The SAFER Trial SVG Angioplasty Free of Emboli

The SAFER Trial “SVG Angioplasty Free of Emboli Randomized” Evaluation of the Clinical Safety and Efficacy of the Percu. Surge Guard. Wire in Saphenous Vein Graft Intervention As presented at TCT 2000 by: Donald S. Baim, MD FACC Harvard Medical School Brigham and Women’s Hospital

The SAFER Trial Background • The average longevity of a vein graft is 8 -10 years • 40% of occlude • 75% develop severe narrowing • Vein graft atherosclerosis is diffuse and friable • Intervention may cause distal embolization • Embolization compromises the distal microcirculation • Manifest as no-reflow (8 -10%) and CK elevation (17 -20%) • Mortality of 3. 4% at 30 days (14% with CK-MB > 3 x normal) A device that could capture and remove embolic particles before they reach the myocardium could reduce these complications
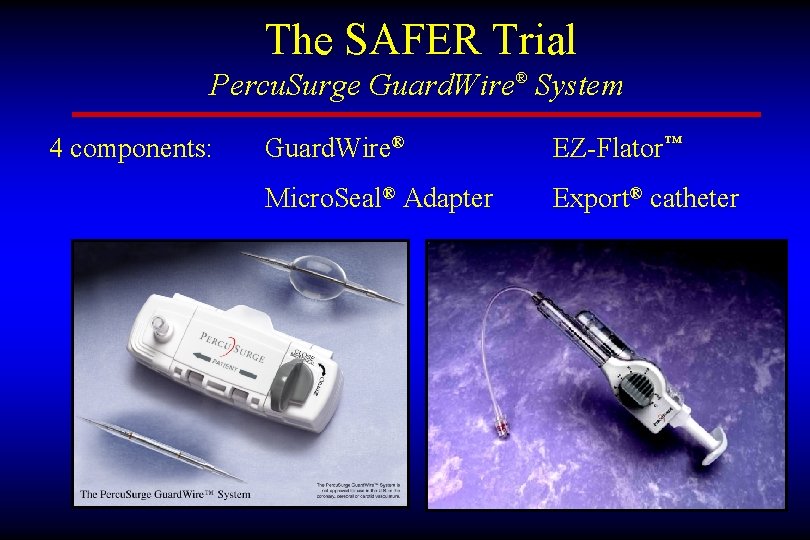
The SAFER Trial Percu. Surge Guard. Wire® System 4 components: Guard. Wire® EZ-Flator™ Micro. Seal® Adapter Export® catheter
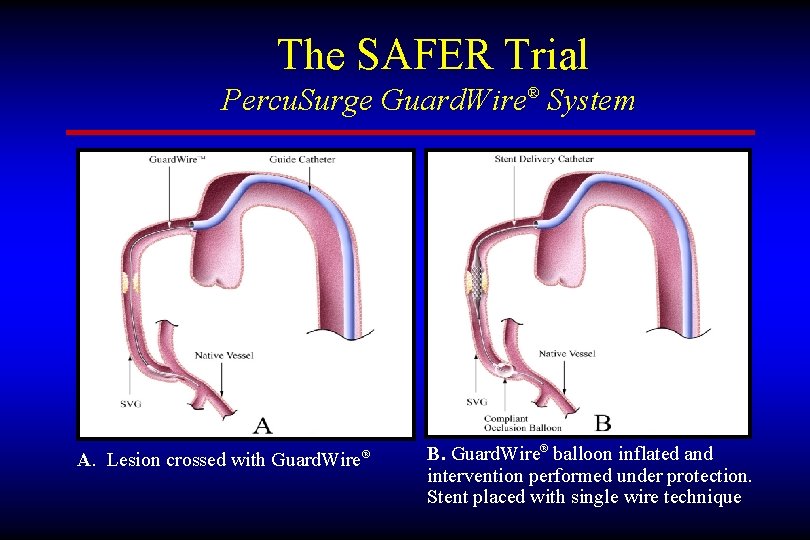
The SAFER Trial Percu. Surge Guard. Wire® System A. Lesion crossed with Guard. Wire® B. Guard. Wire® balloon inflated and intervention performed under protection. Stent placed with single wire technique
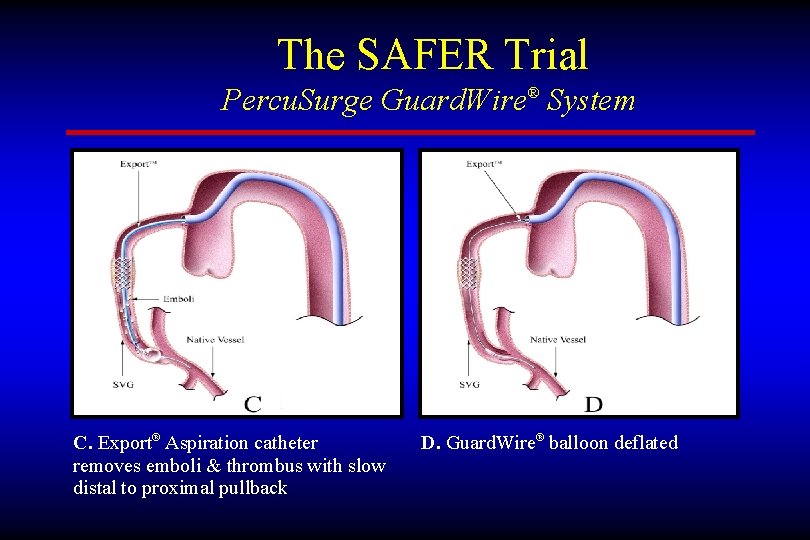
The SAFER Trial Percu. Surge Guard. Wire® System C. Export® Aspiration catheter removes emboli & thrombus with slow distal to proximal pullback D. Guard. Wire® balloon deflated
The SAFER Trial Percu. Surge Guard. Wire® System

The SAFER Trial Prior studies: Webb et al (JACC 34: 461, 1999) • single site study of 27 patients • MACE - 3. 7% SAFE (E. Grube, PI) • • 103 patient European study (7 sites) visible material removed in 95% of cases 81% of material is under 96 micron MACE - 4. 9 % (3. 9% non-Q) The randomized SAFER trial was designed as an 800 patient prospective randomized trial to determine if the Guard. Wire reduced the incidence of MACE compared to conventional, unprotected stenting

The SAFER Trial Study Management Study Coordination: Cardiovascular Data Analysis Center (CDAC) Boston, MA QCA Core Lab: Brigham and Women’s Angiographic Core Laboratory (CDAC) Boston, MA ECG Core Lab: Cardiovascular Data Analysis Center (CDAC) Boston, MA Study Monitoring: Bailer Monitoring Lake Hopatcong, NJ Sponsor: Percu. Surge, Inc. Sunnyvale, CA

The SAFER Trial Study Design Primary endpoint: “Major Adverse Clinical Events” (MACE) during index hospitalization A combined clinical endpoint defined as death, Q wave or Non Q-Wave MI (CK-MB > 3 x ULN), emergent bypass surgery, or repeat target vessel revascularization

The SAFER Trial Study Design Inclusion criteria Lesions (50 -100%) in SVG 3 -6 mm in diameter More than 5 mm from ostium; 20 mm from distal anastomosis At least TIMI 1 flow at baseline Exclusion criteria Ongoing MI with + CK-MB EF < 25%, Cr > 2. 5 unless on hemodialysis Planned use of atherectomy device Enrollment 551 patients at 47 sites
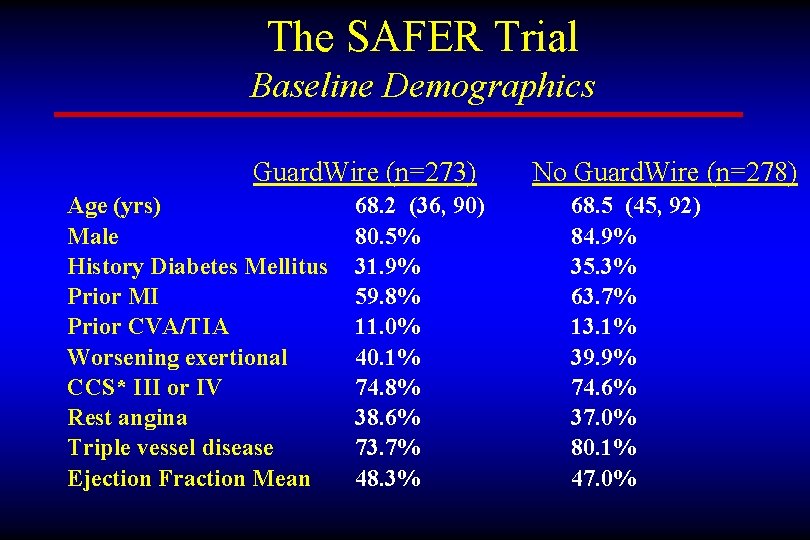
The SAFER Trial Baseline Demographics Guard. Wire (n=273) Age (yrs) Male History Diabetes Mellitus Prior MI Prior CVA/TIA Worsening exertional CCS* III or IV Rest angina Triple vessel disease Ejection Fraction Mean 68. 2 (36, 90) 80. 5% 31. 9% 59. 8% 11. 0% 40. 1% 74. 8% 38. 6% 73. 7% 48. 3% No Guard. Wire (n=278) 68. 5 (45, 92) 84. 9% 35. 3% 63. 7% 13. 1% 39. 9% 74. 6% 37. 0% 80. 1% 47. 0%
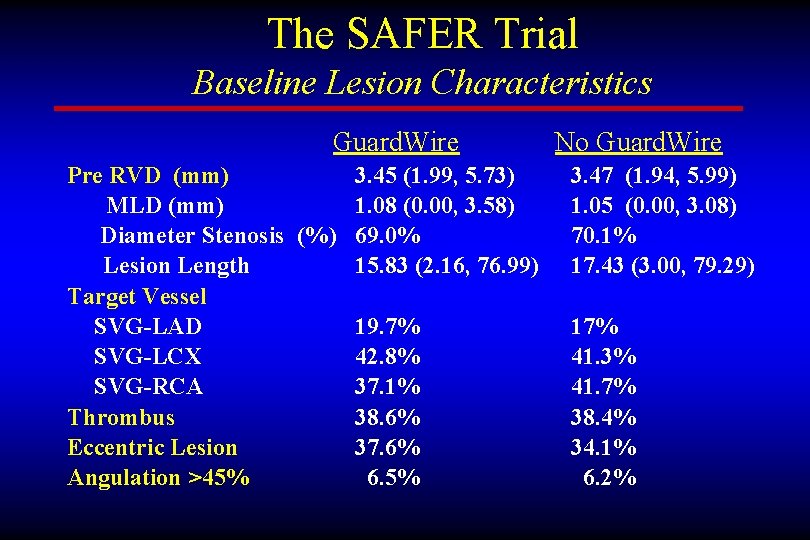
The SAFER Trial Baseline Lesion Characteristics Guard. Wire Pre RVD (mm) 3. 45 (1. 99, 5. 73) MLD (mm) 1. 08 (0. 00, 3. 58) Diameter Stenosis (%) 69. 0% Lesion Length 15. 83 (2. 16, 76. 99) Target Vessel SVG-LAD 19. 7% SVG-LCX 42. 8% SVG-RCA 37. 1% Thrombus 38. 6% Eccentric Lesion 37. 6% Angulation >45% 6. 5% No Guard. Wire 3. 47 (1. 94, 5. 99) 1. 05 (0. 00, 3. 08) 70. 1% 17. 43 (3. 00, 79. 29) 17% 41. 3% 41. 7% 38. 4% 34. 1% 6. 2%

The SAFER Trial Technical Success was defined as successful delivery of the Guard. Wire to the intended target site, inflation of the occlusion balloon, aspiration using the Export catheter and balloon deflation according to the Instructions For Use Procedure Success was defined as achievement of a final DS < 50% with no inhospital MACE Technical Success Guard. Wire 93. 0% No Guard. Wire n/a Procedure success 90. 5% 82. 0% Number of stents 1. 38 1. 35 Inflation time ~ 4 minutes, preliminary observation ~ 2% intolerant
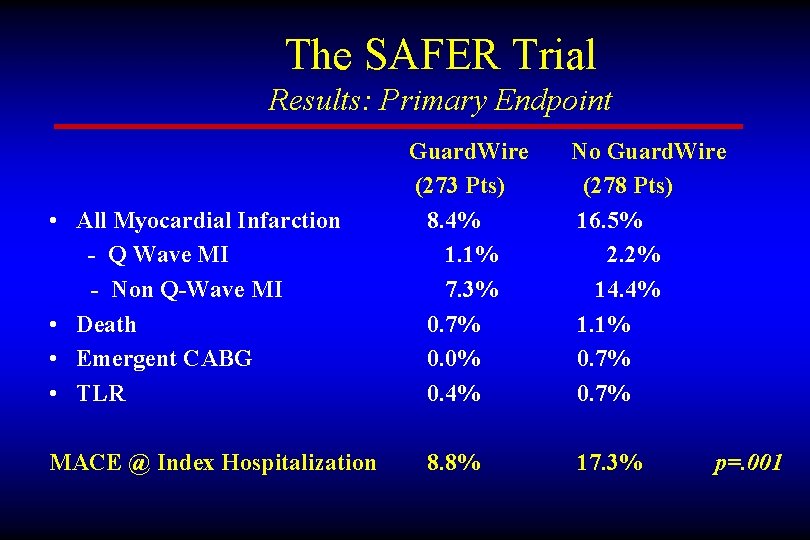
The SAFER Trial Results: Primary Endpoint • All Myocardial Infarction - Q Wave MI - Non Q-Wave MI • Death • Emergent CABG • TLR MACE @ Index Hospitalization Guard. Wire (273 Pts) 8. 4% 1. 1% 7. 3% 0. 7% 0. 0% 0. 4% 8. 8% No Guard. Wire (278 Pts) 16. 5% 2. 2% 14. 4% 1. 1% 0. 7% 17. 3% p=. 001
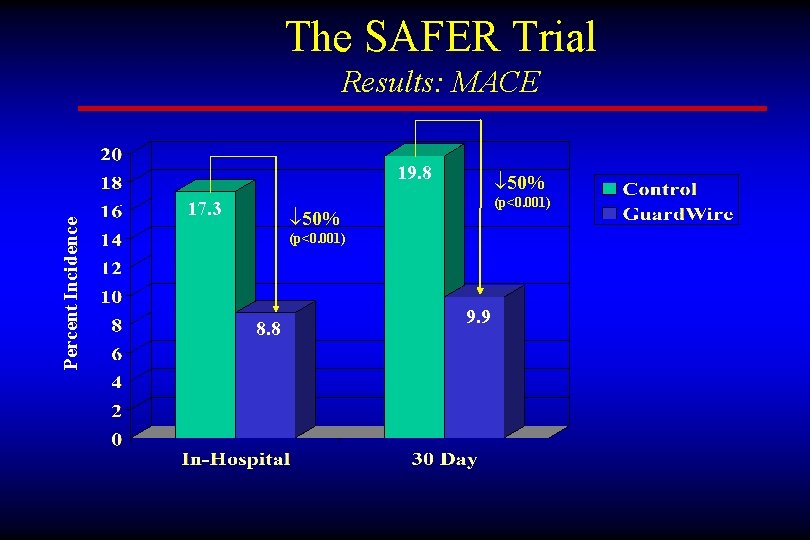
The SAFER Trial Results: MACE Percent Incidence 19. 8 17. 3 ¯ 50% (p<0. 001) 8. 8 9. 9
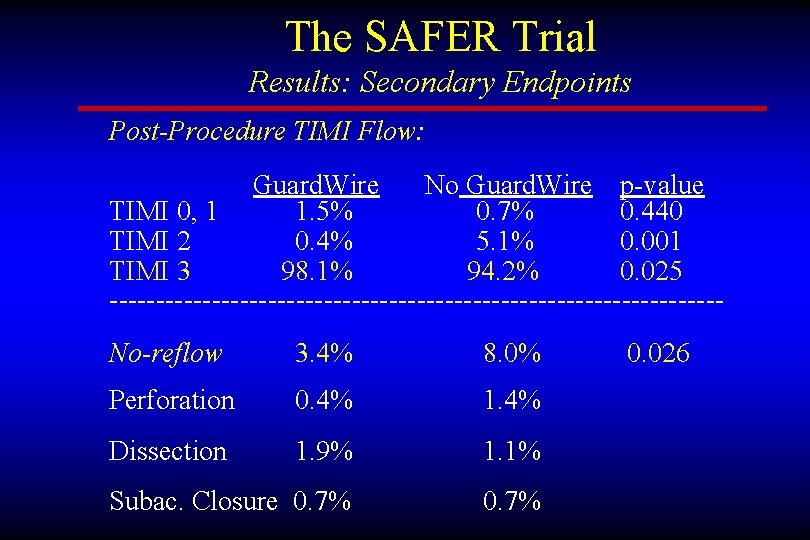
The SAFER Trial Results: Secondary Endpoints Post-Procedure TIMI Flow: Guard. Wire No Guard. Wire p-value TIMI 0, 1 1. 5% 0. 7% 0. 440 TIMI 2 0. 4% 5. 1% 0. 001 TIMI 3 98. 1% 94. 2% 0. 025 ---------------------------------No-reflow 3. 4% 8. 0% Perforation 0. 4% 1. 4% Dissection 1. 9% 1. 1% Subac. Closure 0. 7% 0. 026

The SAFER Trial GP IIb/IIIa Use GP IIb/IIIa inhibitor were used in >60% in both arms, mostly before intervention Percu. Surge had MACE benefit with or without them Guard. Wire No Guardwire p IIb/IIIa (164 – 182) 11. 6% 21. 6% 0. 007 No IIb/IIIa (109 – 96) 7. 3 13. 5% 0. 17 Why? GP IIb/IIIa’s prevent platelet thrombi, but they do not dissolve atherosclerotic plaque.

The SAFER Trial Conclusions The Percu. Surge Guard. Wire® system proved safe and effective in • recovering potentially embolic material • preserving normal flow • and reducing MACE by 50% during the percutaneous interventional treatment of saphenous vein bypass grafts This underscores the importance of distal embolization protection in SVG intervention
- Slides: 18