The Role of ACEIs in Unstable Angina Sukhjinder

The Role of ACEIs in Unstable Angina Sukhjinder Sidhu Interior Health Pharmacy Resident Cardiology Rotation February 21, 2014

Learning Objectives • By the end of this 20 -min session the audience should be able to: – Describe the pathophysiology and clinical presentation of unstable angina (UA) – Compare and contrast the diagnostic criteria of UA, NSTEMI and STEMI – State the evidence for ACEI in UA – Be able to determine the need for ACEI in an UA patient
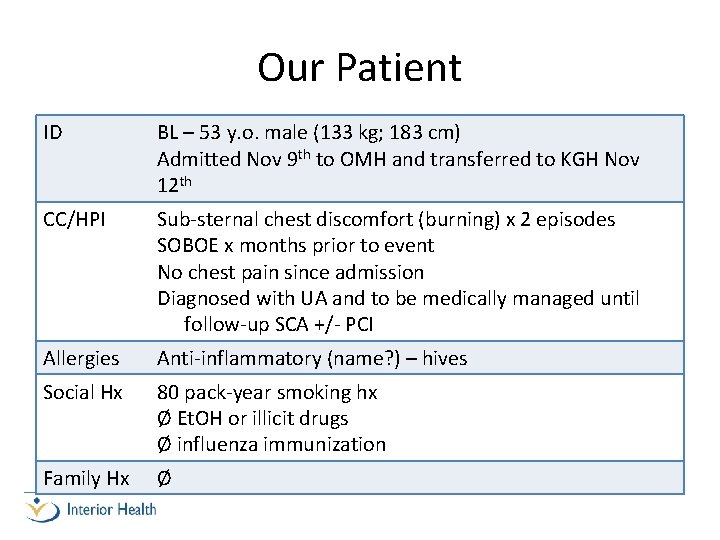
Our Patient ID BL – 53 y. o. male (133 kg; 183 cm) Admitted Nov 9 th to OMH and transferred to KGH Nov 12 th CC/HPI Sub-sternal chest discomfort (burning) x 2 episodes SOBOE x months prior to event No chest pain since admission Diagnosed with UA and to be medically managed until follow-up SCA +/- PCI Allergies Anti-inflammatory (name? ) – hives Social Hx 80 pack-year smoking hx Ø Et. OH or illicit drugs Ø influenza immunization Family Hx Ø
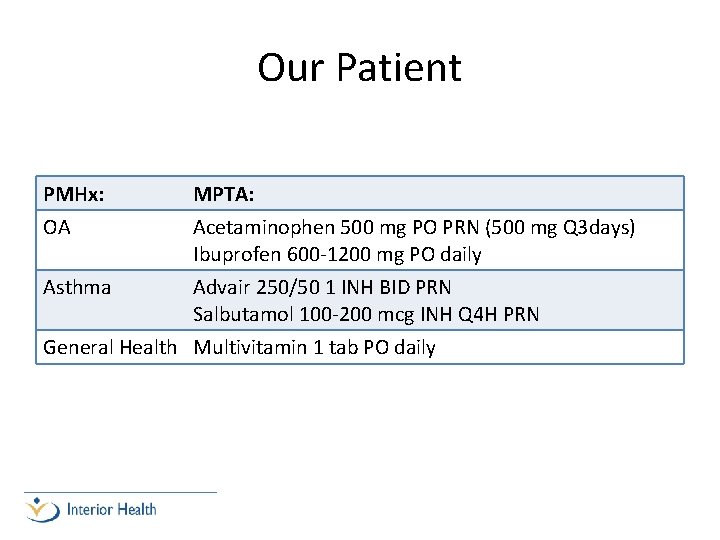
Our Patient PMHx: MPTA: OA Acetaminophen 500 mg PO PRN (500 mg Q 3 days) Ibuprofen 600 -1200 mg PO daily Asthma Advair 250/50 1 INH BID PRN Salbutamol 100 -200 mcg INH Q 4 H PRN General Health Multivitamin 1 tab PO daily
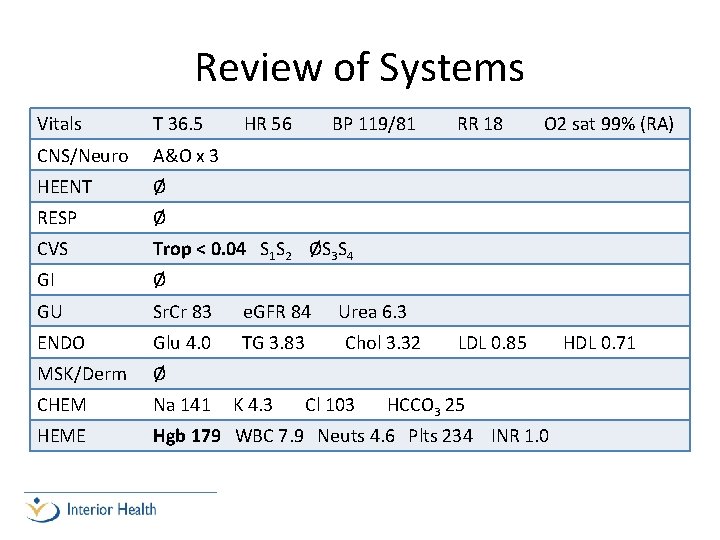
Review of Systems Vitals T 36. 5 HR 56 CNS/Neuro A&O x 3 HEENT Ø RESP Ø CVS Trop < 0. 04 S 1 S 2 ØS 3 S 4 GI Ø GU Sr. Cr 83 e. GFR 84 ENDO Glu 4. 0 TG 3. 83 MSK/Derm Ø CHEM Na 141 HEME Hgb 179 WBC 7. 9 Neuts 4. 6 Plts 234 INR 1. 0 K 4. 3 BP 119/81 RR 18 O 2 sat 99% (RA) Urea 6. 3 Chol 3. 32 Cl 103 LDL 0. 85 HCCO 3 25 HDL 0. 71
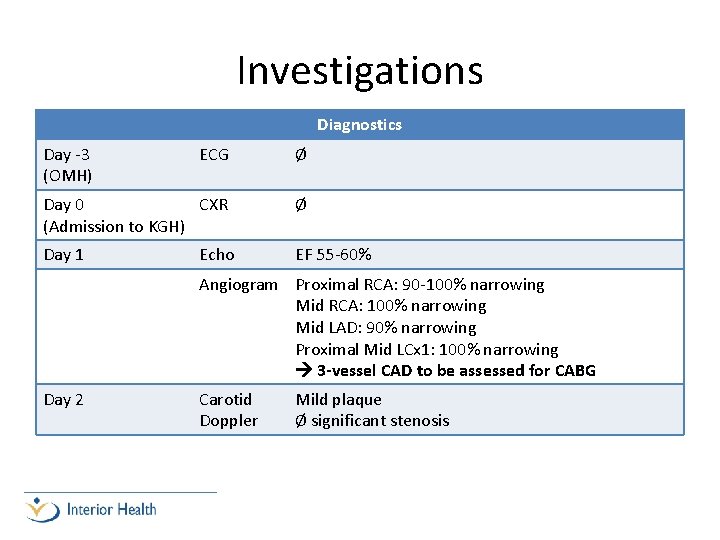
Investigations Diagnostics Day -3 (OMH) ECG Ø Day 0 CXR (Admission to KGH) Ø Day 1 EF 55 -60% Echo Angiogram Proximal RCA: 90 -100% narrowing Mid RCA: 100% narrowing Mid LAD: 90% narrowing Proximal Mid LCx 1: 100% narrowing 3 -vessel CAD to be assessed for CABG Day 2 Carotid Doppler Mild plaque Ø significant stenosis
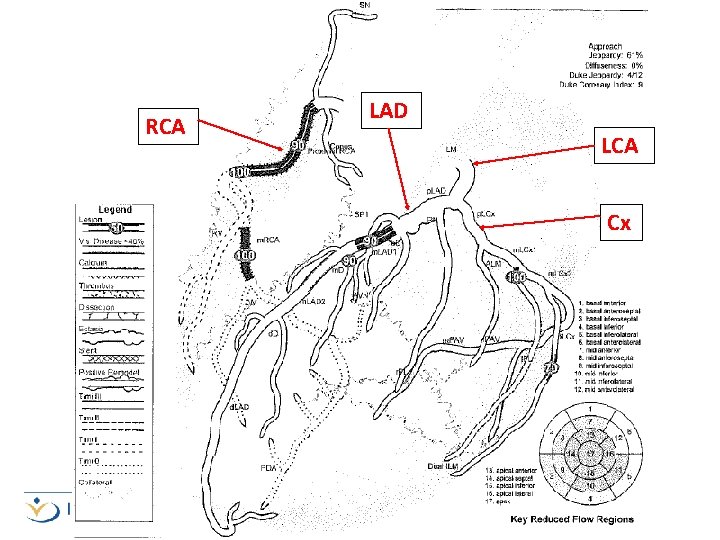
RCA LAD LCA Cx
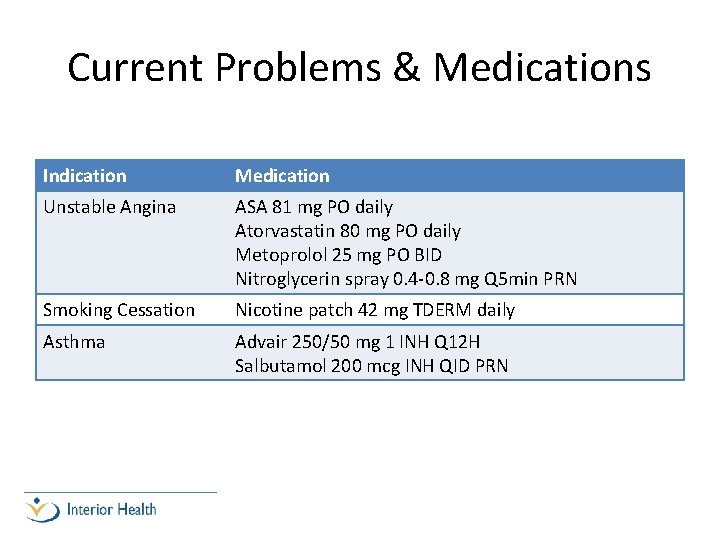
Current Problems & Medications Indication Medication Unstable Angina ASA 81 mg PO daily Atorvastatin 80 mg PO daily Metoprolol 25 mg PO BID Nitroglycerin spray 0. 4 -0. 8 mg Q 5 min PRN Smoking Cessation Nicotine patch 42 mg TDERM daily Asthma Advair 250/50 mg 1 INH Q 12 H Salbutamol 200 mcg INH QID PRN

DRP’s • BL is at risk of recurrent MI and death secondary to not receiving an ACEI and would benefit from optimization of ACS therapy. • BL is at risk of experiencing recurrent MI and death secondary to smoking and would benefit from smoking cessation. • BL is at risk of experiencing recurrent MI, worsening heart function and death secondary to ibuprofen use and would benefit from discontinuing ibuprofen and counseling on its adverse effects. • BL is at risk of experiencing influenza (fever, night sweats, myalgias, fatigue, nausea, vomiting, diarrhea) secondary to not receiving an influenza vaccine and would benefit from receiving the influenza vaccine. • BL is experiencing a mild rash secondary to the adhesives on the ECG strips and nicotine patch and would benefit from receiving a topical corticosteroid formulation.

Unstable Angina • Angina is caused by poor blood flow through the coronary vessels of the myocardium – Acute reduction in myocardial oxygen supply • CAD due to atherosclerosis is the most common cause of UA heartcurrents. com
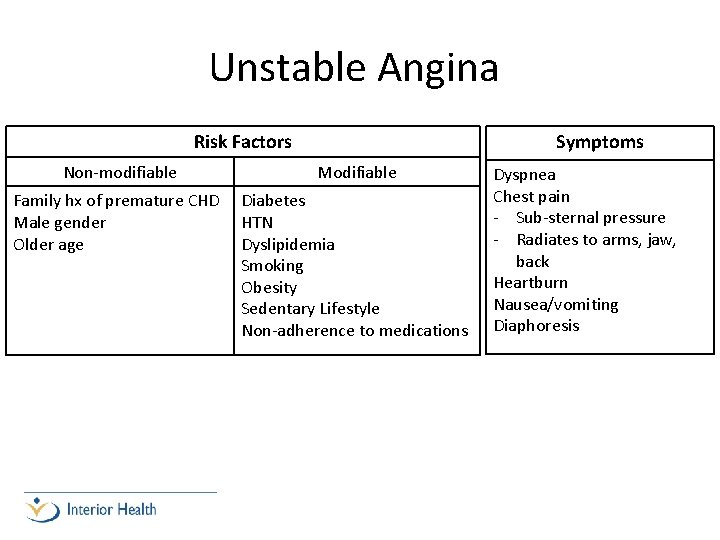
Unstable Angina Risk Factors Symptoms Non-modifiable Modifiable Family hx of premature CHD Male gender Older age Diabetes HTN Dyslipidemia Smoking Obesity Sedentary Lifestyle Non-adherence to medications Dyspnea Chest pain - Sub-sternal pressure - Radiates to arms, jaw, back Heartburn Nausea/vomiting Diaphoresis
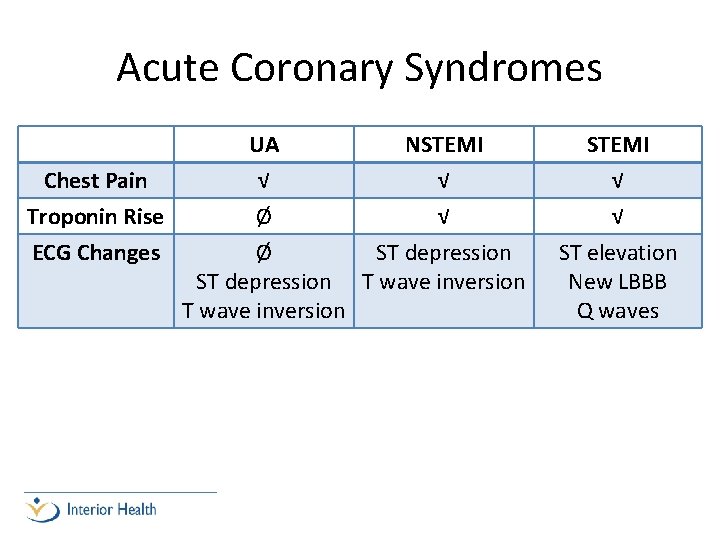
Acute Coronary Syndromes UA NSTEMI Chest Pain √ √ Troponin Rise Ø √ ECG Changes Ø ST depression T wave inversion STEMI √ √ ST elevation New LBBB Q waves

Goals of Therapy • Prevent mortality • Minimize myocardial damage and total ischemic time • Establish and maintain patency of the infarct-related artery • Alleviate signs and symptoms • Prevent re-occlusion, re-infarction, re-hospitalization • Minimize adverse events • Promote smoking cessation

Therapeutic Approach • • • ASA 81 mg PO daily P 2 Y 12 inhibitors High dose statin Beta-blockers RAAS inhibitors Nitroglycerin PRN

RAAS Inhibitors • Improve vascular endothelial function • Inhibit hypertrophy • Increase bradykinin – Increases nitric oxide production = vasodilation • Anti-atherosclerotic effects – Antagonize the rupture of plaques – Enhance fibrinolysis • Blood pressure control

Background • ACC/AHA Guidelines for UA/NSTEMI – ACEI should be given and continued indefinitely for patients recovering from UA/NSTEMI with HF, LV dysfunction, HTN, or DM, unless contraindicated (Class I A) – ACEIs have been shown to reduce mortality rates in patients with AMI and in patients with recent MI or with LV systolic dysfunction, in diabetic patients with LV dysfunction, and in a broad spectrum of patients with high-risk chronic CAD – ACEI are reasonable for patients recovering from UA/NSTEMI in the absence of LV dysfunction, HTN, or DM, unless contraindicated (Class IIa B) ACC/AHA 2007 Guidelines for UA/NSTEMI

Clinical Question P I C O UA patient awaiting CABG with no hx of type II DM or HTN and preserved LVEF ACEI + ACS therapy (ASA, atorvastatin, beta-blocker) Placebo + ACS therapy (ASA, atorvastatin, beta-blocker) Decrease mortality Prevent future MIs Ø Increased risk of adverse events

Literature Search Databases Google Scholar, Medline, Embase Search Terms Angiotensin-Converting Enzyme Inhibitors Cardiovascular diseases or heart diseases Limits English language and (guideline or meta analysis or RCT or systematic review) Results 2 relevant meta-analysis 6 relevant RCTs - HOPE - EUROPA - PEACE - IMAGINE 1 relevant observational study
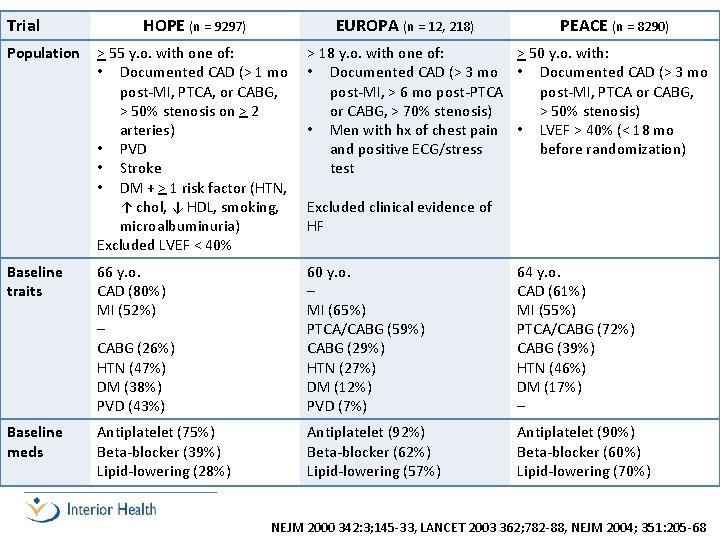
Trial Population HOPE (n = 9297) EUROPA (n = 12, 218) PEACE (n = 8290) > 55 y. o. with one of: • Documented CAD (> 1 mo post-MI, PTCA, or CABG, > 50% stenosis on > 2 arteries) • PVD • Stroke • DM + > 1 risk factor (HTN, ↑ chol, ↓ HDL, smoking, microalbuminuria) Excluded LVEF < 40% > 18 y. o. with one of: > 50 y. o. with: • Documented CAD (> 3 mo post-MI, > 6 mo post-PTCA post-MI, PTCA or CABG, > 70% stenosis) > 50% stenosis) • Men with hx of chest pain • LVEF > 40% (< 18 mo and positive ECG/stress before randomization) test Baseline traits 66 y. o. CAD (80%) MI (52%) – CABG (26%) HTN (47%) DM (38%) PVD (43%) 60 y. o. – MI (65%) PTCA/CABG (59%) CABG (29%) HTN (27%) DM (12%) PVD (7%) 64 y. o. CAD (61%) MI (55%) PTCA/CABG (72%) CABG (39%) HTN (46%) DM (17%) – Baseline meds Antiplatelet (75%) Beta-blocker (39%) Lipid-lowering (28%) Antiplatelet (92%) Beta-blocker (62%) Lipid-lowering (57%) Antiplatelet (90%) Beta-blocker (60%) Lipid-lowering (70%) Excluded clinical evidence of HF NEJM 2000 342: 3; 145 -33, LANCET 2003 362; 782 -88, NEJM 2004; 351: 205 -68
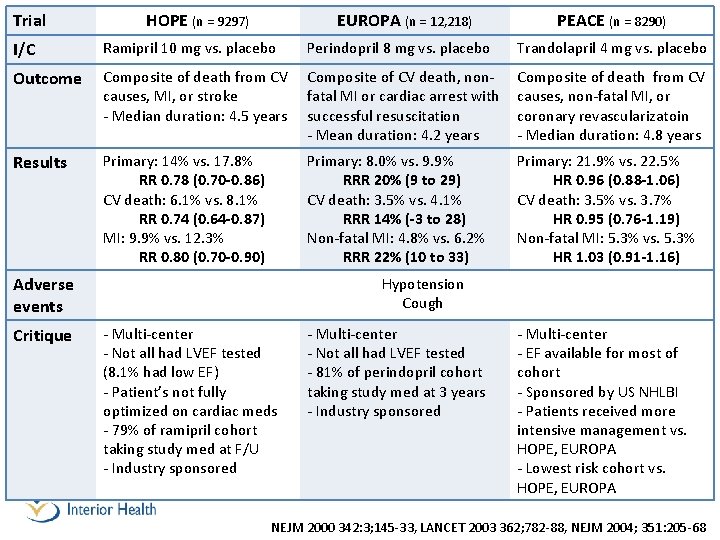
Trial HOPE (n = 9297) EUROPA (n = 12, 218) PEACE (n = 8290) I/C Ramipril 10 mg vs. placebo Perindopril 8 mg vs. placebo Trandolapril 4 mg vs. placebo Outcome Composite of death from CV causes, MI, or stroke - Median duration: 4. 5 years Composite of CV death, nonfatal MI or cardiac arrest with successful resuscitation - Mean duration: 4. 2 years Composite of death from CV causes, non-fatal MI, or coronary revascularizatoin - Median duration: 4. 8 years Results Primary: 14% vs. 17. 8% RR 0. 78 (0. 70 -0. 86) CV death: 6. 1% vs. 8. 1% RR 0. 74 (0. 64 -0. 87) MI: 9. 9% vs. 12. 3% RR 0. 80 (0. 70 -0. 90) Primary: 8. 0% vs. 9. 9% RRR 20% (9 to 29) CV death: 3. 5% vs. 4. 1% RRR 14% (-3 to 28) Non-fatal MI: 4. 8% vs. 6. 2% RRR 22% (10 to 33) Primary: 21. 9% vs. 22. 5% HR 0. 96 (0. 88 -1. 06) CV death: 3. 5% vs. 3. 7% HR 0. 95 (0. 76 -1. 19) Non-fatal MI: 5. 3% vs. 5. 3% HR 1. 03 (0. 91 -1. 16) Adverse events Critique Hypotension Cough - Multi-center - Not all had LVEF tested (8. 1% had low EF) - Patient’s not fully optimized on cardiac meds - 79% of ramipril cohort taking study med at F/U - Industry sponsored - Multi-center - Not all had LVEF tested - 81% of perindopril cohort taking study med at 3 years - Industry sponsored - Multi-center - EF available for most of cohort - Sponsored by US NHLBI - Patients received more intensive management vs. HOPE, EUROPA - Lowest risk cohort vs. HOPE, EUROPA NEJM 2000 342: 3; 145 -33, LANCET 2003 362; 782 -88, NEJM 2004; 351: 205 -68

IMAGINE D DB, PC, PG, MC RCT, N=2553 P Inclusion: > 18 y. o; post-CABG < 7 -10 days; stable after operation; LVEF > 40% Exclusion: Insulin-dependent DM or type 2 DM with microalbuminuria; significant valve stenosis/cardiomyopathy; K > 5. 6; Cr > 200 umol/L; BP > 160/90; significant perioperative MI Baseline: Mean age 61; men (87%); DM (10%); HTN (47%); LVEF 60%; BP 122/70 Meds – ASA (91%); statin (65%); BB (79%) I Quinapril 10 or 20 mg PO daily to target of 40 mg PO daily C Placebo O Composite of CV death, resuscitated cardiac arrest, nonfatal MI, coronary revascularization, UA requiring hospitalization, angina, stroke and CHF • median 2. 95 yrs Circulation. 2008; 117: 24 -31
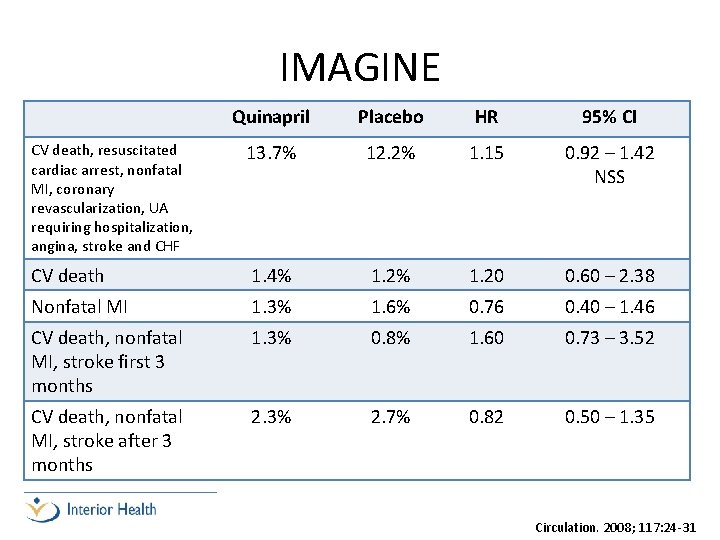
IMAGINE Quinapril Placebo HR 95% CI 13. 7% 12. 2% 1. 15 0. 92 – 1. 42 NSS CV death 1. 4% 1. 20 0. 60 – 2. 38 Nonfatal MI 1. 3% 1. 6% 0. 76 0. 40 – 1. 46 CV death, nonfatal MI, stroke first 3 months 1. 3% 0. 8% 1. 60 0. 73 – 3. 52 CV death, nonfatal MI, stroke after 3 months 2. 3% 2. 7% 0. 82 0. 50 – 1. 35 CV death, resuscitated cardiac arrest, nonfatal MI, coronary revascularization, UA requiring hospitalization, angina, stroke and CHF Circulation. 2008; 117: 24 -31

IMAGINE • Author’s conclusions: – At least in low risk-patients treated with contemporary therapy, early initiation of an ACEI after CABG has no benefit, and this strategy may even be associated with an increase in adverse events Circulation. 2008; 117: 24 -31

IMAGINE • Strengths – High compliance – Large number of patients receiving appropriate cardiac medications • Limitations – 11% of placebo cohort was taking open-label ACEI at 3 years – Industry sponsored • Generalizability – Patient had CABG surgery – Normal LVEF – Excluded majority of DM patients Circulation. 2008; 117: 24 -31
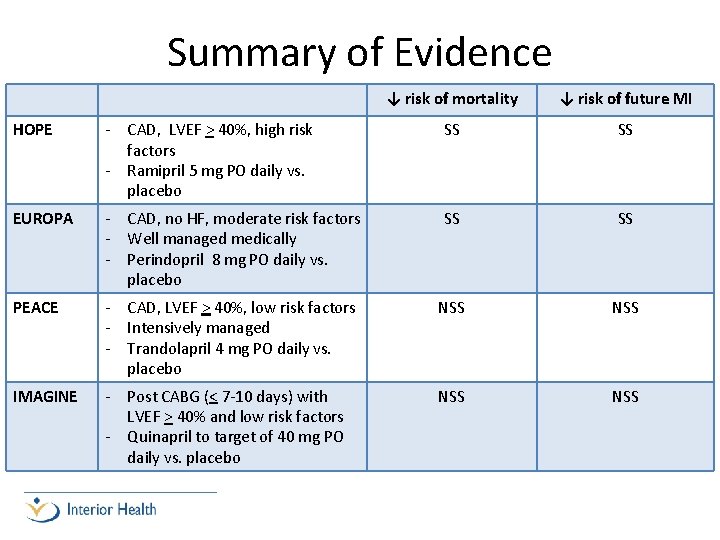
Summary of Evidence ↓ risk of mortality ↓ risk of future MI HOPE - CAD, LVEF > 40%, high risk factors - Ramipril 5 mg PO daily vs. placebo SS SS EUROPA - CAD, no HF, moderate risk factors - Well managed medically - Perindopril 8 mg PO daily vs. placebo SS SS PEACE - CAD, LVEF > 40%, low risk factors - Intensively managed - Trandolapril 4 mg PO daily vs. placebo NSS IMAGINE - Post CABG (< 7 -10 days) with LVEF > 40% and low risk factors - Quinapril to target of 40 mg PO daily vs. placebo NSS

Application • Necessary – No prior risk factors, such as DM, HTN or ↓ LVEF • Effective – In CABG patients with no clear indication for ACEI, they have conflicting evidence in reducing CV death, non-fatal MI, and revascularization • Safety – Risk of hypotension and cough – BP was consistently 95 -110/60 -70 • Adherence – Minimal non-adherence risk • Patient Factors – Increased pill and cost burden

Therapeutic Plan • Do not initiate an ACEI at present • Administer influenza vaccine 0. 5 m. L IM x 1 • Hydrocortisone cream 1% apply to affected areas BID PRN • Provided counseling regarding A/E of NSAID use • Provided counseling on the use of acetaminophen over NSAIDs for OA pain • Provided counseling and reinforcement regarding smoking cessation
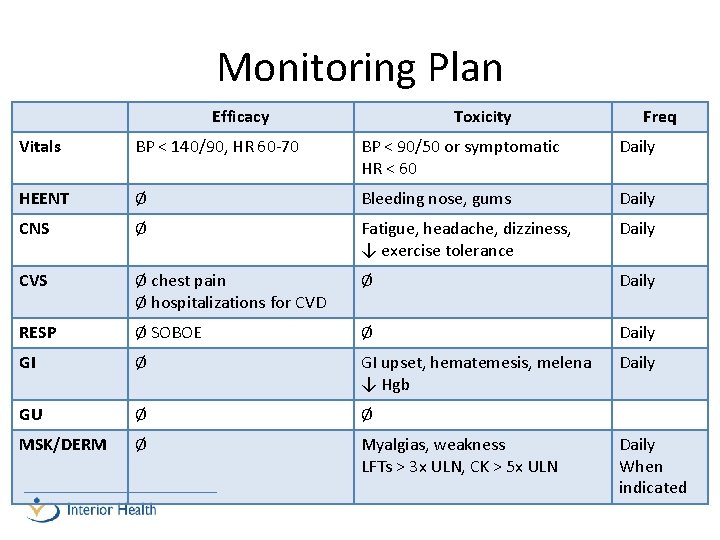
Monitoring Plan Efficacy Toxicity Freq Vitals BP < 140/90, HR 60 -70 BP < 90/50 or symptomatic HR < 60 Daily HEENT Ø Bleeding nose, gums Daily CNS Ø Fatigue, headache, dizziness, ↓ exercise tolerance Daily CVS Ø chest pain Ø hospitalizations for CVD Ø Daily RESP Ø SOBOE Ø Daily GI Ø GI upset, hematemesis, melena ↓ Hgb Daily GU Ø Ø MSK/DERM Ø Myalgias, weakness LFTs > 3 x ULN, CK > 5 x ULN Daily When indicated

Follow Up ACEI not initiated at this time Influenza vaccine administered BL receptive to smoking cessation BL receptive to avoiding use of NSAIDs and using acetaminophen for pain control • Successful CABG • •

Conclusion • No studies have evaluated ACEI solely in UA patients • ACEI decreased CV death and non-fatal MI in CAD patients if other risk factors are present – HTN, DM, ↑ chol, or ↓ LVEF • ACEI have not shown similarly consistent results in lower CV risk patients

Questions? IMAGE: http: //comicsthatsaysomething. quora. com/A-Day-at-the-Park? ref=fb

NSAIDs & CV Risk • COX-1 is active in platelets - Thromboxane A 2 is a vasoconstrictor and potent stimulator of platelet aggregation - TXA 2 increases renal salt & fluid retention, increases BP, enhances MI & vascular remodeling • COX-2 is active in cells that line blood vessels - Prostacyclin is a potent vasodilator and inhibitor of platelet function = vasculoprotective - PGI 2 facilitates renal salt & fluid excretion, lowers BP - inhibition decreases prostacyclin - Endothelial cells are a source of prostacyclin • NSAIDs tip the TXA 2/PGI 2 balance increasing CV risk - Nonselective NSAIDs with high COX 2 inhibition seem to have higher cardiovascular risk - Also increase risk of fluid retention and edema
- Slides: 32