The RAVEL Study A RAndomised double blind study

The RAVEL Study A RAndomised (double blind) study with the Sirolimus coated BX™ VElocity balloon expandable stent (CYPHER™) in the treatment of patients with De Novo native coronary artery Lesions M. C. Morice, P. W Serruys, J. E. Sousa, J. Fajadet, M. Perin, E. Ban Hayashi, A. Colombo, G. Schuler, P. Barragan, C. Bode on behalf of the RAVEL Study Group. www. Clinical trial results. org

RAVEL: Background • Sirolimus (Rapamune® - Wyeth Ayerst) is a naturally occurring macrocyclic antibiotic • It is a potent immunosuppressive agent inducing late G 1 cell cycle arrest www. Clinical trial results. org

Results of the RAVEL Trial Bx velocity™ stent coated with Sirolimus™ (Cypher™) versus placebo Inclusion Criteria • Single de novo lesions • • Native coronary arteries <18 mm in length Native coronary arteries 2. 5 to 3. 5 mm in diameter Design: • • • Double-blinded, placebo-controlled, randomized trial Enrolled 237 patients from several countries in Europe and Latin America All patients received Clopidogrel for a 2 -month period Results: • • • There was no case of restenosis in patients receiving the Sirolimus coated stent Rate was 26% in the uncoated stent group Late lumen loss of only 0. 01 mm with Sirolimus coated stent No reported cases of subacute thrombosis in patients treated with the Sirolimus coated stent Event-free survival was 97% with Sirolimus coated stent www. Clinical trial results. org

RAVEL: Study Design 237 patients with single de novo lesions in native coronary arteries < 18 mm in length and 2. 5 to 3. 5 mm in diameter, randomized trial in Europe and Latin America to Uncoated BX Velocity Stent (n=118) BX Velocity Stent Coated with Sirolimus (n=120) + + Clopidogrel for 2 months Major Adverse Cardiac Events (MACE) (Death / MI / Urgent TVR) Angiographic study www. Clinical trial results. org
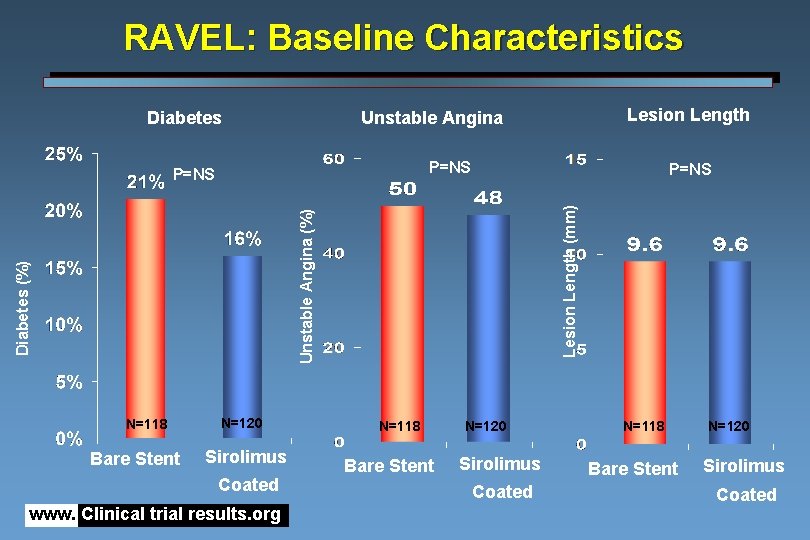
RAVEL: Baseline Characteristics Diabetes P=NS Diabetes (%) Unstable Angina (%) Lesion Length (mm) P=NS N=118 Bare Stent Lesion Length Unstable Angina N=120 Sirolimus Coated www. Clinical trial results. org N=118 Bare Stent N=120 Sirolimus Coated
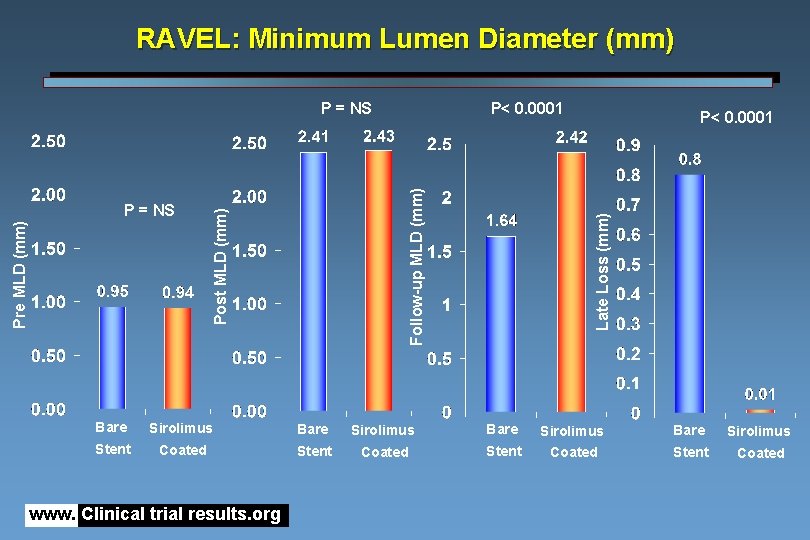
RAVEL: Minimum Lumen Diameter (mm) P< 0. 0001 Late Loss (mm) Post MLD (mm) Pre MLD (mm) P = NS P< 0. 0001 Follow-up MLD (mm) P = NS Bare Sirolimus Stent Coated www. Clinical trial results. org
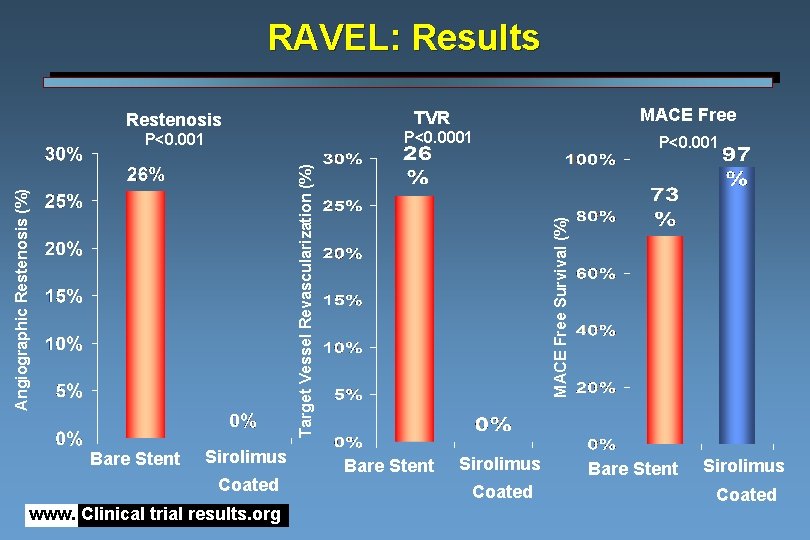
RAVEL: Results MACE Free TVR Restenosis P<0. 0001 Bare Stent Sirolimus Coated www. Clinical trial results. org P<0. 001 MACE Free Survival (%) Angiographic Restenosis (%) Target Vessel Revascularization (%) P<0. 001 Bare Stent Sirolimus Coated
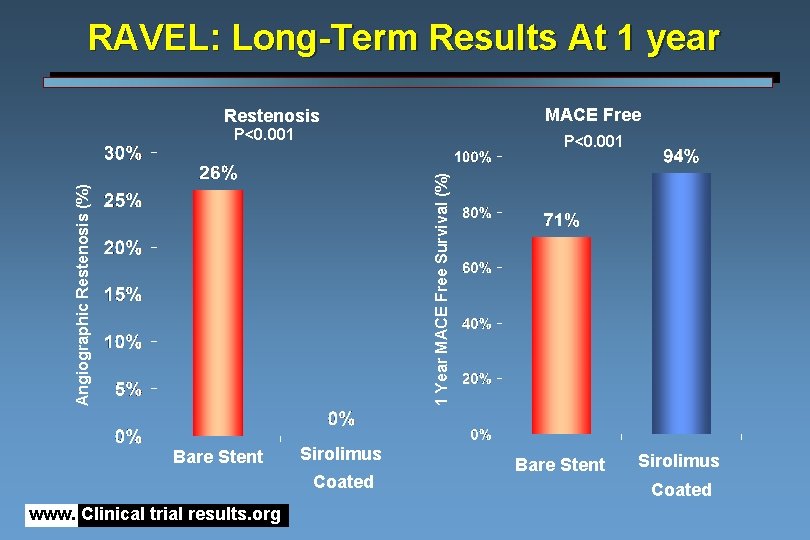
RAVEL: Long-Term Results At 1 year MACE Free Restenosis P<0. 001 Angiographic Restenosis (%) 1 Year MACE Free Survival (%) P<0. 001 Bare Stent Sirolimus Coated www. Clinical trial results. org Bare Stent Sirolimus Coated
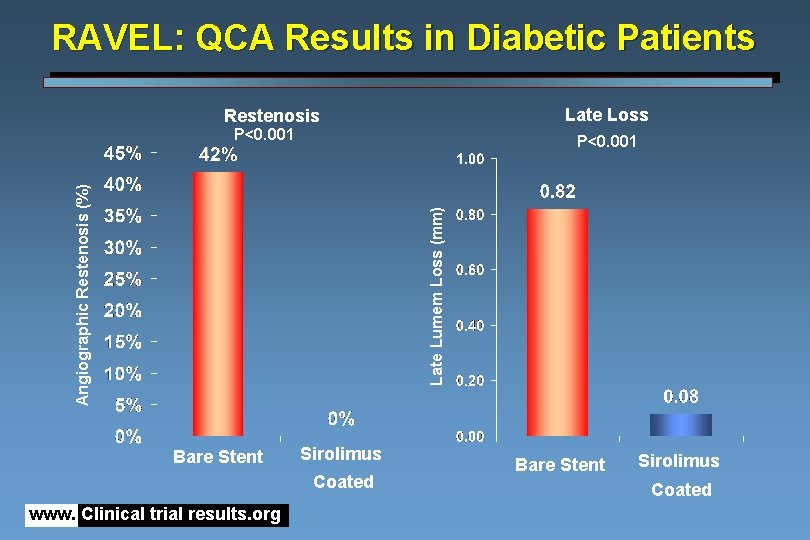
RAVEL: QCA Results in Diabetic Patients Late Loss Restenosis P<0. 001 Late Lumem Loss (mm) Angiographic Restenosis (%) P<0. 001 Bare Stent Sirolimus Coated www. Clinical trial results. org Bare Stent Sirolimus Coated

RAVEL: Cautionary Notes • In the First In Man study of 45 patients followed over 45 months, there was one stent thrombosis between months 12 and 13 • While data is available to just over 6 months, more long term data is needed to exclude a risk of late thrombosis • Fortunately, no aneuryms or pseudoaneurysms have been observed with 2 year follow-up in the First In Man study (FIM) • RAVEL may be underpowered to demonstrate late safety signals, the ongoing SIRIUS trial enrolling 1101 patients at 53 centers will provide more information in this regard • The efficacy of coated stenting in the management of in stent restenosis has not been studied to date www. Clinical trial results. org

RAVEL : Conclusions The treatment of a de novo lesion with CYPHER™ appears feasible and safe : no acute, subacute (30 days) or late occlusion occurred although clopidogrel / ticlopidine was administered for only 2 months Virtual elimination of neo-intimal in-stent proliferation: MLD post deployment (2. 43 mm) remains essentially unchanged at 6 months (2. 42 mm) with no measurable late loss (-0. 05 mm) Restenosis (0%) and no evidence of edge effect www. Clinical trial results. org

RAVEL: Conclusions Angiographic results are confirmed in a subset of patients evaluated with IVUS: Percentage of volume obstruction of the stent is less than 2%, No evidence of negative remodelling at the edges of the stent. This virtual elimination of neo-intimal hyperplasia was shown to be clinically translated into an absence of reintervention so that event free survival at 6 months is close to 97% and significantly (P<0. 0001) superior to the 72% event free survival of the patients treated with the bare stent. www. Clinical trial results. org
- Slides: 12