The relationship between temperature and volume How Volume

The relationship between temperature and volume

How Volume Varies With Temperature If we place a balloon in liquid nitrogen it shrinks: So, gases shrink if cooled. Conversely, if we heat a gas it expands (as in a hot air balloon). Let’s take a closer look at temperature before we try to find the exact relationship of V vs. T.

Temperature scales Is 20 C twice as hot as 10 C? No. 68 F (20 C) is not double 50 F (10 C) Is 20 kg twice as heavy as 10 kg? Yes. 44 lb (20 kg) is double 22 lb (10 kg) What’s the difference? • Weights (kg or lb) have a minimum value of 0. • But the smallest temperature is not 0 C. • We saw that doubling P yields half the V. • Yet, to investigate the effect of doubling temperature, we first have to know what that means. • An experiment with a fixed volume of gas in a cylinder will reveal the relationship of V vs. T…
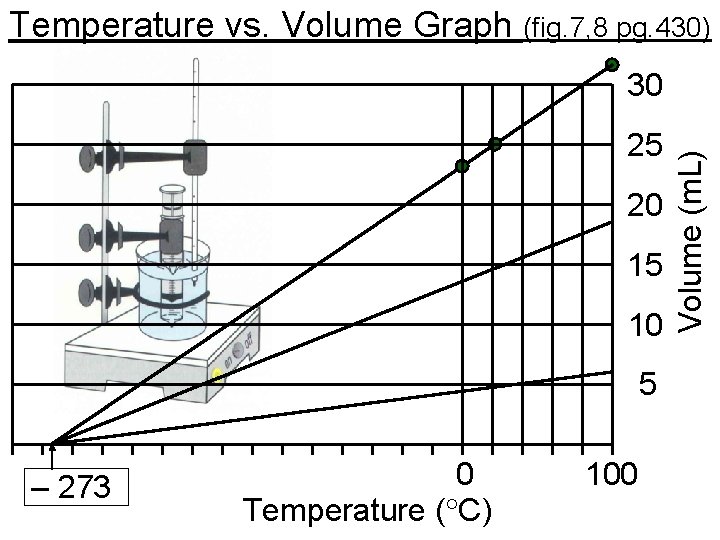
Temperature vs. Volume Graph (fig. 7, 8 pg. 430) 25 20 15 10 5 – 273 0 Temperature ( C) 100 Volume (m. L) 30

The Kelvin Temperature Scale • If a volume vs. temperature graph is plotted for gases, most lines can be interpolated so that when volume is 0 the temperature is -273 C. • Naturally, gases don’t really reach a 0 volume, but the spaces between molecules approach 0. • At this point all molecular movement stops. • – 273 C is known as “absolute zero” (no EK) • Lord Kelvin suggested that a reasonable temperature scale should start at a true zero value. • He kept the convenient units of C, but started at absolute zero. Thus, K = C + 273. 62 C = ? K: K= C+273 = 62 + 273 = 335 K • Notice that kelvin is represented as K not K.

Kelvin Practice What is the approximate temperature for absolute zero in degrees Celsius and kelvin? Absolute zero is – 273 C or 0 K Calculate the missing temperatures 100 C 373 = _______ K – 173 C 100 K = _______ – 30 C 243 = _______ K 300 K = _______ C 27 C 298 403 K = _______ 130 0 C 25 C 273 = _______ K – 273 0 K = _______ C

Charles’s Law • Looking back at the temperature vs. volume graph, notice that there is a direct relationship. • It can be shown that V/T = constant Read pages 432 -3. Answer these questions: 1. Give Charles’s law in words & as an equation. Charles’s Law: as the temperature of a gas increases, the volume increases proportionally, provided that the pressure and amount of gas remain constant, V 1/T 1 = V 2/T 2

2. A sample of gas occupies 3. 5 L at 300 K. What volume will it occupy at 200 K? V 1 = 3. 5 L, T 1 = 300 K, V 2 = ? , T 2 = 200 K Using Charles’ law: V 1/T 1 = V 2/T 2 3. 5 L / 300 K = V 2 / 200 K V 2 = (3. 5 L/300 K) x (200 K) = 2. 3 L 3. If a 1 L balloon is heated from 22°C to 100°C, what will its new volume be? V 1 = 1 L, T 1 = 22°C = 295 K V 2 = ? , T 2 = 100 °C = 373 K V 1/T 1 = V 2/T 2, 1 L / 295 K = V 2 / 373 K V 2 = (1 L/295 K) x (373 K) = 1. 26 L 4. Do questions 16, 17, 19 on page 434 For more lessons, visit www. chalkbored. com
- Slides: 8