The Shapes of Molecules The shape of a











- Slides: 48

The Shapes of Molecules The shape of a molecule has an important bearing on its reactivity and behavior. The shape of a molecule depends a number of factors. These include: 1. Atoms forming the bonds 2. Bond distance 3. Bond angles 1

Valence Shell Electron Pair Repulsion (VSEPR) theory can be used to predict the geometric shapes of molecules. VSEPR that is revolves around the principle. One can predict the shape of a molecule by finding a pattern where electron pairs are as far from each other as possible. 2

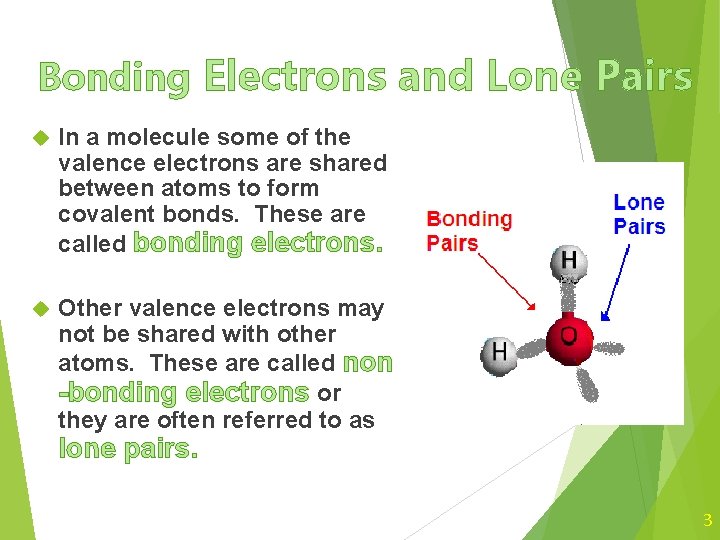
Bonding Electrons and Lone Pairs In a molecule some of the valence electrons are shared between atoms to form covalent bonds. These are called bonding electrons. Other valence electrons may not be shared with other atoms. These are called non -bonding electrons or they are often referred to as lone pairs. 3

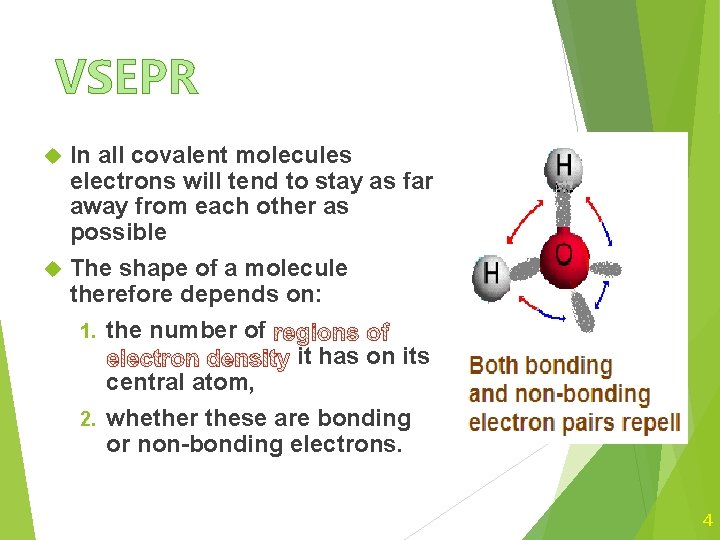
VSEPR In all covalent molecules electrons will tend to stay as far away from each other as possible The shape of a molecule therefore depends on: 1. the number of it has on its central atom, 2. whether these are bonding or non-bonding electrons. 4

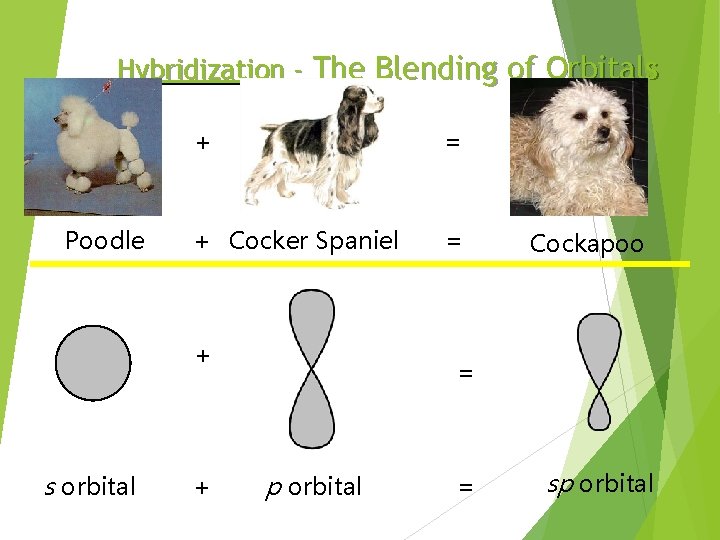
Hybridization - The Blending of Orbitals Poodle + = + Cocker Spaniel = + s orbital + Cockapoo = p orbital = sp orbital

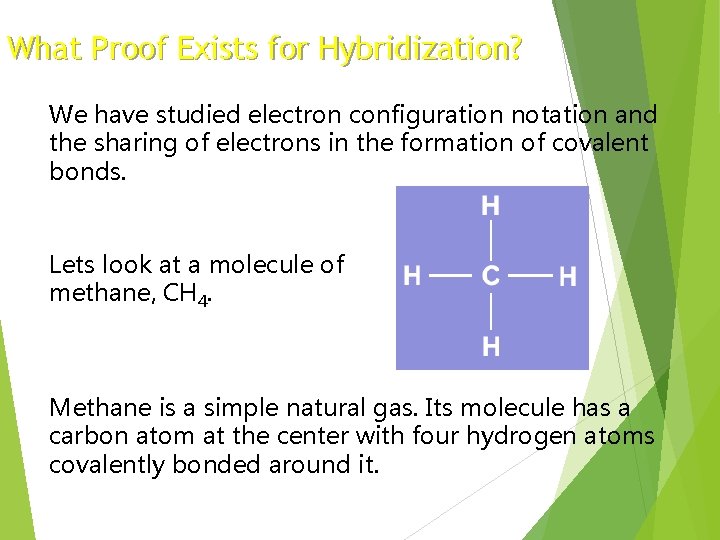
What Proof Exists for Hybridization? We have studied electron configuration notation and the sharing of electrons in the formation of covalent bonds. Lets look at a molecule of methane, CH 4. Methane is a simple natural gas. Its molecule has a carbon atom at the center with four hydrogen atoms covalently bonded around it.

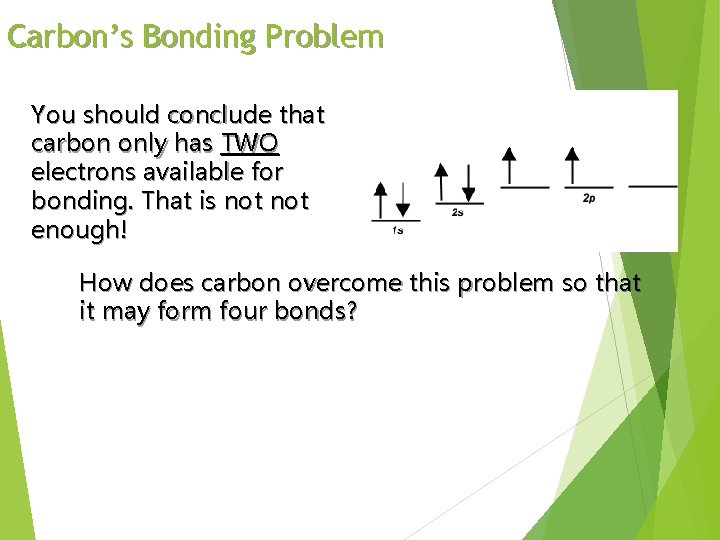
Carbon ground state configuration What is the expected orbital notation of carbon in its ground state? Can you see a problem with this? (Hint: How many unpaired electrons does this carbon atom have available for bonding? )

Carbon’s Bonding Problem You should conclude that carbon only has TWO electrons available for bonding. That is not enough! How does carbon overcome this problem so that it may form four bonds?

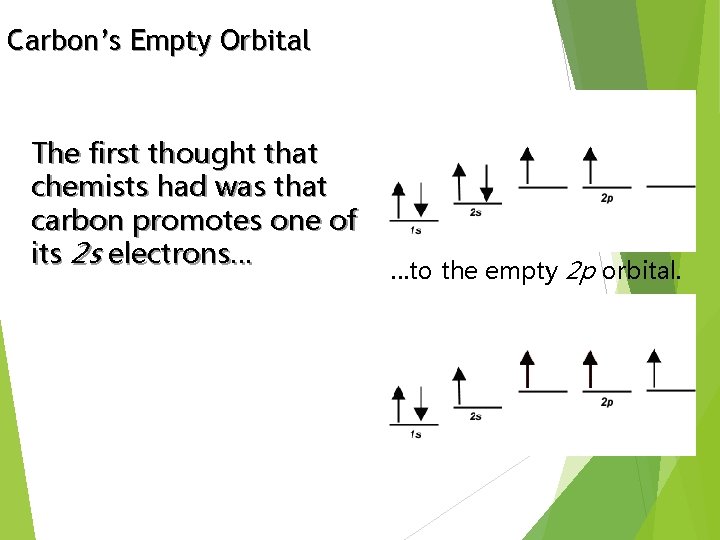
Carbon’s Empty Orbital The first thought that chemists had was that carbon promotes one of its 2 s electrons… …to the empty 2 p orbital.

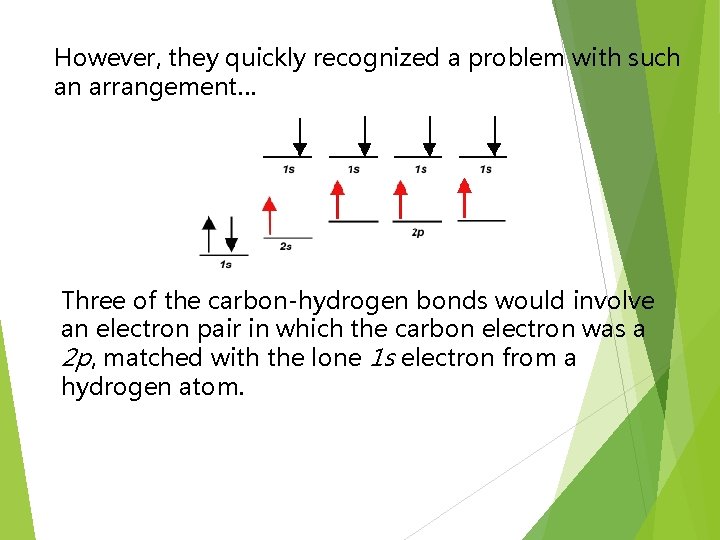
However, they quickly recognized a problem with such an arrangement… Three of the carbon-hydrogen bonds would involve an electron pair in which the carbon electron was a 2 p, matched with the lone 1 s electron from a hydrogen atom.

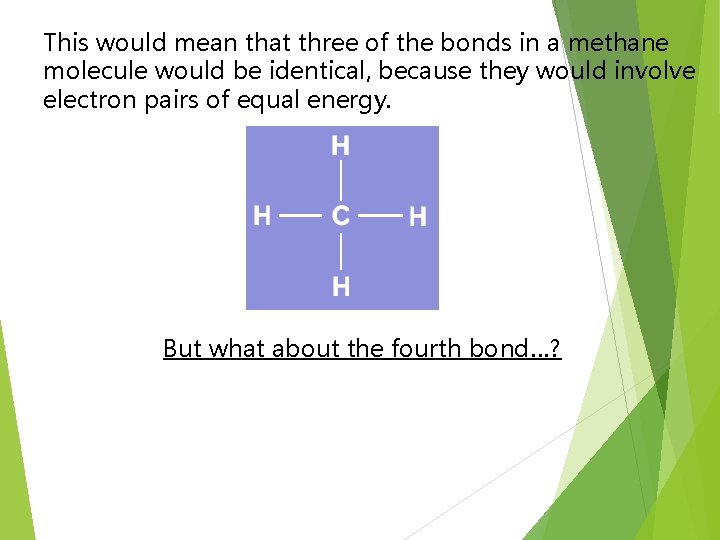
This would mean that three of the bonds in a methane molecule would be identical, because they would involve electron pairs of equal energy. But what about the fourth bond…?

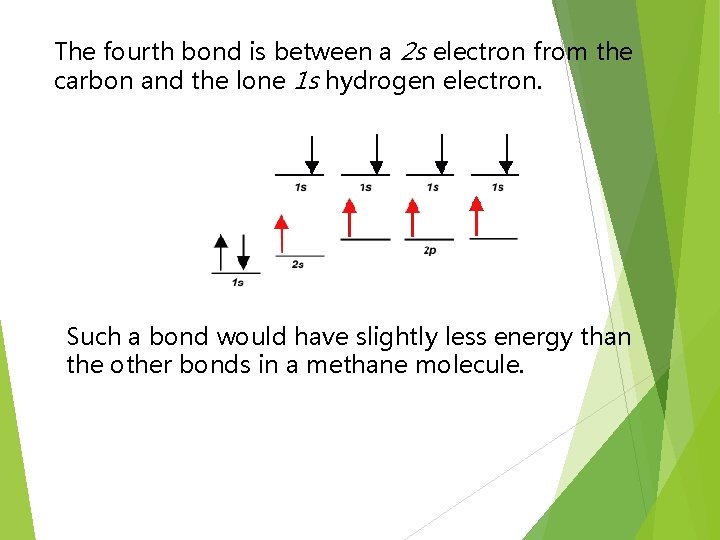
The fourth bond is between a 2 s electron from the carbon and the lone 1 s hydrogen electron. Such a bond would have slightly less energy than the other bonds in a methane molecule.

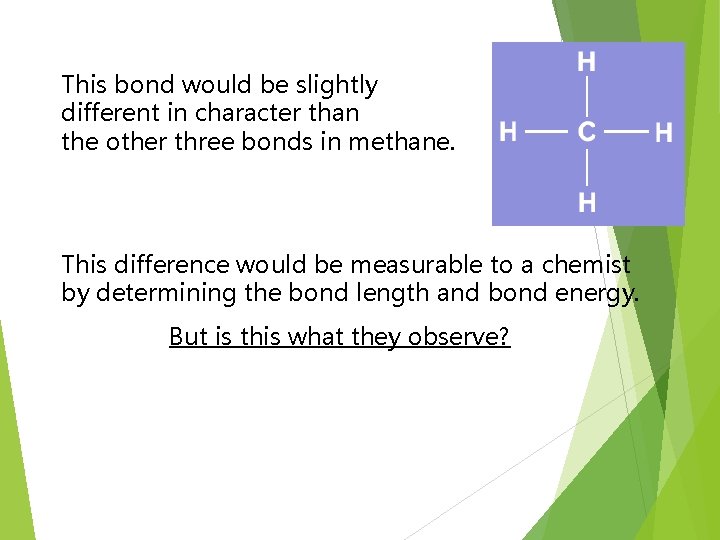
This bond would be slightly different in character than the other three bonds in methane. This difference would be measurable to a chemist by determining the bond length and bond energy. But is this what they observe?

The simple answer is, “No”. Measurements show that all four bonds in methane are equal. Thus, we need a new explanation for the bonding in methane. Chemists have proposed an explanation – they call it Hybridization is the combining of two or more orbitals of nearly equal energy within the same atom into orbitals of equal energy.

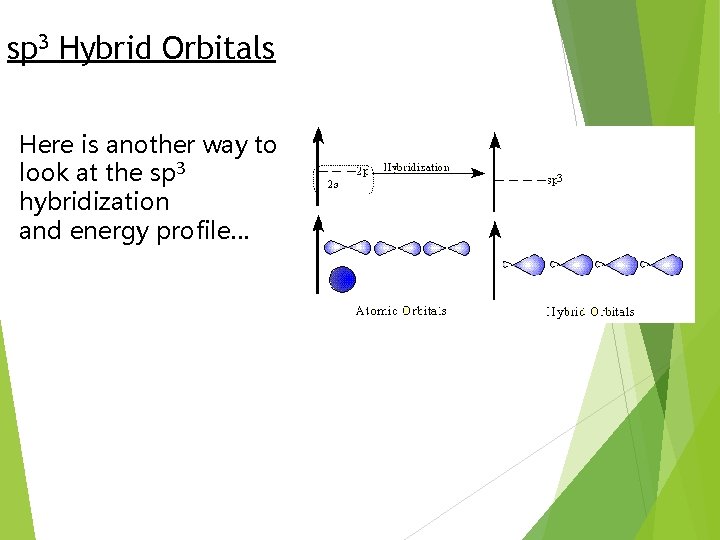
In the case of methane, they call the hybridization sp 3, meaning that an s orbital is combined with three p orbitals to create four equal hybrid orbitals. These new orbitals have slightly MORE energy than the 2 s orbital… … and slightly LESS energy than the 2 p orbitals.

sp 3 Hybrid Orbitals Here is another way to look at the sp 3 hybridization and energy profile…

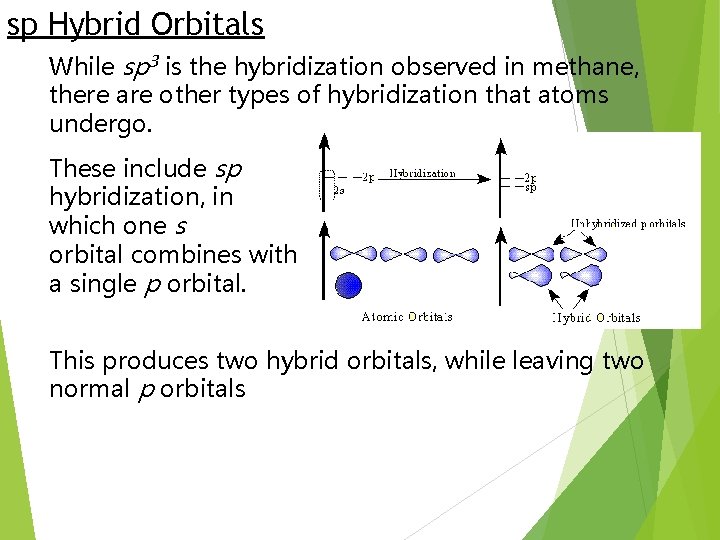
sp Hybrid Orbitals While sp 3 is the hybridization observed in methane, there are other types of hybridization that atoms undergo. These include sp hybridization, in which one s orbital combines with a single p orbital. This produces two hybrid orbitals, while leaving two normal p orbitals

sp 2 Hybrid Orbitals Another hybrid is the sp 2, which combines two orbitals from a p sublevel with one orbital from an s sublevel. One p orbital remains unchanged.

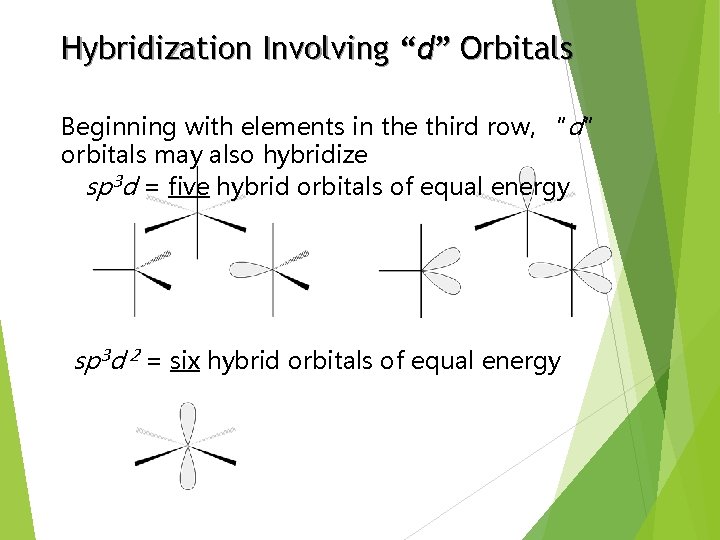
Hybridization Involving “d” Orbitals Beginning with elements in the third row, “d” orbitals may also hybridize sp 3 d = five hybrid orbitals of equal energy sp 3 d 2 = six hybrid orbitals of equal energy

VSEPR Predicting Shapes

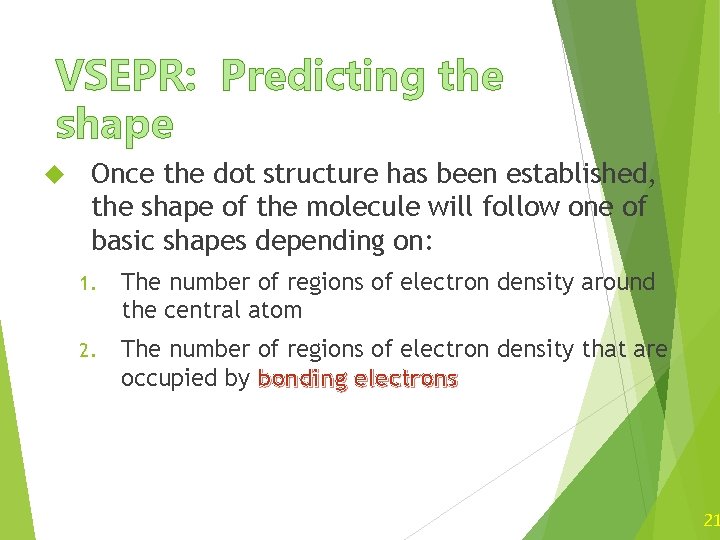
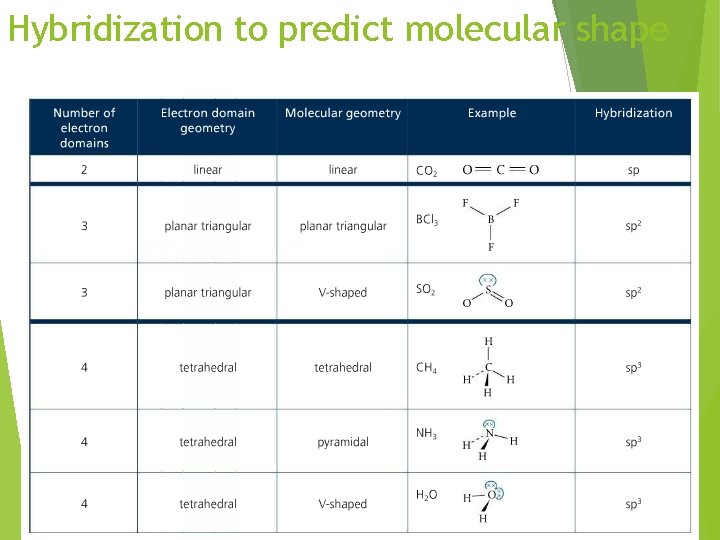
VSEPR: Predicting the shape Once the dot structure has been established, the shape of the molecule will follow one of basic shapes depending on: 1. The number of regions of electron density around the central atom 2. The number of regions of electron density that are occupied by bonding electrons 21

VSEPR: Predicting the shape The number of regions of electron density around the central atom determines the electron geometry The number of regions of electron density that are occupied by bonding electrons and hence other atoms determines the actual shape https: //phet. colorado. edu/sims/html/moleculeshapes/latest/molecule-shapes_en. html 22

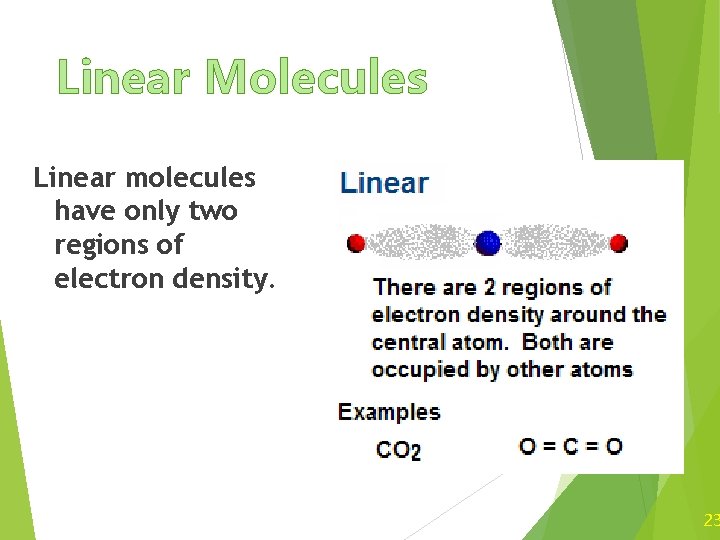
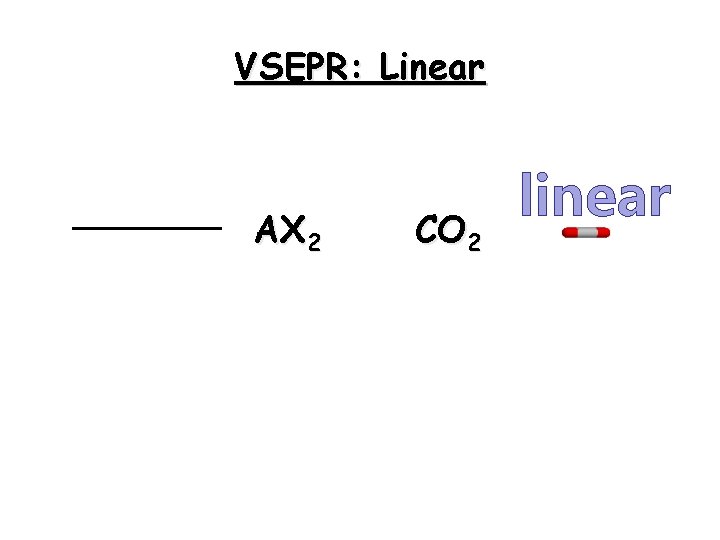
Linear Molecules Linear molecules have only two regions of electron density. 23

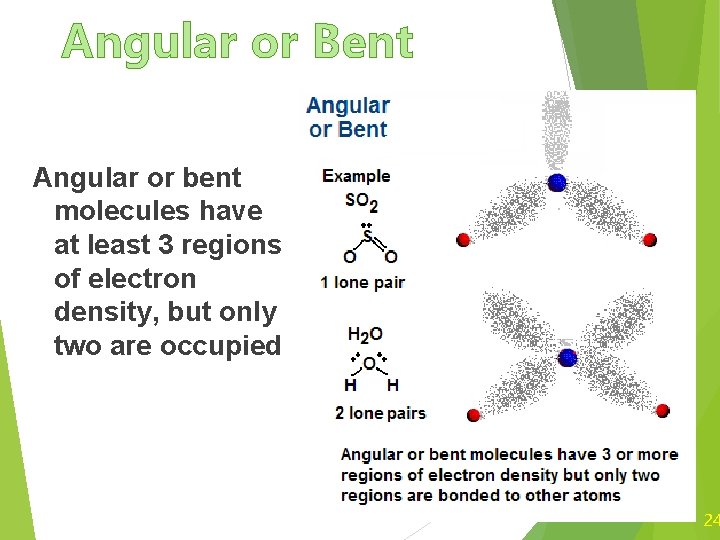
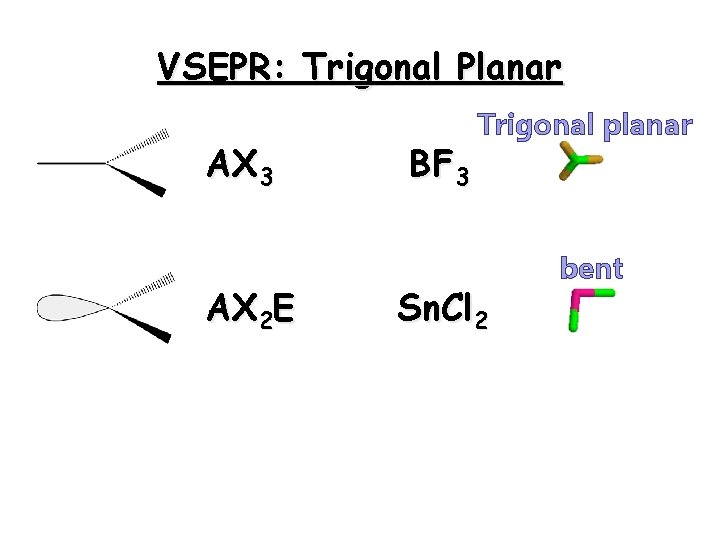
Angular or Bent Angular or bent molecules have at least 3 regions of electron density, but only two are occupied 24

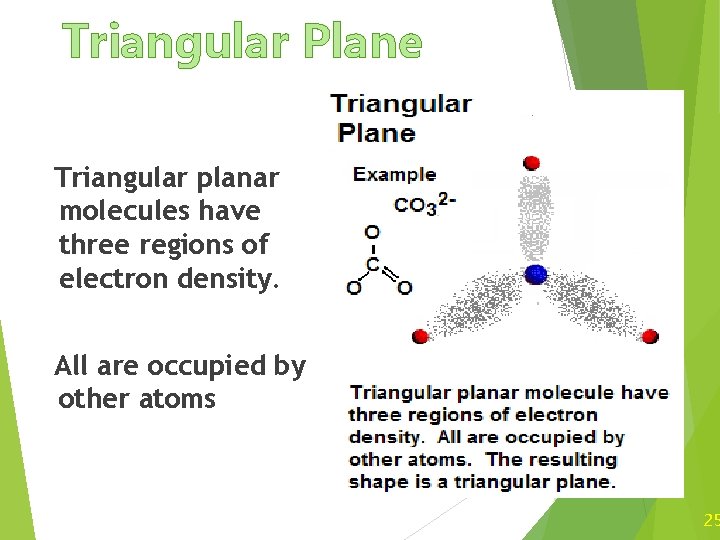
Triangular Plane Triangular planar molecules have three regions of electron density. All are occupied by other atoms 25

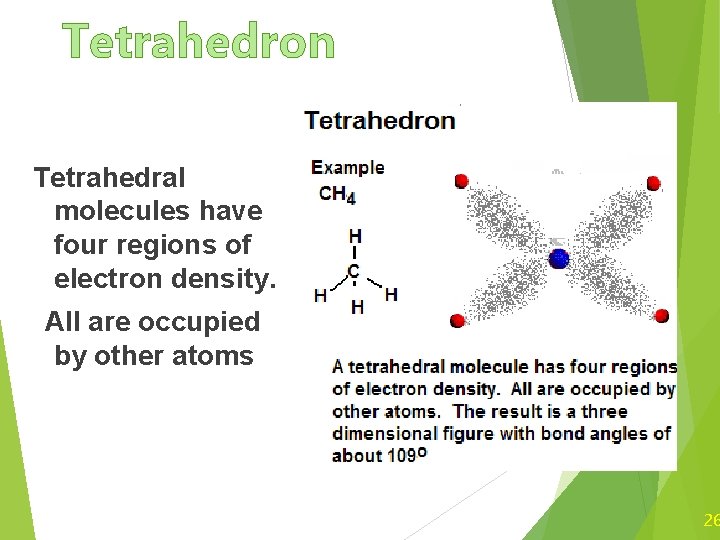
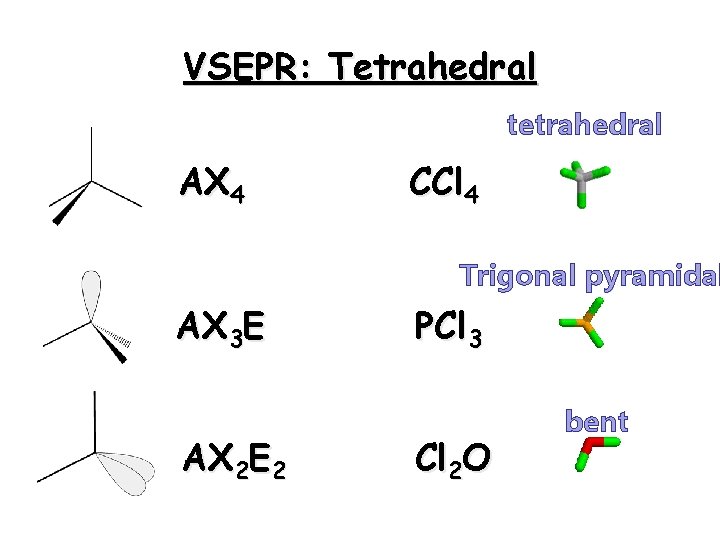
Tetrahedron Tetrahedral molecules have four regions of electron density. All are occupied by other atoms 26

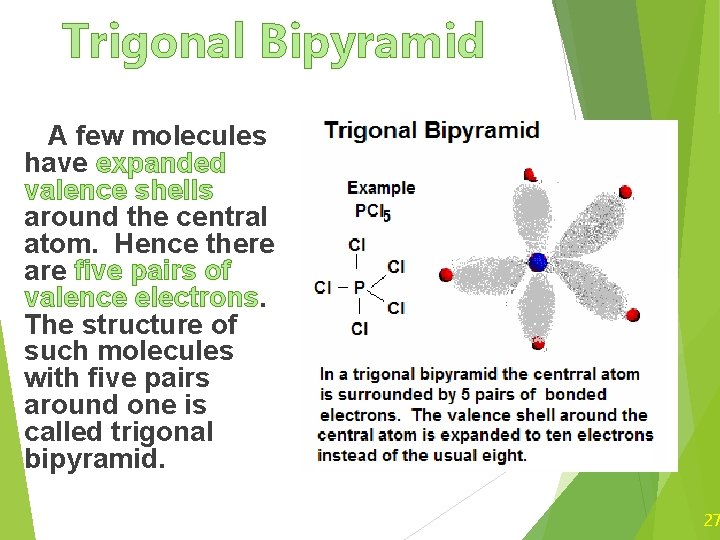
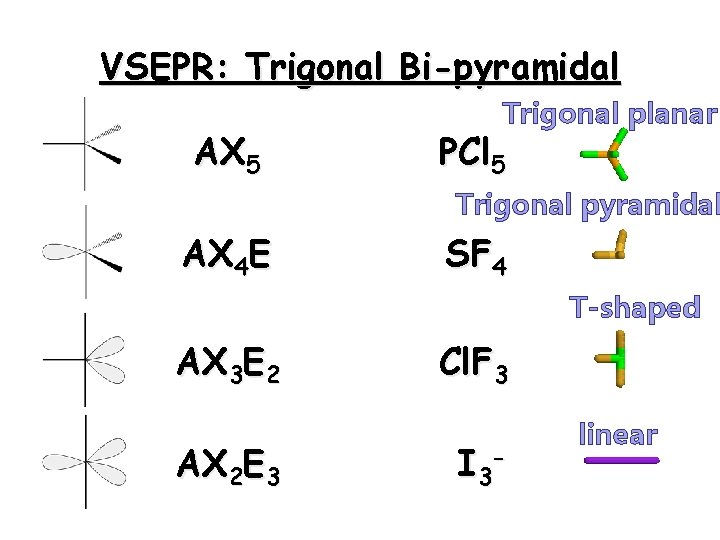
Trigonal Bipyramid A few molecules have expanded valence shells around the central atom. Hence there are five pairs of valence electrons. The structure of such molecules with five pairs around one is called trigonal bipyramid. 27

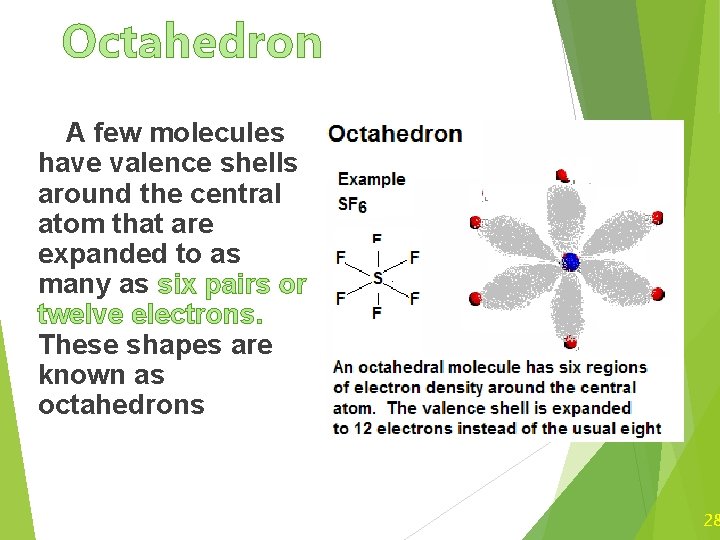
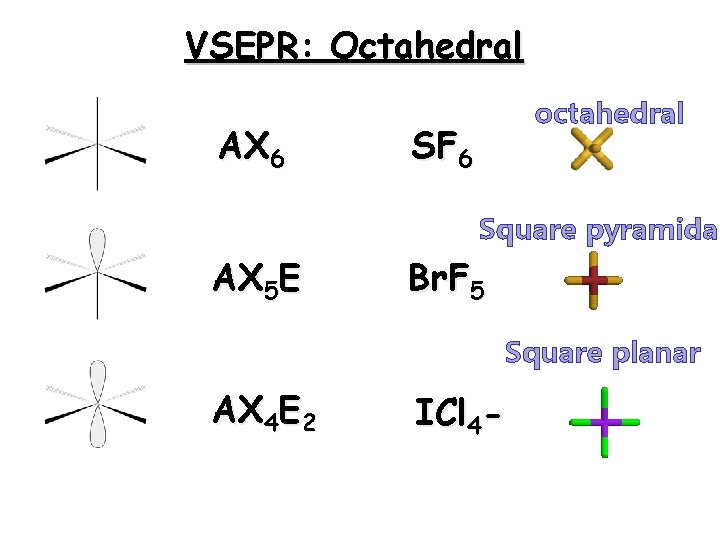
Octahedron A few molecules have valence shells around the central atom that are expanded to as many as six pairs or twelve electrons. These shapes are known as octahedrons 28

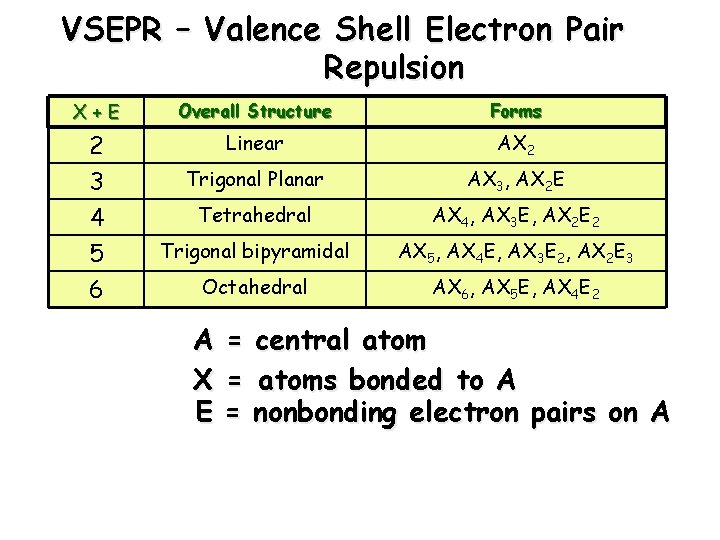
VSEPR – Valence Shell Electron Pair Repulsion X+E Overall Structure Forms 2 Linear AX 2 3 Trigonal Planar AX 3, AX 2 E 4 Tetrahedral AX 4, AX 3 E, AX 2 E 2 5 Trigonal bipyramidal AX 5, AX 4 E, AX 3 E 2, AX 2 E 3 6 Octahedral AX 6, AX 5 E, AX 4 E 2 A = central atom X = atoms bonded to A E = nonbonding electron pairs on A

VSEPR: Linear AX 2 CO 2 linear

VSEPR: Trigonal Planar AX 3 AX 2 E BF 3 Trigonal planar Sn. Cl 2 bent

VSEPR: Tetrahedral tetrahedral AX 4 CCl 4 Trigonal pyramidal AX 3 E AX 2 E 2 PCl 3 Cl 2 O bent

VSEPR: Trigonal Bi-pyramidal AX 5 Trigonal planar PCl 5 Trigonal pyramidal AX 4 E SF 4 T-shaped AX 3 E 2 AX 2 E 3 Cl. F 3 I 3 - linear

VSEPR: Octahedral AX 6 octahedral SF 6 Square pyramidal AX 5 E Br. F 5 Square planar AX 4 E 2 ICl 4 -

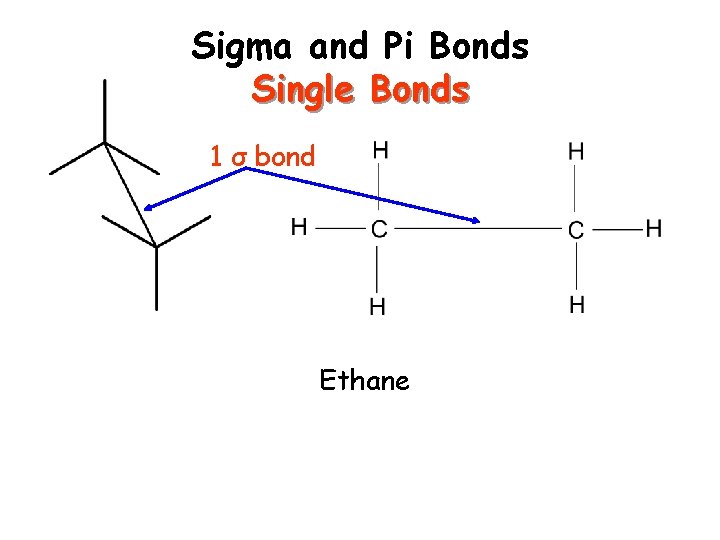
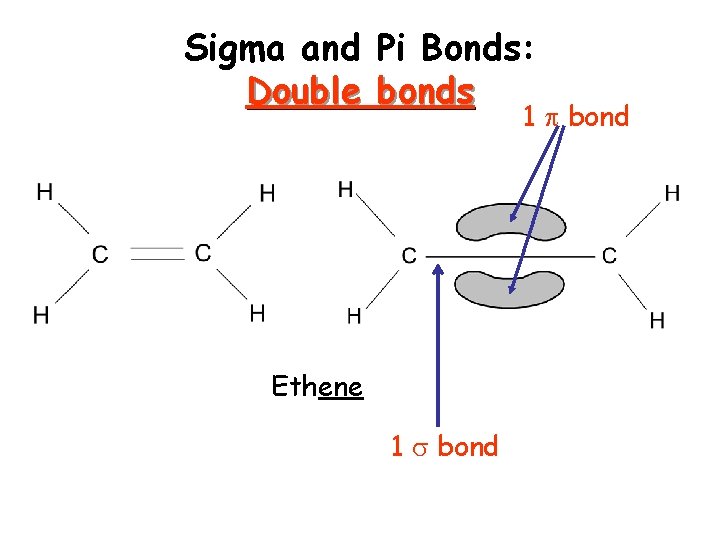
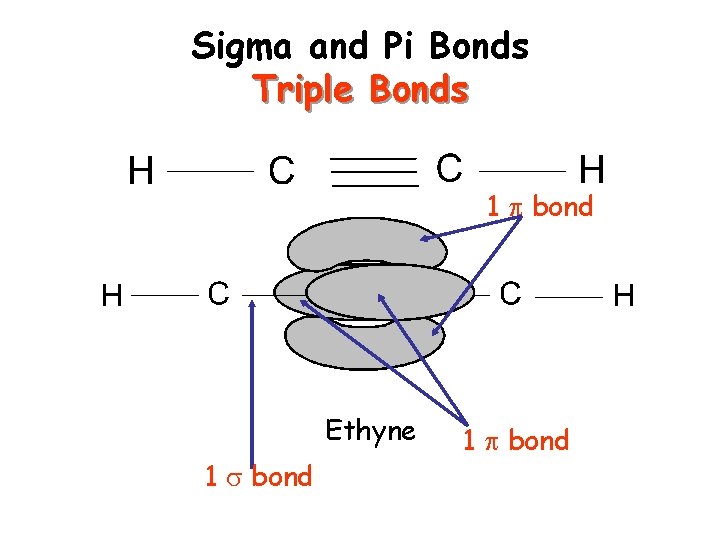
Sigma and Pi Bonds Sigma ( ) bonds exist in the region directly between two bonded atoms. Pi ( ) bonds exist in the region above and below a line drawn between two bonded atoms. Single bond 1 sigma bond Double Bond 1 sigma, 1 pi bond Triple Bond 1 sigma, 2 pi bonds Visual of how this happens…

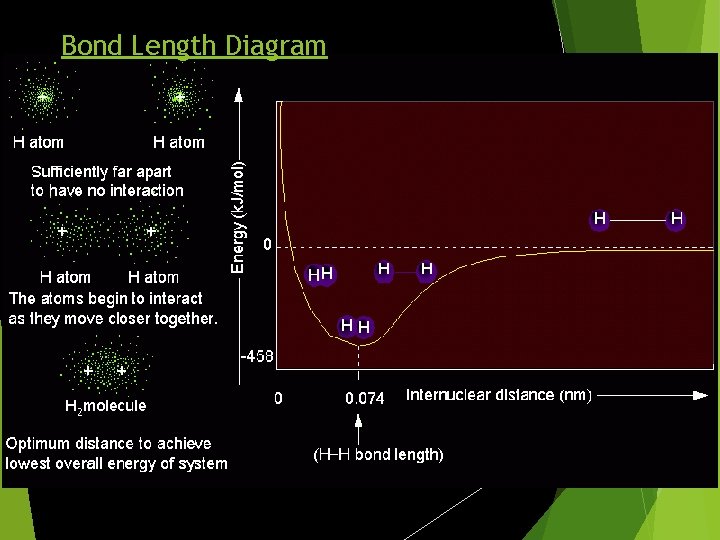
Bond Length Diagram

Atomic orbitals overlap When covalent bonds are formed, the orbitals overlap to create a new molecular orbital. Simply put, this happens because the new orbital is of lower energy. There are two main types.

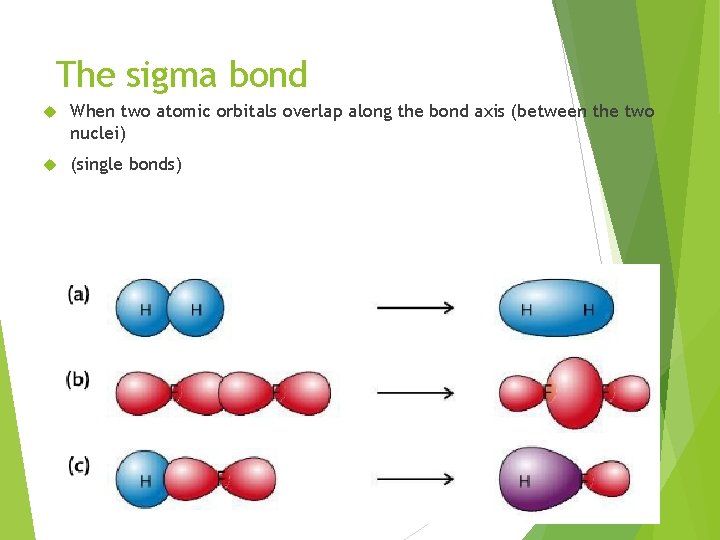
The sigma bond When two atomic orbitals overlap along the bond axis (between the two nuclei) (single bonds)

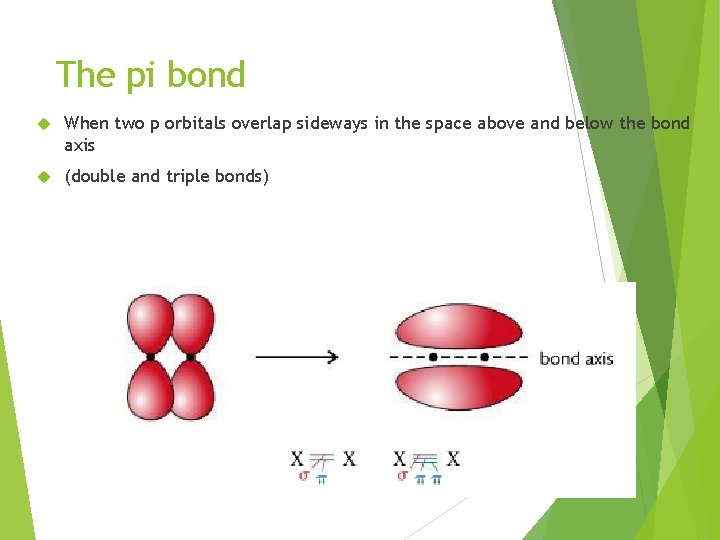
The pi bond When two p orbitals overlap sideways in the space above and below the bond axis (double and triple bonds)

Identifying sigma and pi orbitals

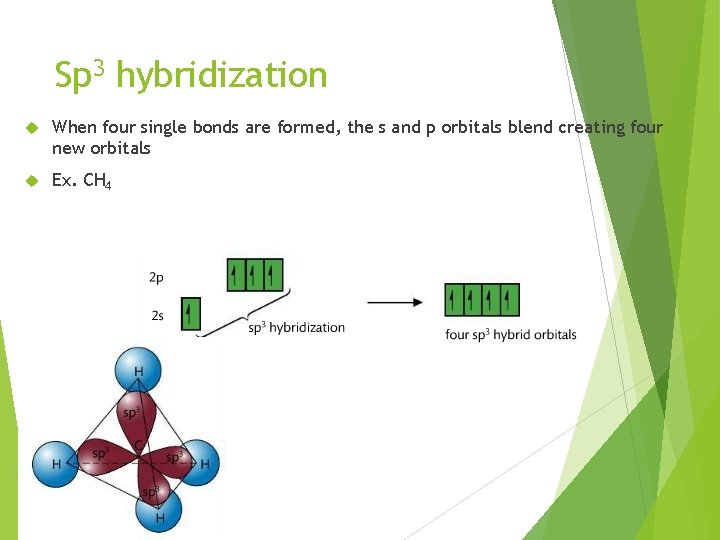
Sp 3 hybridization When four single bonds are formed, the s and p orbitals blend creating four new orbitals Ex. CH 4

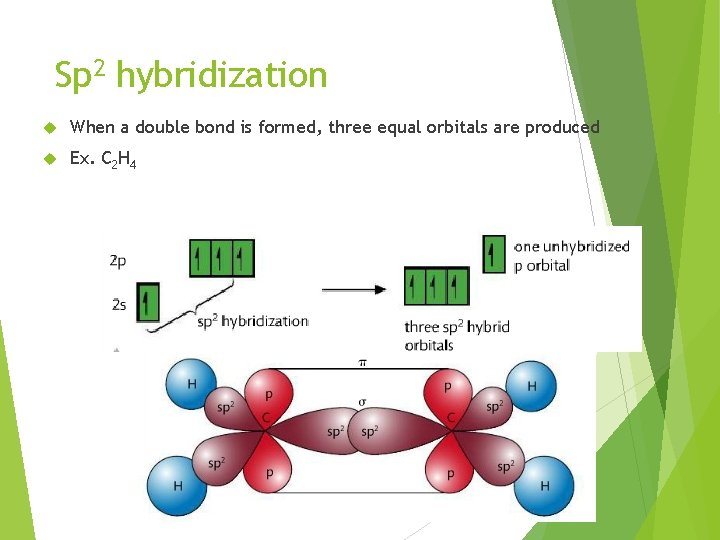
Sp 2 hybridization When a double bond is formed, three equal orbitals are produced Ex. C 2 H 4

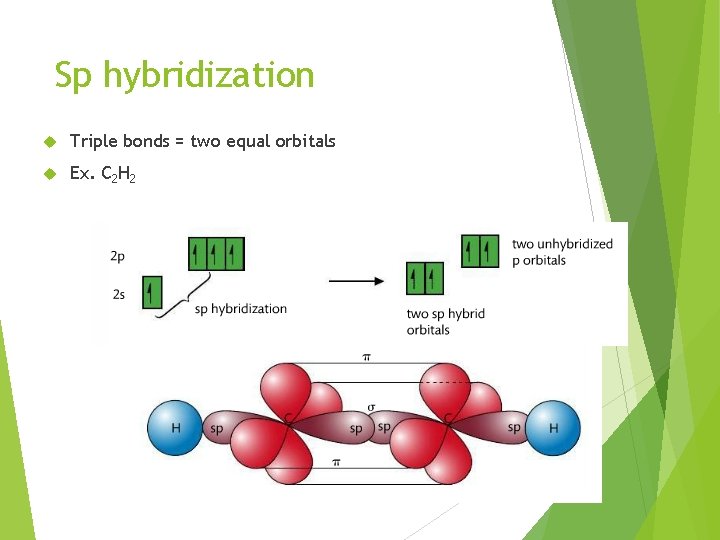
Sp hybridization Triple bonds = two equal orbitals Ex. C 2 H 2

Hybridization to predict molecular shape

Sigma and Pi Bonds Single Bonds 1 σ bond Ethane

Sigma and Double Pi Bonds: bonds 1 bond Ethene 1 bond

Sigma and Pi Bonds Triple Bonds 1 bond Ethyne 1 bond

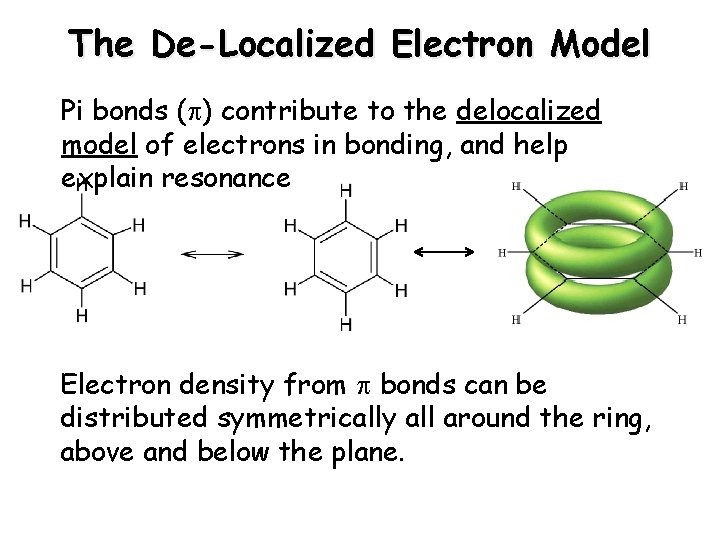
The De-Localized Electron Model Pi bonds ( ) contribute to the delocalized model of electrons in bonding, and help explain resonance Electron density from bonds can be distributed symmetrically all around the ring, above and below the plane.