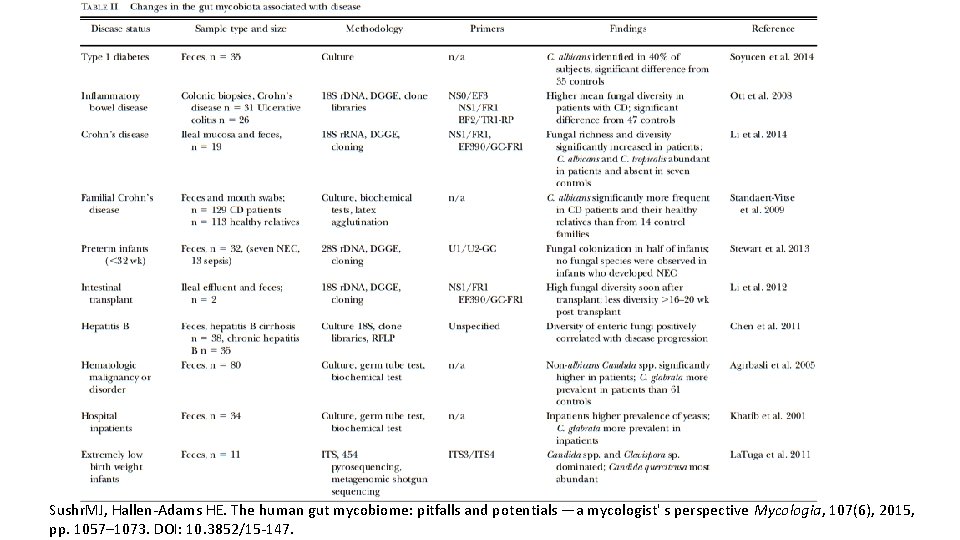
Sushr. MJ, Hallen-Adams HE. The human gut mycobiome: pitfalls and potentials —a mycologist' s perspective Mycologia, 107(6), 2015, pp. 1057– 1073. DOI: 10. 3852/15 -147.

Prebiotics = Food for healthy gut microbiota • Bran, psyllium, resistant starch (high amylose), breast milk, oligofructose, inulin, germinated barley foodstuff (GBF), synthetic oligosaccharides, guar, & lactulose. • FOS (fructooligosaccharides) 4 g+/day - found in onions, garlic, rye, chicory, blueberries, and bananas. • promote growth of bifidobacteria • Lower colon p. H • Discourage growth of clostridia • Inulins 1 -10 g/day - derived from chicory and artichoke. • reduce biomarkers of metabolic syndrome. • Modified citrus pectin 3 -5 g BID. • Larch (arabinogalactans) 500 -5000 mg BID Langen L, Dieleman L. Prebiotics in chronic intestinal inflammation. Inflammatory bowel diseases. 2008; 15(3): 454– 62.

Prebiotic Rich Foods • • • Jerusalem artichokes Onions Chicory root Garlic Leeks Bananas Fruit Soybeans Burdock root Asparagus Agave • • • Sugar maple Chinese chives Peas Legumes Eggplant Honey Green Tea Agave Pine nut Fava bean Yacon root

Dietary Fiber • Soluble fiber is completely fermented by the bacterial: pectins, gums, inulin type fructans, and mucilages. • Insoluble fiber is only slightly fermented: cellulose, waxes, beta glucan, and lignins (mainly in plant cell walls). • Wheat is 90% insoluble and 10% soluble • Oats are 50% insoluble and 50% soluble • Psyllium is 10% insoluble and 90% soluble J. M. Lattimer & M. D. Haub. Nutrients 2010, 2, 1266 -1289

Dietary Fiber • Recommended intake for adult is 30 -40 g/day (for child is age + 5). • Forms gel, increases stool bulk, holds water, binds minerals. • Inverse correlation to obesity, DM 2, CVD, cancer. • > SCFA, > gut hormones, < transit time, change in intestinal viscosity (less contact with mucosal cells) • > estrogen excretion, > antioxidant nutrients, < inflammation

Probiotic Rich Foods • Mushrooms • Yeast • Olives • Fermented cabbage (sauerkraut, kimchi) • Vinegars, wines • Fermented dairy (cheeses, kefir, buttermilk, sour cream, yogurt) • Cucumber pickles

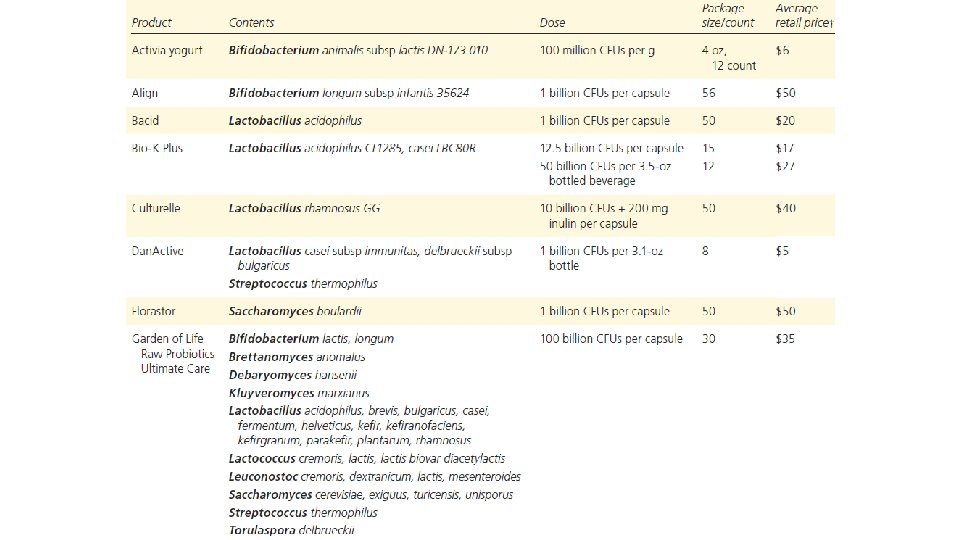
Probiotics • Probiotics are only recoverable in stool samples for one to two weeks. • Use the specific probiotic species and strains that have been studied. • Make sure that the product states that the dosage recommended is viable at the expiration date, and that the expiration date is printed on the bottle. Good Manufacturing Practice! • Their viability is temperature-dependent. Keep in fridge. Give in cool food/drink. • They are not well regulated in general.

Risks of Probiotics • Bacteremia (rare) • Endocarditis: 16 reported cases (1992 2001) 5 cases with yogurt or probiotic Only 1 confirmed to be identical to probiotic • Liver abscess (74 year old woman) Daily consumption of probiotic drink Indistinguishable from probiotic strain • Several reports of fungemia associated with use of yeast-based probiotics (S. boulardii) – usually critically ill host and polymicrobial

• “Only three out of the seven products (43%) contained the claimed culture concentration or more. ” • None of the multispecies products contained all the labeled probiotic bacteria. • Misidentification of some species occurred. Fredua-Agyeman M. , Parab S. & Gaisford S. (2016) “Evaluation of Commercial Probiotic Products”, British Journal of Pharmacy. 1(1). doi: https: //doi. org/10. 5920/bjpharm. 2016. 11

• Only 4 of 13 products (31%) were in accordance with label claims. • Our results suggest the need for adequate control of probiotic production as well as periodical screenings by competent organizations to monitor the effect of storage on product quality. Drago L, Rodighiero V, Celeste T, Rovetto L, De Vecchi E. Microbiological evaluation of commercial probiotic products available in the USA in 2009. J Chemother. 2010 Dec; 22(6): 373 -7.
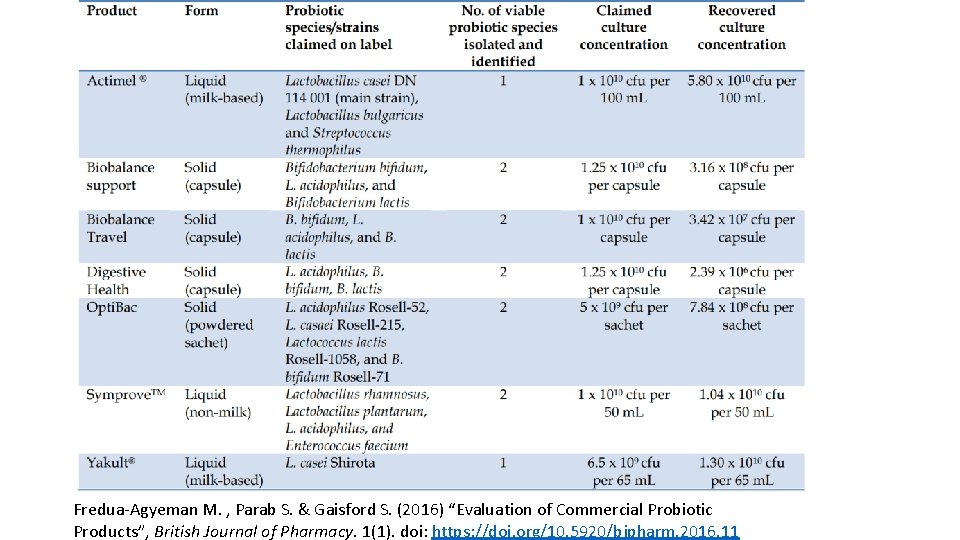
Fredua-Agyeman M. , Parab S. & Gaisford S. (2016) “Evaluation of Commercial Probiotic Products”, British Journal of Pharmacy. 1(1). doi: https: //doi. org/10. 5920/bjpharm. 2016. 11

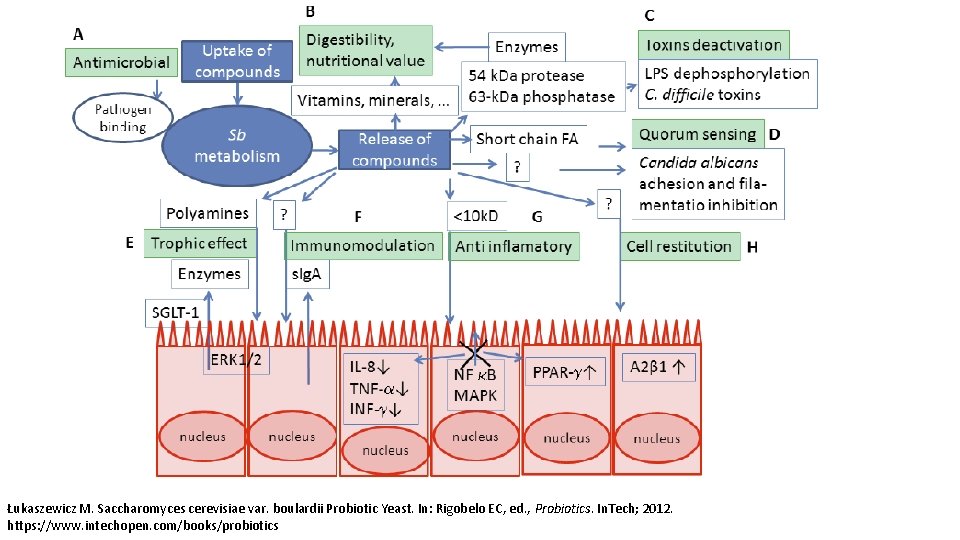
Łukaszewicz M. Saccharomyces cerevisiae var. boulardii Probiotic Yeast. In: Rigobelo EC, ed. , Probiotics. In. Tech; 2012. https: //www. intechopen. com/books/probiotics
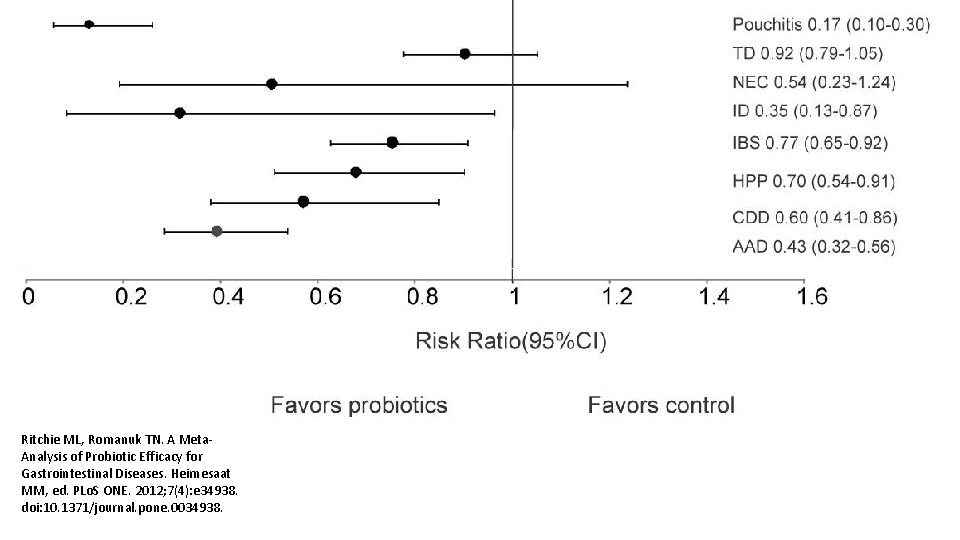
Ritchie ML, Romanuk TN. A Meta. Analysis of Probiotic Efficacy for Gastrointestinal Diseases. Heimesaat MM, ed. PLo. S ONE. 2012; 7(4): e 34938. doi: 10. 1371/journal. pone. 0034938.

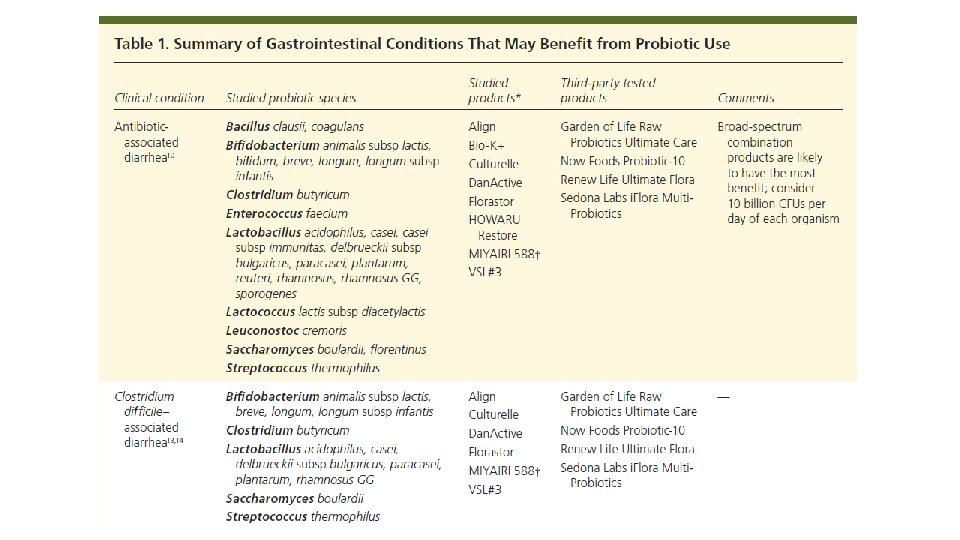
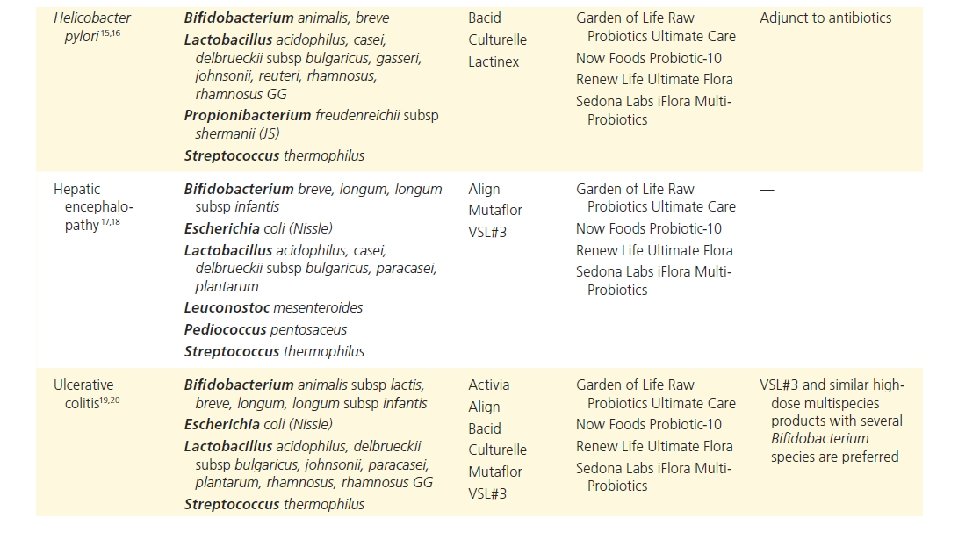
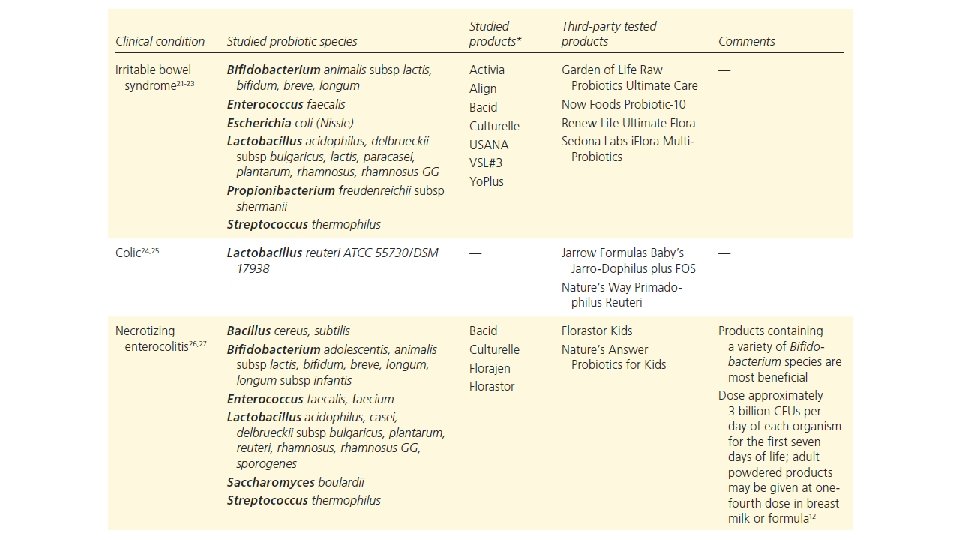
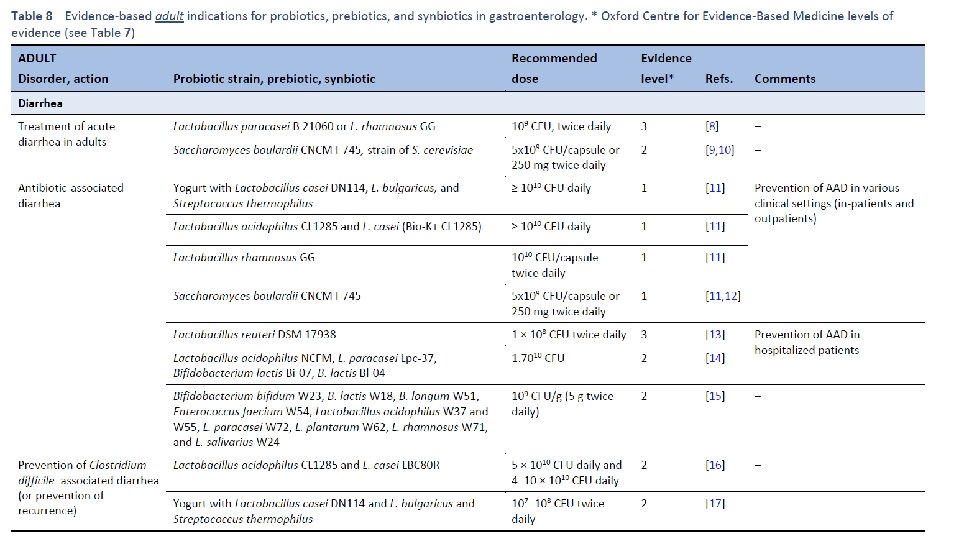
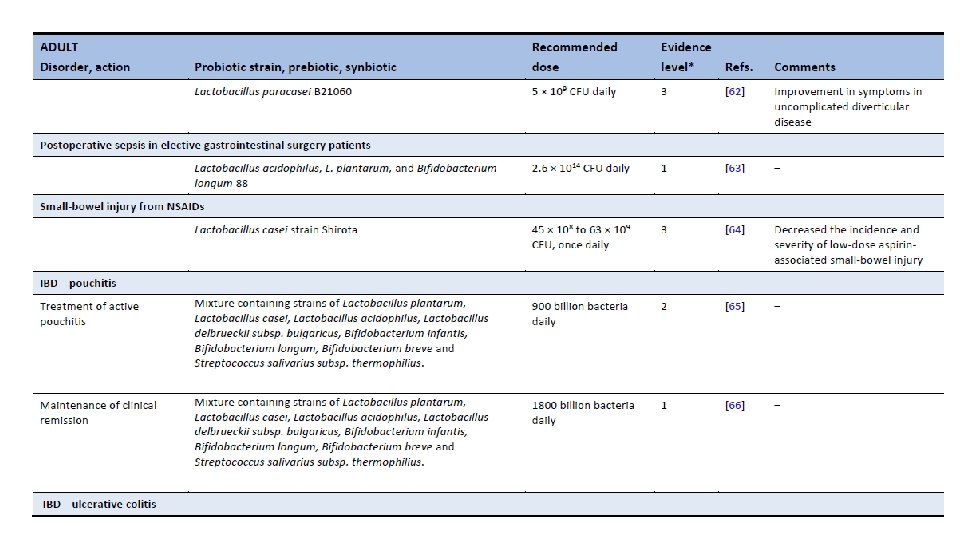
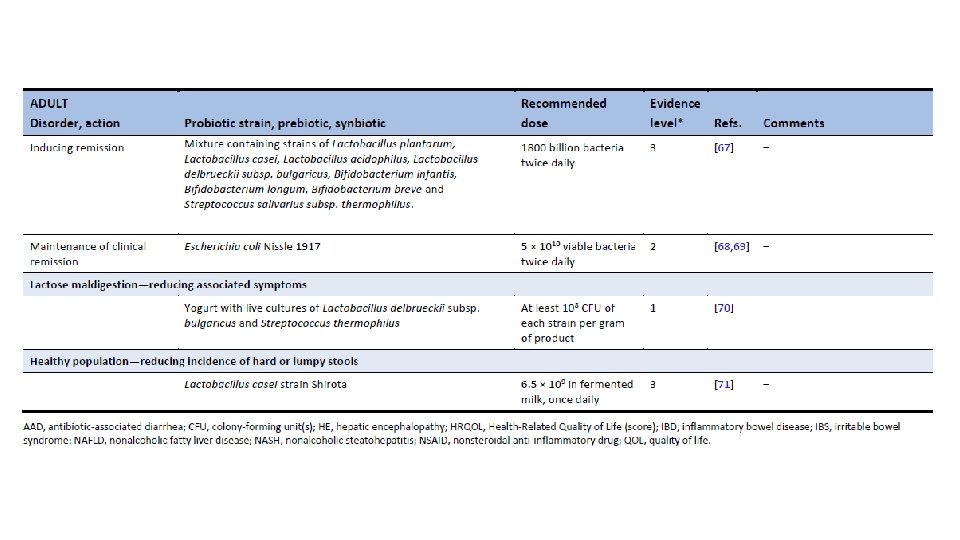
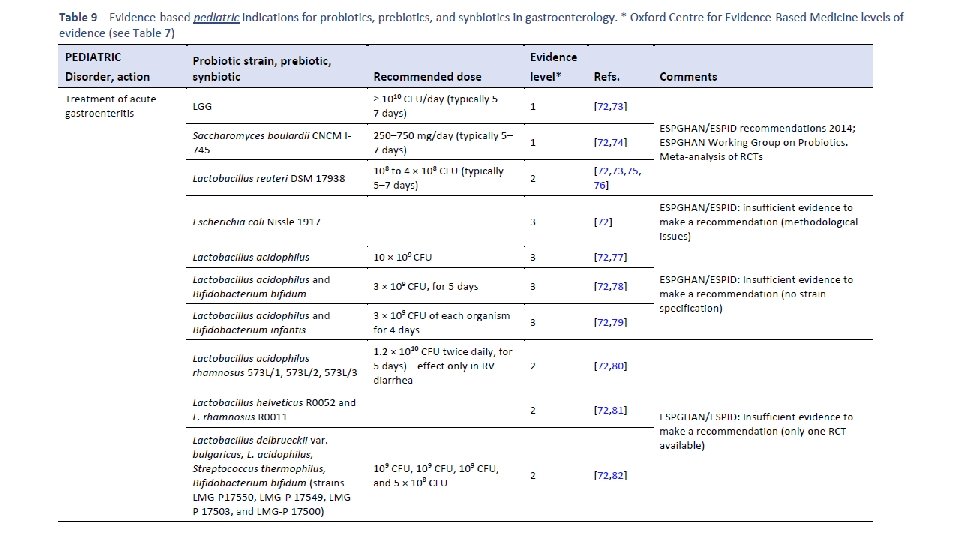
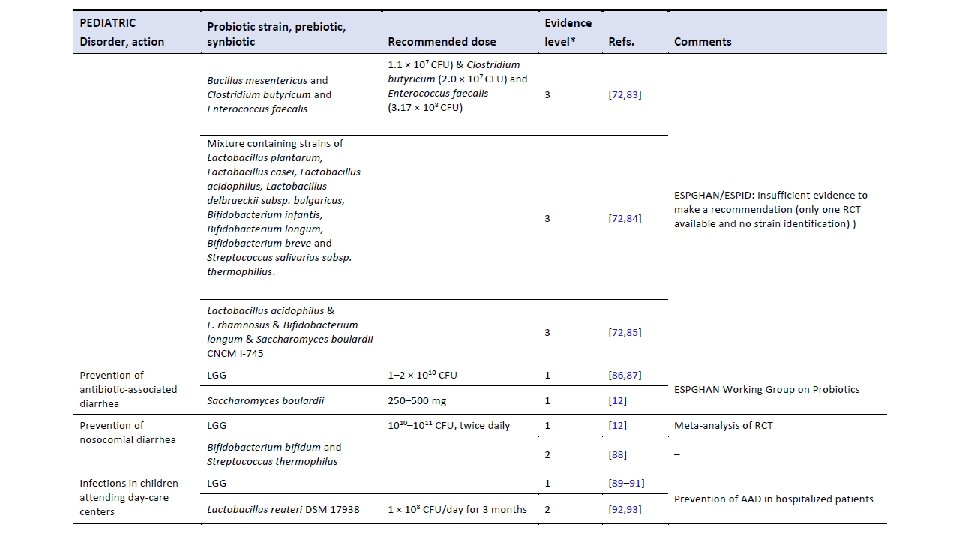
Am J Gastroenterol. 2006 Apr; 101(4): 812 -22. Meta-analysis of probiotics for the prevention of antibiotic associated diarrhea and the treatment of Clostridium difficile disease. Mc. Farland LV 1. Department of Health Services Research and Development, Veterans Administration Puget Sound Health Care System, Seattle, Washington 98101, USA. CONTEXT: Antibiotic-associated diarrhea (AAD) is a common complication of most antibiotics and Clostridium difficile disease (CDD), which also is incited by antibiotics, is a leading cause of nosocomial outbreaks of diarrhea and colitis. The use of probiotics for these two related diseases remains controversial. OBJECTIVE: To compare the efficacy of probiotics for the prevention of AAD and the treatment of CDD based on the published randomized, controlled clinical trials. DATA SOURCES: Pub. Med, Medline, Google Scholar, NIH registry of clinical trials, meta. Register, and Cochrane Central Register of Controlled Trials were searched from 1977 to 2005, unrestricted by language. Secondary searches of reference lists, authors, reviews, commentaries, associated diseases, books, and meeting abstracts. STUDY SELECTION: Trials were included in which specific probiotics given to either prevent or treat the diseases of interest. Trials were required to be randomized, controlled, blinded efficacy trials in humans published in peer-reviewed journals. Trials that were excluded were pre-clinical, safety, Phase 1 studies in volunteers, reviews, duplicate reports, trials of unspecified probiotics, trials of prebiotics, not the disease being studied, or inconsistent outcome measures. Thirty-one of 180 screened studies (totally 3, 164 subjects) met the inclusion and exclusion criteria. DATA EXTRACTION: One reviewer identified studies and abstracted data on sample size, population characteristics, treatments, and outcomes. DATA SYNTHESIS: From 25 randomized controlled trials (RCTs), probiotics significantly reduced the relative risk of AAD (RR = 0. 43, 95% CI 0. 31, 0. 58, p < 0. 001). From six randomized trials, probiotics had significant efficacy for CDD (RR = 0. 59, 95% CI 0. 41, 0. 85, p = 0. 005). CONCLUSION: A variety of different types of probiotics show promise as effective therapies for these two diseases. Using meta-analyses, three types of probiotics (Saccharomyces boulardii, Lactobacillus rhamnosus GG, and probiotic mixtures) significantly reduced the development of antibiotic-associated diarrhea. Only S. boulardii was effective for CDD.
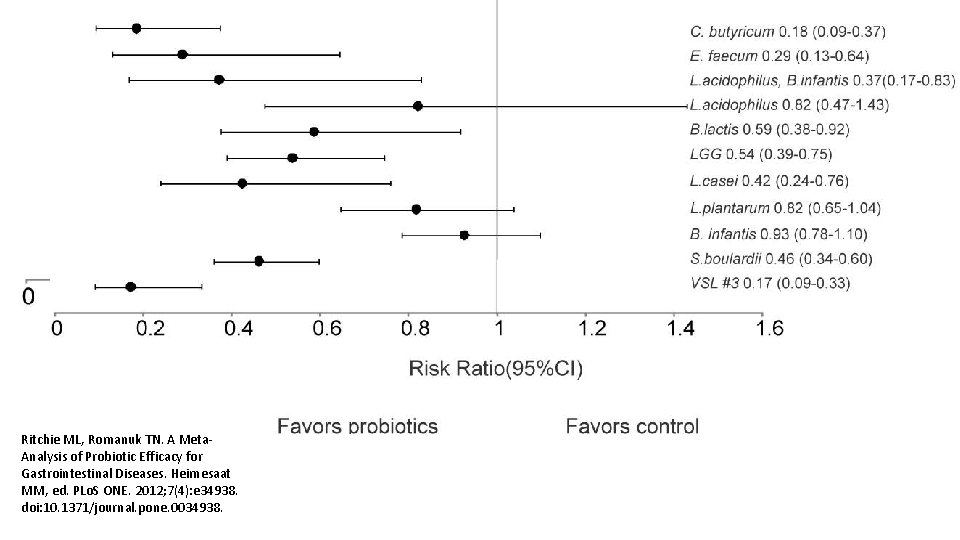
Ritchie ML, Romanuk TN. A Meta. Analysis of Probiotic Efficacy for Gastrointestinal Diseases. Heimesaat MM, ed. PLo. S ONE. 2012; 7(4): e 34938. doi: 10. 1371/journal. pone. 0034938.
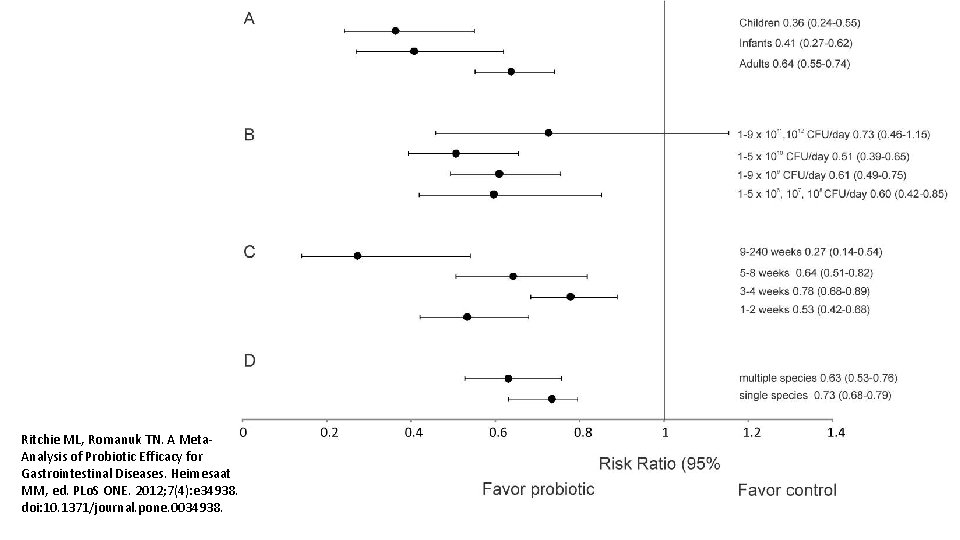
Ritchie ML, Romanuk TN. A Meta. Analysis of Probiotic Efficacy for Gastrointestinal Diseases. Heimesaat MM, ed. PLo. S ONE. 2012; 7(4): e 34938. doi: 10. 1371/journal. pone. 0034938.





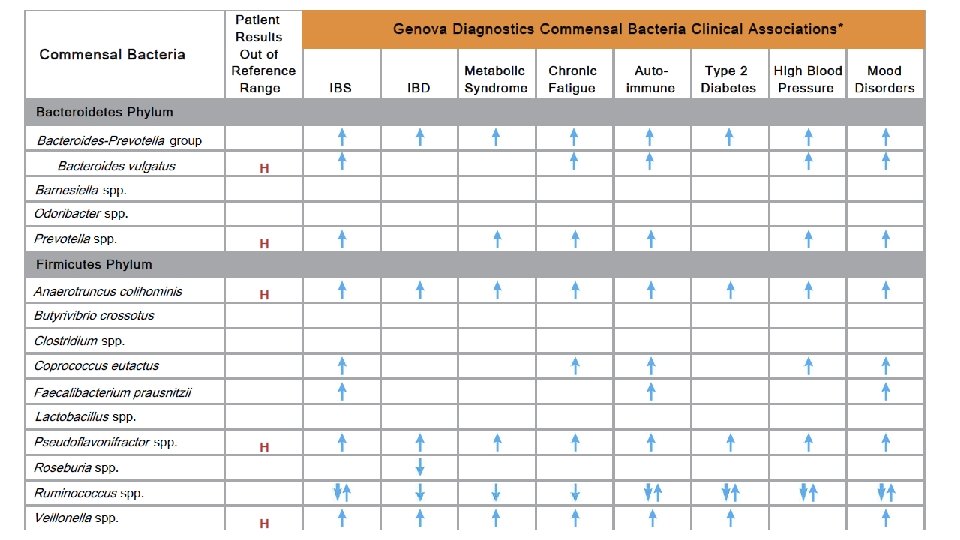
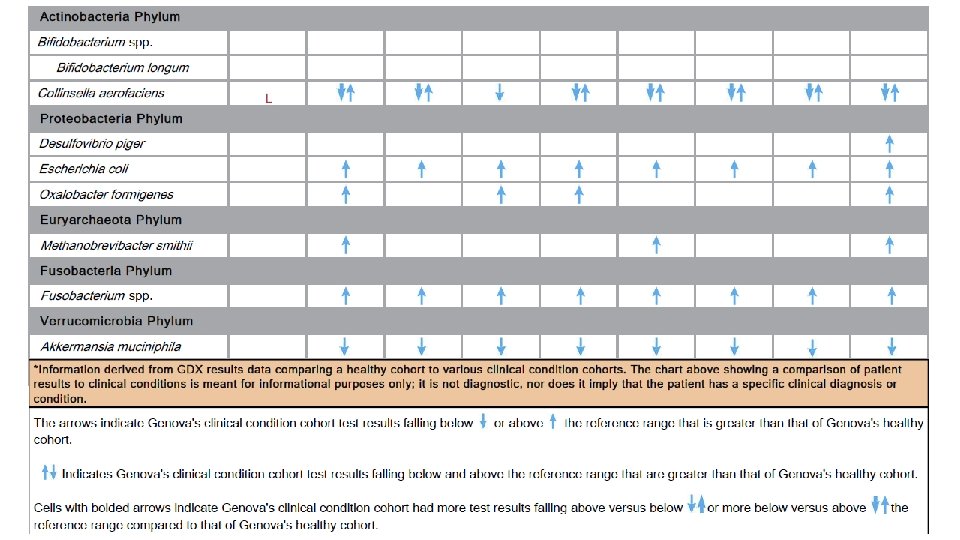
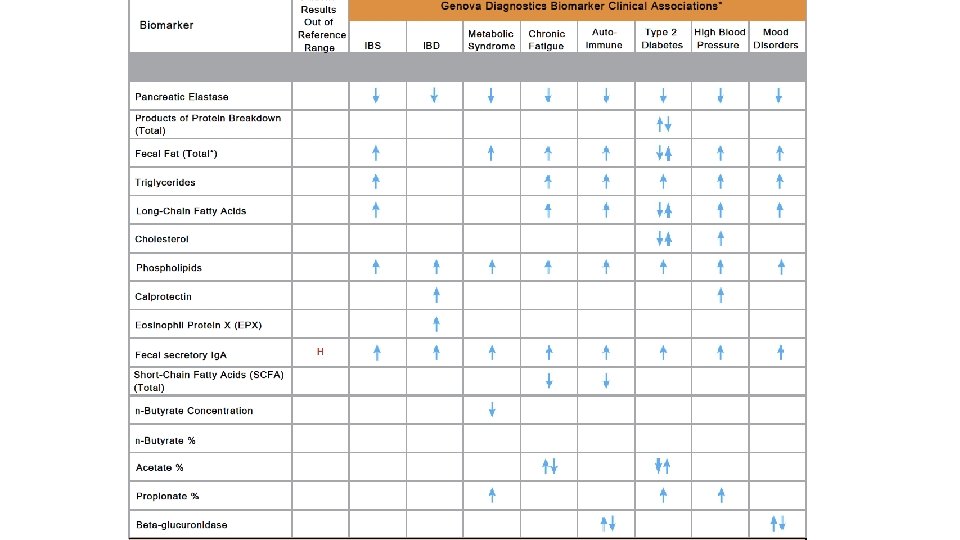
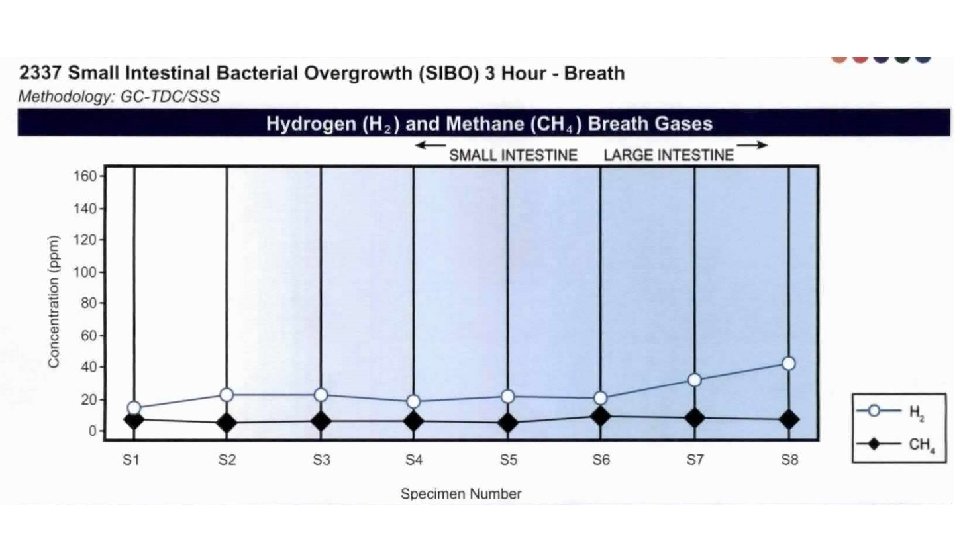
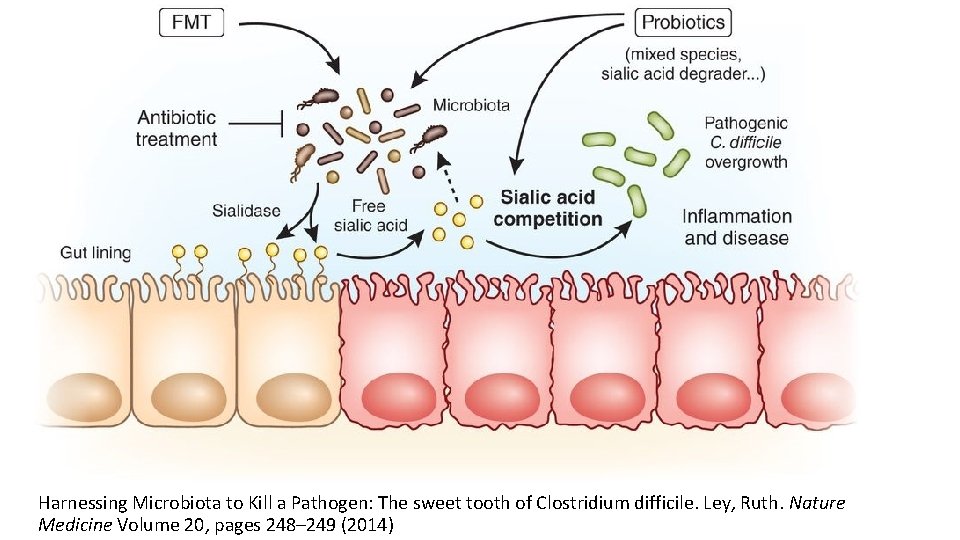
Harnessing Microbiota to Kill a Pathogen: The sweet tooth of Clostridium difficile. Ley, Ruth. Nature Medicine Volume 20, pages 248– 249 (2014)
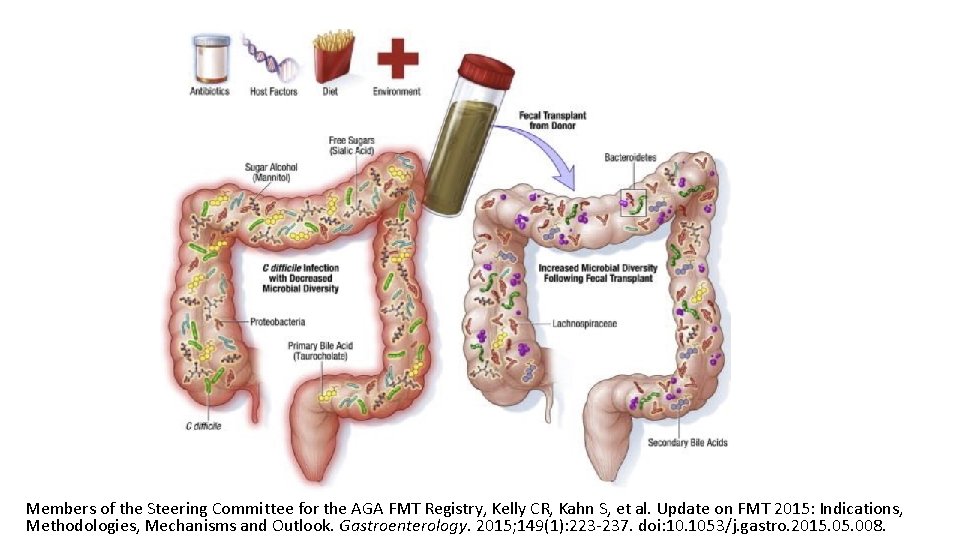
Members of the Steering Committee for the AGA FMT Registry, Kelly CR, Kahn S, et al. Update on FMT 2015: Indications, Methodologies, Mechanisms and Outlook. Gastroenterology. 2015; 149(1): 223 -237. doi: 10. 1053/j. gastro. 2015. 008.

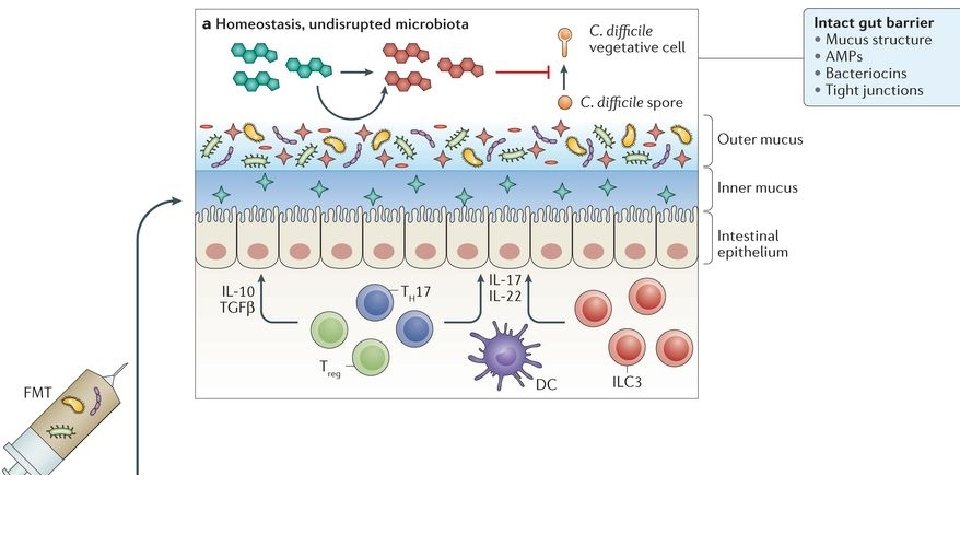
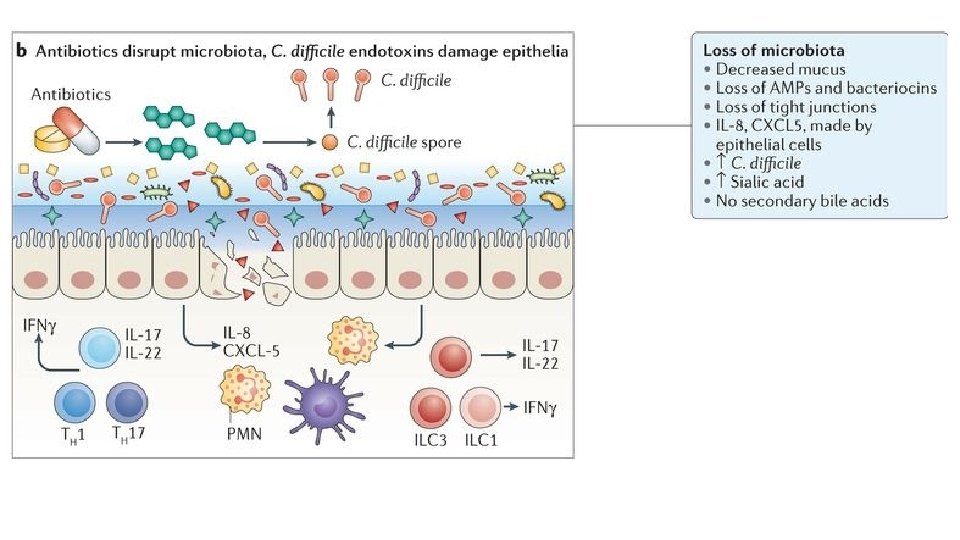
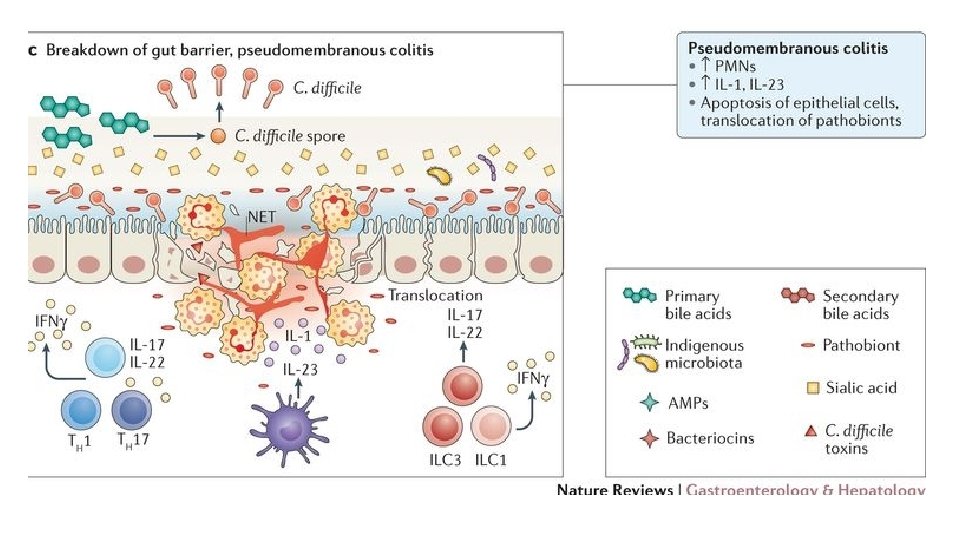
FMT for C. diff infection • Fecal microbiota transplantation (FMT) involves administration of the whole microbial community from healthy donor stool into the recipient's intestinal tract to normalize or modify intestinal microbiota composition and function • Overall suppression of microbiota and disruption of its community structure in the colon, most commonly resulting from antibiotic therapies, is the fundamental problem underlying the pathogenesis of Clostridium difficile infection (CDI) • FMT results in normalization of microbial diversity and community structure in patients being treated for CDI, with high rates of clinical cure • The restored colon microbial community could inhibit C. difficile by multiple mechanisms: competition for nutrients; direct suppression by antimicrobial peptides; bile-acid-mediated inhibition of spore germination and vegetative growth; and activation of immune-mediated colonization resistance Understanding the mechanisms of faecal microbiota transplantation. Khoruts A. & Sadowsky M. Nature Reviews Gastroenterology & Hepatology Volume 13, pages 508– 516 (2016)
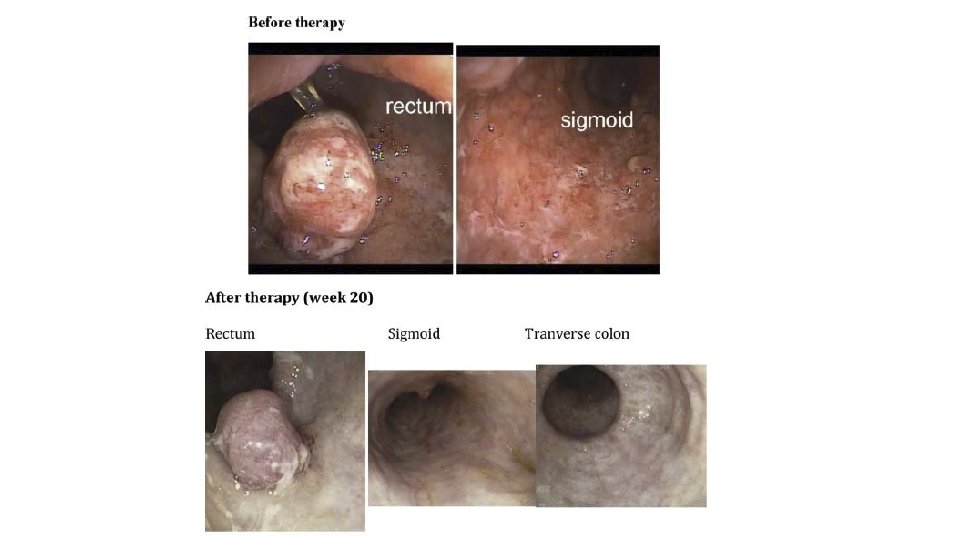


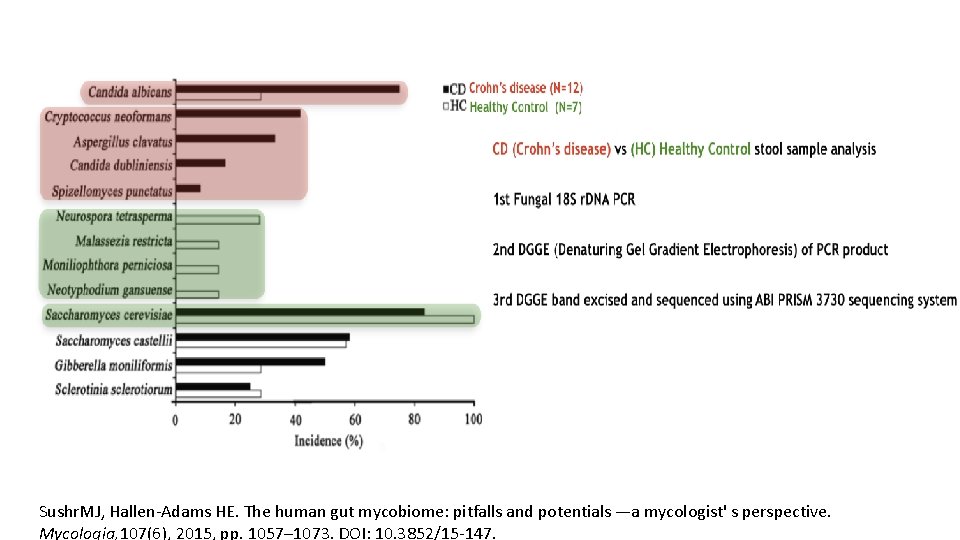
Sushr. MJ, Hallen-Adams HE. The human gut mycobiome: pitfalls and potentials —a mycologist' s perspective. Mycologia, 107(6), 2015, pp. 1057– 1073. DOI: 10. 3852/15 -147.
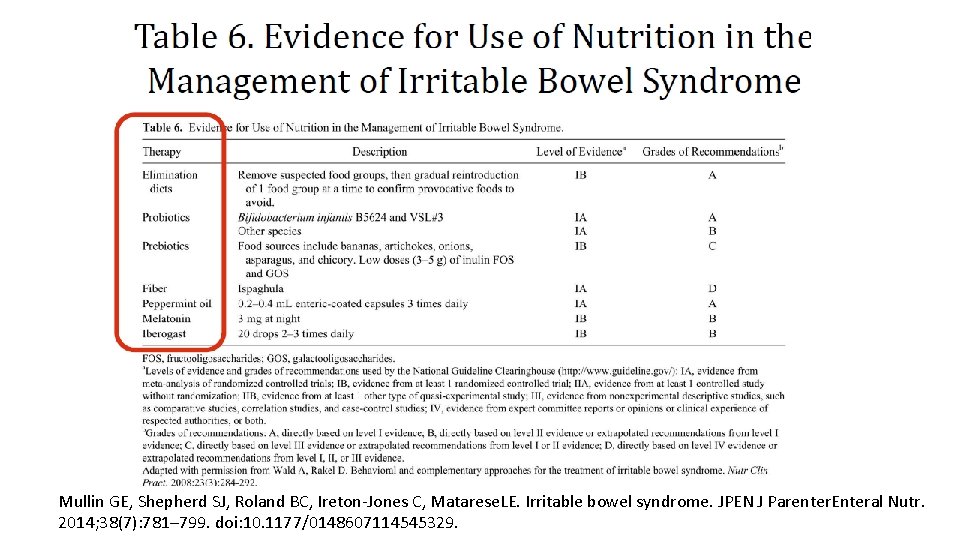
Mullin GE, Shepherd SJ, Roland BC, Ireton-Jones C, Matarese. LE. Irritable bowel syndrome. JPEN J Parenter. Enteral Nutr. 2014; 38(7): 781– 799. doi: 10. 1177/0148607114545329.
- Slides: 94