The Role of DPPIV Inhibitors in the Management

The Role of DPP-IV Inhibitors in the Management of Type 2 Diabetes 1
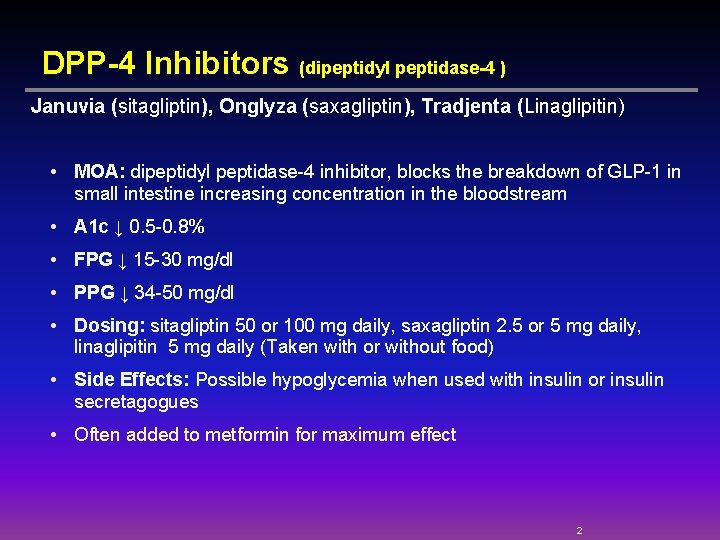
DPP-4 Inhibitors (dipeptidyl peptidase-4 ) Januvia (sitagliptin), Onglyza (saxagliptin), Tradjenta (Linaglipitin) • MOA: dipeptidyl peptidase-4 inhibitor, blocks the breakdown of GLP-1 in small intestine increasing concentration in the bloodstream • A 1 c ↓ 0. 5 -0. 8% • FPG ↓ 15 -30 mg/dl • PPG ↓ 34 -50 mg/dl • Dosing: sitagliptin 50 or 100 mg daily, saxagliptin 2. 5 or 5 mg daily, linaglipitin 5 mg daily (Taken with or without food) • Side Effects: Possible hypoglycemia when used with insulin or insulin secretagogues • Often added to metformin for maximum effect 2
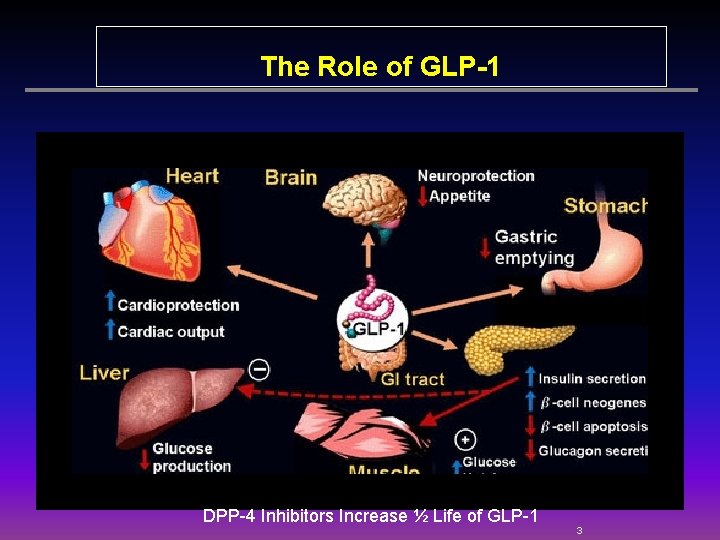
The Role of GLP-1 DPP-4 Inhibitors Increase ½ Life of GLP-1 3

DPP-IV ACTION • Cleaves GLP-1 • Results in decreased signal to the pancreas —limiting insulin response. X • That in turn decreases the signal to the liver resulting in increased hepatic glucose production. • HYPERGLYCEMIA 4

5
6

Major features of AACE 2009 Guidelines 1. Most Important Principle : recognition of the importance of avoiding hypoglycemia (24 -28). 2. It favors the use of GLP-1 agonists and DPP-4 inhibitors with higher priorityeffectiveness and overall safety profiles. 3. It moves sulfonylureas to a lower priority because of the associated risks a. hypoglycemia b. weight gain c. glycemic control only for relatively short period (1 to 2 years in typical patients). 7

Major features of AACE 2009 Guidelines 5. TZDs as “well-validated”, effective durable 6. It considers 3 other classes of agents (AGIs, colesevelam, and glinides) only for relatively narrow, well-defined clinical situations in view of their limited efficacy. 7. Rapid-acting insulin analogues are superior to “regular human insulin” - safer alternative. 8. NPH insulin is not recommended. - superseded by synthetic analogues insulin glargine and insulin detemir, which provide a relative peakless profile , yield better reproducibility and consistency, corresponding reduction in the risk of hypoglycemia. 8
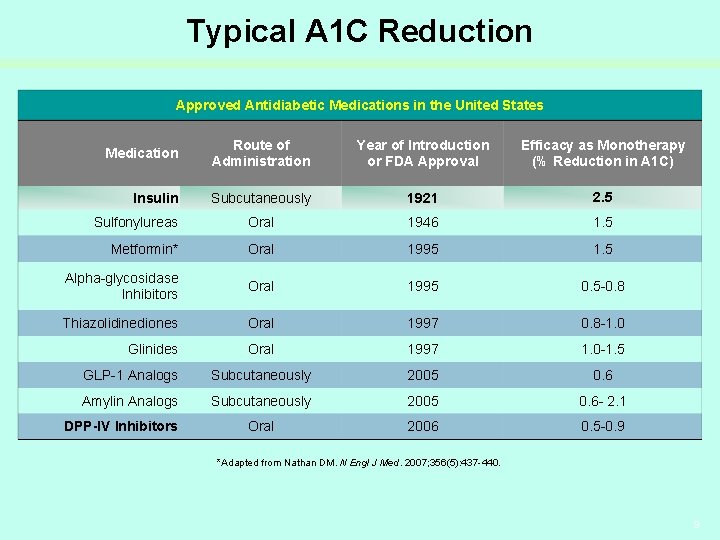
Typical A 1 C Reduction Approved Antidiabetic Medications in the United States Medication Route of Administration Year of Introduction or FDA Approval Efficacy as Monotherapy (% Reduction in A 1 C) Insulin Subcutaneously 1921 2. 5 Sulfonylureas Oral 1946 1. 5 Metformin* Oral 1995 1. 5 Alpha-glycosidase Inhibitors Oral 1995 0. 5 -0. 8 Thiazolidinediones Oral 1997 0. 8 -1. 0 Glinides Oral 1997 1. 0 -1. 5 GLP-1 Analogs Subcutaneously 2005 0. 6 Amylin Analogs Subcutaneously 2005 0. 6 - 2. 1 Oral 2006 0. 5 -0. 9 DPP-IV Inhibitors *Adapted from Nathan DM. N Engl J Med. 2007; 356(5): 437 -440. 9
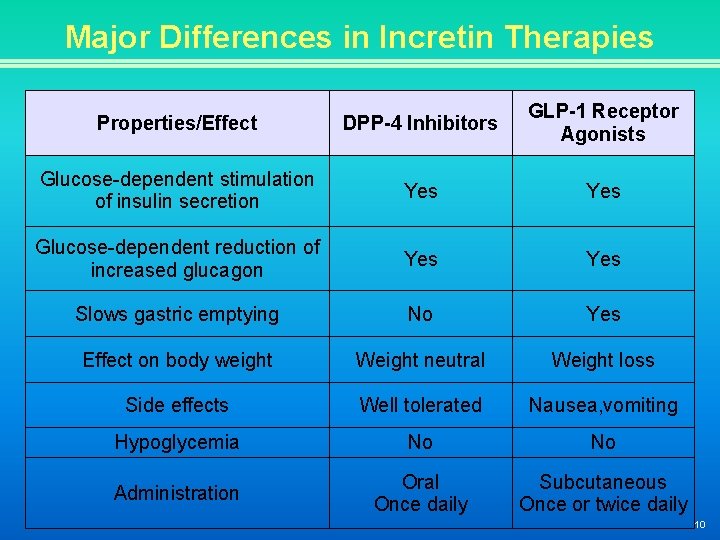
Major Differences in Incretin Therapies Properties/Effect DPP-4 Inhibitors GLP-1 Receptor Agonists Glucose-dependent stimulation of insulin secretion Yes Glucose-dependent reduction of increased glucagon Yes Slows gastric emptying No Yes Effect on body weight Weight neutral Weight loss Side effects Well tolerated Nausea, vomiting Hypoglycemia No No Administration Oral Once daily Subcutaneous Once or twice daily 10

Short-Version AACE Guidelines, 2009 11
- Slides: 11