The RapoportLuebering Pathway Dr Sooad AlDaihan Biochemistry department

The Rapoport-Luebering Pathway Dr. Sooad Al-Daihan Biochemistry department

Overview ü In RBCs, glycolysis is modified by the Rapoport. Luebering shunt. ü It is a biochemical pathway in mature erythrocytes involving the formation of 2, 3 -bisphoglycerate and which regulates oxygen release from hemoglobin and delivery to tissues. Hence, the name “ 2, 3 bisphosphglycerate (2, 3 -BPG) shunt.
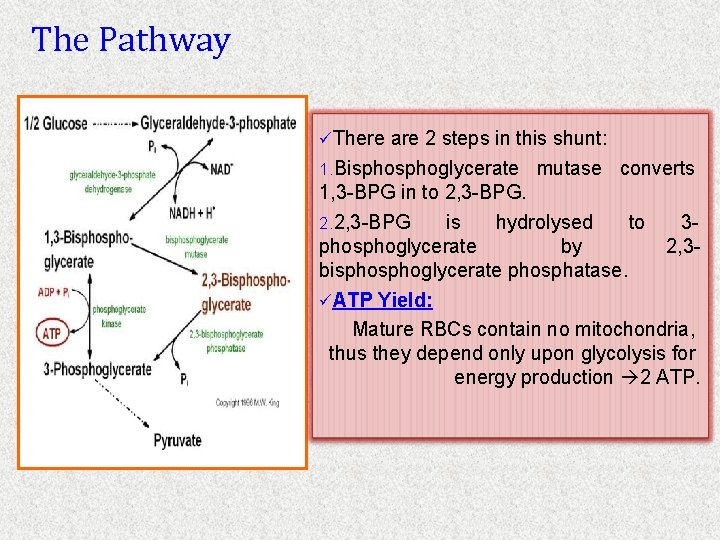
The Pathway üThere are 2 steps in this shunt: 1. Bisphoglycerate mutase converts 1, 3 -BPG in to 2, 3 -BPG. 2. 2, 3 -BPG is hydrolysed to 3 phosphoglycerate by 2, 3 bisphoglycerate phosphatase. üATP Yield: Mature RBCs contain no mitochondria, thus they depend only upon glycolysis for energy production 2 ATP.

Role of 2, 3 -BPG in haemoglobin function 2, 3 BPG acts as an allosteric regulator of hemoglobin ü that has the ability to decrease the affinity of O 2 to hemoglobin, . When a hemoglobin molecule is O 2 deficient, 2, 3 ü BPG inserts itself between the two beta chains, where it contains positively charged amino acids that form salt bridges with the negatively charged phosphate groups of 2, 3 -BPG. The lower affinity for O 2 means that the hemoglobin’s ü delivery of O 2 to the tissues is enhanced when needed.

Sorbitol Metabolism (polyol pathway)
Sorbitol, a polyol , is a bulk sweetener found in ü numerous food products. In addition to providing sweetness, it is an excellent ü humectant and texturizing agent.
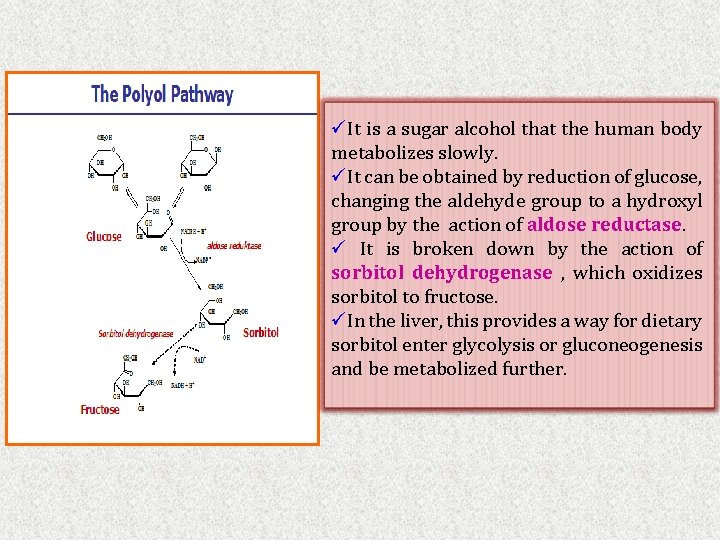
üIt is a sugar alcohol that the human body metabolizes slowly. üIt can be obtained by reduction of glucose, changing the aldehyde group to a hydroxyl group by the action of aldose reductase. ü It is broken down by the action of sorbitol dehydrogenase , which oxidizes sorbitol to fructose. üIn the liver, this provides a way for dietary sorbitol enter glycolysis or gluconeogenesis and be metabolized further.
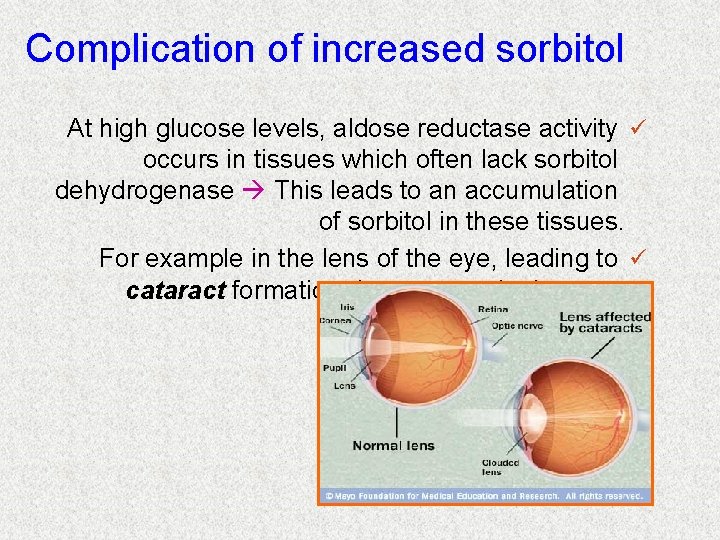
Complication of increased sorbitol At high glucose levels, aldose reductase activity ü occurs in tissues which often lack sorbitol dehydrogenase This leads to an accumulation of sorbitol in these tissues. For example in the lens of the eye, leading to ü cataract formation due to osmotic damage.

Ethanol Catabolism
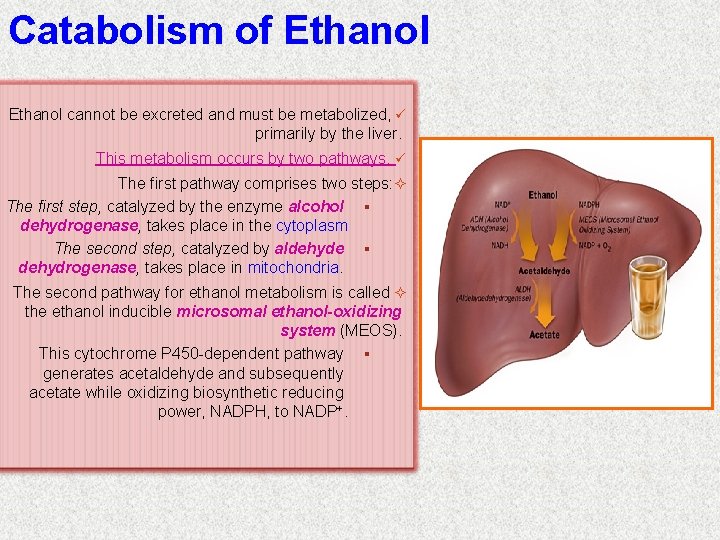
Catabolism of Ethanol cannot be excreted and must be metabolized, ü primarily by the liver. This metabolism occurs by two pathways. ü The first pathway comprises two steps: The first step, catalyzed by the enzyme alcohol § dehydrogenase, takes place in the cytoplasm The second step, catalyzed by aldehyde § dehydrogenase, takes place in mitochondria. The second pathway for ethanol metabolism is called the ethanol inducible microsomal ethanol-oxidizing system (MEOS). This cytochrome P 450 -dependent pathway § generates acetaldehyde and subsequently acetate while oxidizing biosynthetic reducing power, NADPH, to NADP+.

Metabolic effect of ethanol: The fate of acetate depends on the ratio of NADH to ü NAD+. Both alcohol dehydrogenase and aldehyde ü dehydrogenase consume NAD+, contributing to a high NADH: NAD+, resulting in: Inhibition of the citric acid cycle, because NADH ü inhibits two important regulatory enzymes: isocitrate dehydrogenase and α-ketoglutarate dehydrogenase Inhibition of gluconeogenesis by preventing the ü oxidation of lactate to pyruvate. In fact, the high concentration of NADH will cause the reverse reaction to predominate, and lactate will accumulate. The consequences may be hypoglycemia and lactic
- Slides: 11