The Quantum Mechanical Atom Chapter 8 Electron Distribution

The Quantum Mechanical Atom Chapter 8

Electron Distribution • When 2 or more atoms join to form a compound, the nuclei of the atoms stay relatively far apart. Only the atoms outer regions-the regions inhabited by electrons-come in close contact. • Valence electrons (VE)-are the outermost electrons of an atom. They are involved in bonding events. • The # of valence electrons (VE) for the representative elements groups 1 A-8 A can be predicted from the periodic table***. Group # = VE# ***Note: He is in group 8 A and has only 2 VE

Electron Configuration and Chemical Properties of Elements • Elements within the same group have the same outer electron configuration (same # of VE), and thus similar chemical properties. • The electronic structure of atoms has been elucidated based on studies regarding light which can be emitted or absorbed by an atom.

Electromagnetic Radiation • Light is a form of energy that travels through space or matter by wavelike oscillations. • Electromagnetic energy has an electric component and a magnetic component.
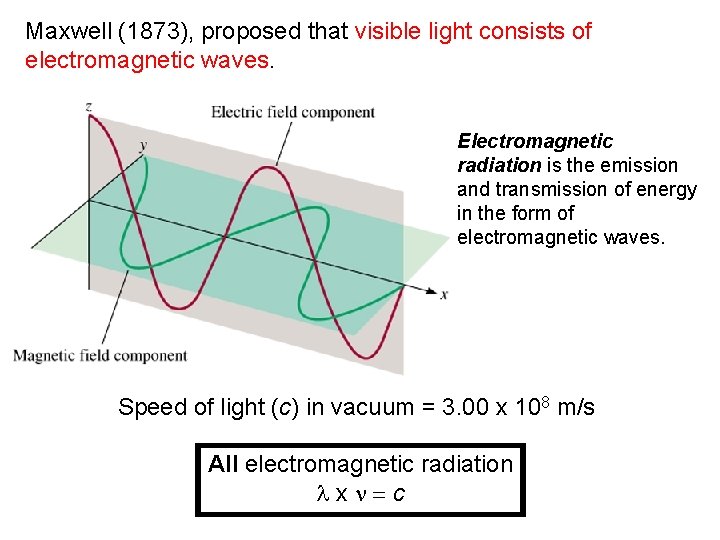
Maxwell (1873), proposed that visible light consists of electromagnetic waves. Electromagnetic radiation is the emission and transmission of energy in the form of electromagnetic waves. Speed of light (c) in vacuum = 3. 00 x 108 m/s All electromagnetic radiation lxn=c
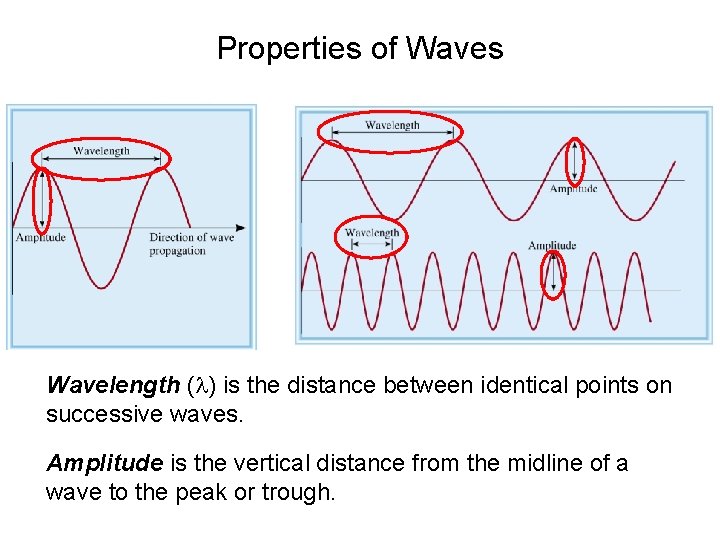
Properties of Waves Wavelength (l) is the distance between identical points on successive waves. Amplitude is the vertical distance from the midline of a wave to the peak or trough.
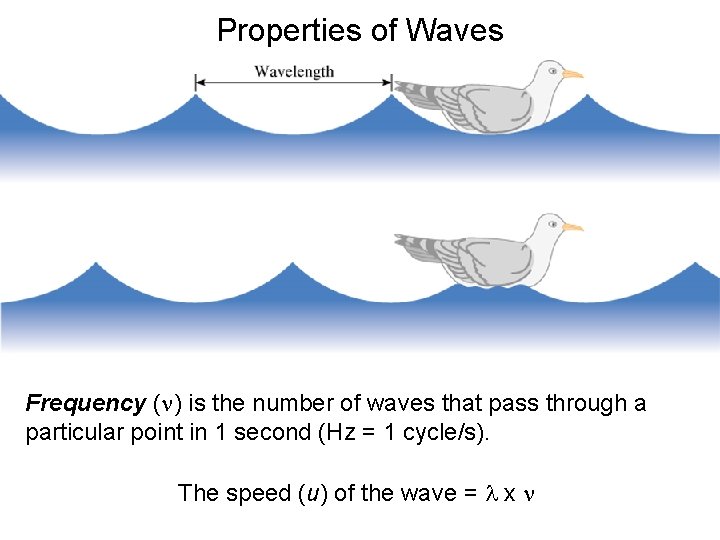
Properties of Waves Frequency (n) is the number of waves that pass through a particular point in 1 second (Hz = 1 cycle/s). The speed (u) of the wave = l x n

The Electromagnetic Spectrum • Electromagnetic radiation comes from a broad range of frequencies called the electromagnetic spectrum. • Only a small portion of the electromagnetic spectrum contains visible light with wavelengths 400 -700 nm.
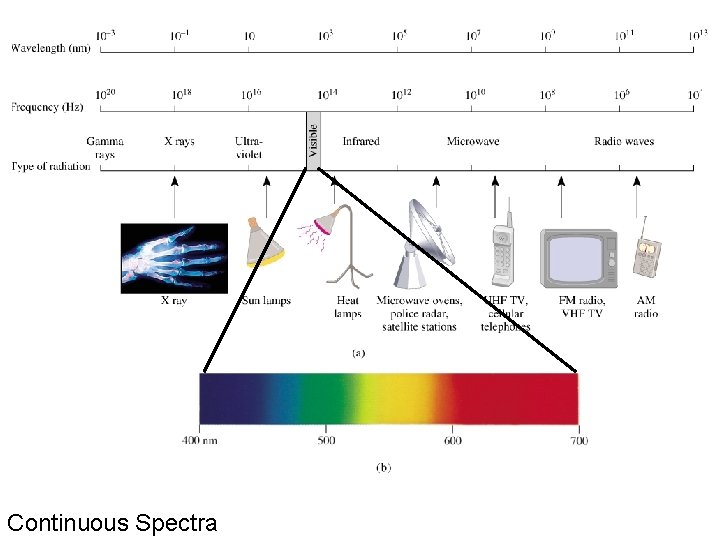
Continuous Spectra

A photon has a frequency of 6. 0 x 104 Hz. Convert this frequency into wavelength (nm). Does this frequency fall in the visible region? l lxn=c n l = c/n l = 3. 00 x 108 m/s / 6. 0 x 104 Hz l = 5. 0 x 103 m l = 5. 0 x 1012 nm Radio wave

Early 1900’s: Great Discoveries by Max Planck and Albert Einstein. Energy (light) is emitted or absorbed in discrete units (quantum). Light has both: 1. wave nature 2. particle nature E=hxn Planck’s constant (h) h = 6. 63 x 10 -34 J • s Photon is a “particle” of light

When copper is bombarded with high-energy electrons, X rays are emitted. Calculate the energy (in joules) associated with the photons if the wavelength of the X rays is 0. 154 nm. E=hxn E=hxc/l E = 6. 63 x 10 -34 (J • s) x 3. 00 x 10 8 (m/s) / 0. 154 x 10 -9 (m) E = 1. 29 x 10 -15 J

Atomic Spectra/Line Spectra • Continuous spectra contains light of all colors (see previous slide) which is much different than an atomic spectra also called line spectra. • Atomic spectra are observed when atoms are energized with a spark. • Every element has its own unique atomic spectra which acts as a fingerprint for that element.
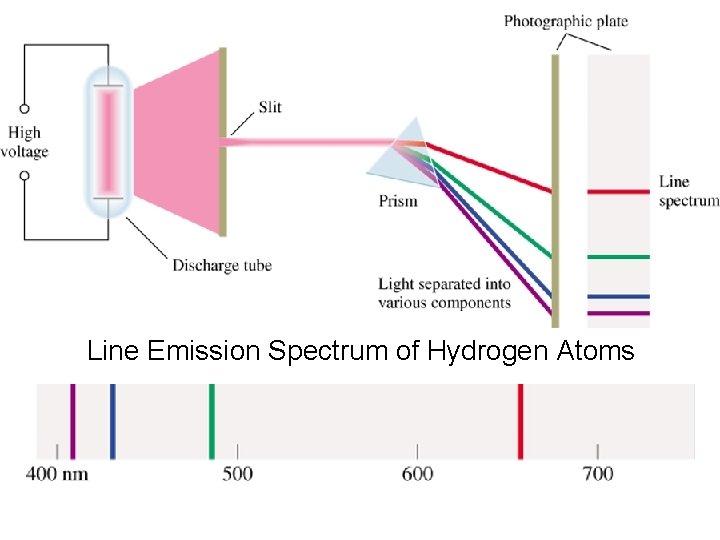
Line Emission Spectrum of Hydrogen Atoms

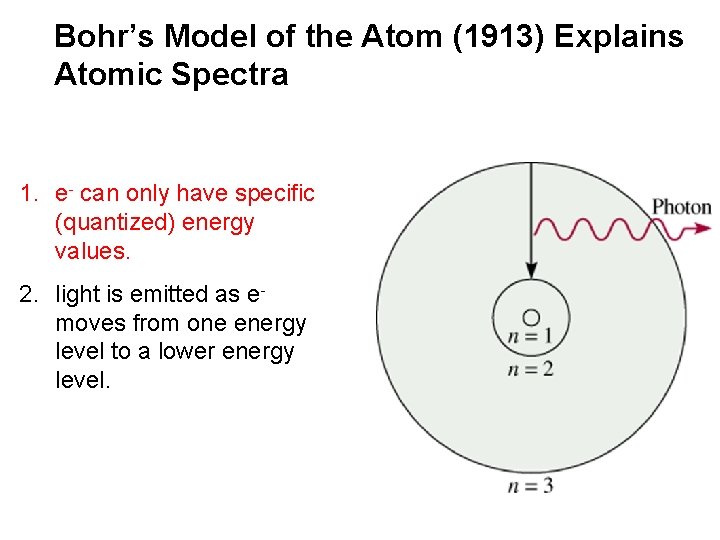
Bohr’s Model of the Atom (1913) Explains Atomic Spectra 1. e- can only have specific (quantized) energy values. 2. light is emitted as emoves from one energy level to a lower energy level.

Wave Properties of Matter and Wave Mechanics • The classical laws of physics do not apply to particles as tiny as electrons. • De Broglie (1924) reasoned that e- is both particle and wave. h l = mv v = velocity of em = mass of e-

Quantum Mechanics • How do we locate the exact location of a wave in space? How do we locate the exact location of a subatomic particle in an atom? • Werner Heisenberg and Erwin Schrodinger attempt to solve these questions. 18

Heisenberg Uncertainty Principle • It is impossible to know simultaneously both the momentum and the position of a particle with certainty in an atom. • Applying this principle we see that in reality electrons do not orbit the nucleus of an atom in a well defined path as Bohr suggested. 19

Schrodinger Wave Equation In 1926 Schrodinger wrote an equation that described both the particle and wave nature of the e. Wave function (y, psi) describes: 1. energy of e- with a given y 2. probability of finding e- in a volume of space Schrodinger’s equation can only be solved exactly for the hydrogen atom. Must approximate its solution for multi-electron systems. 20

Schrodinger Equation • The probability of finding an electron in 3 D space around the nucleus of an atom is proportional to the square of the wave function. • (y)^2 = ( n, l, ms) 21

Quantum Mechanics • Results in our understanding that electrons in atoms exist in certain energy levels around the nucleus called shells. Shells are numbered 1, 2, 3 etc with number 1 being the lowest in energy and closest to the nucleus. • The number of electrons in any particular shell is limited by the equation: Maximum # of electrons per shell= 2 n 2 n=1, 2, 3, 4…

Quantum Mechanics • Each shell consists of subshells. A subshell is a sub-energy level labeled s, p, d, f. • Subshells are composed of orbitals. The number of orbitals per subshell and the maximum number of electrons that can exist within them are described on page 328.

Electron Orbitals • Regions in space around a nucleus where there is a high probability of finding electrons. • Each region has a distinct shape. 24
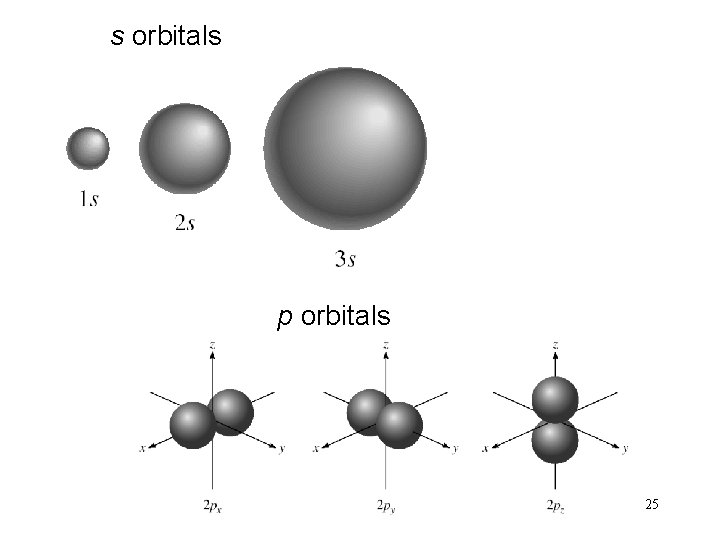
s orbitals p orbitals 25
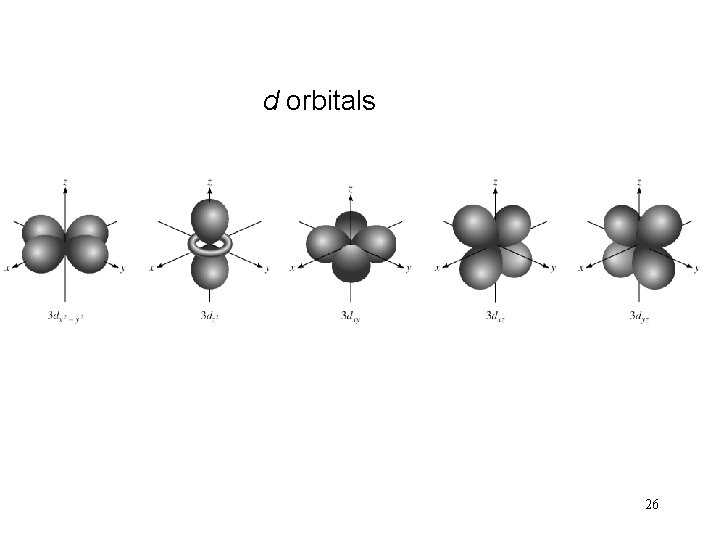
d orbitals 26

Schrodinger Wave Equation In 1926 Schrodinger wrote an equation that described both the particle and wave nature of the e. Wave function (y) describes: 1. energy of e- with a given y 2. probability of finding e- in a volume of space Schrodinger’s equation can only be solved exactly for the hydrogen atom. Must approximate its solution for multi-electron systems. 27

Schrodinger Wave Equation y is a function of four numbers called quantum numbers (n, l, ms) principal quantum number n n = 1, 2, 3, 4, …. distance of e- from the nucleus n=1 n=2 n=3 28

Schrodinger Wave Equation quantum numbers: (n, l, ms) angular momentum quantum number l for a given value of n, l = 0, 1, 2, 3, … n-1 n = 1, l = 0 n = 2, l = 0 or 1 n = 3, l = 0, 1, or 2 l=0 l=1 l=2 l=3 s orbital p orbital d orbital f orbital Shape of the “volume” of space that the e- occupies 29
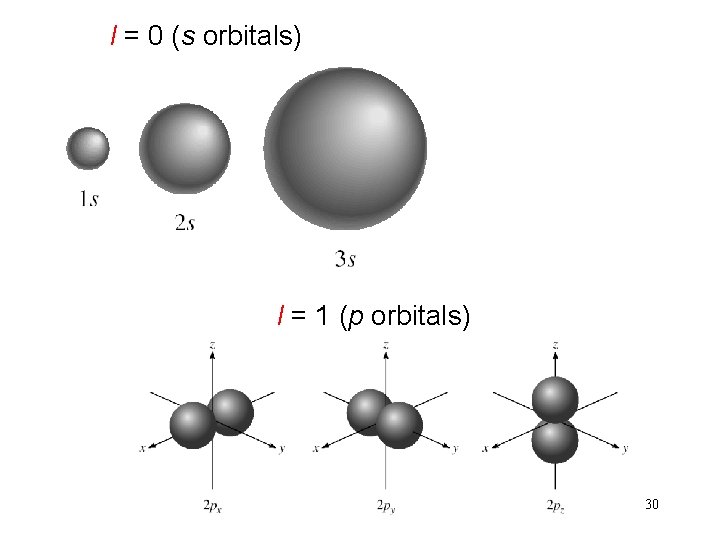
l = 0 (s orbitals) l = 1 (p orbitals) 30
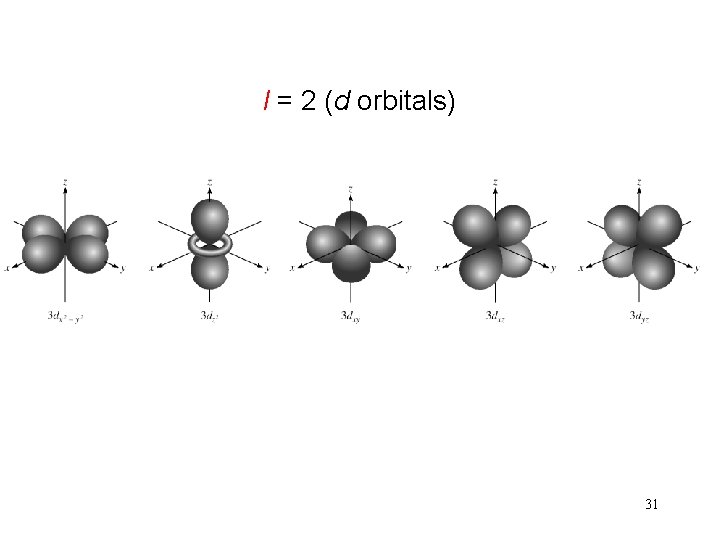
l = 2 (d orbitals) 31

Schrodinger Wave Equation quantum numbers: (n, l, ms) magnetic quantum number ml for a given value of l ml = -l, …. , 0, …. +l if l = 1 (p orbital), ml = -1, 0, or 1 if l = 2 (d orbital), ml = -2, -1, 0, 1, or 2 orientation of the orbital in space 32
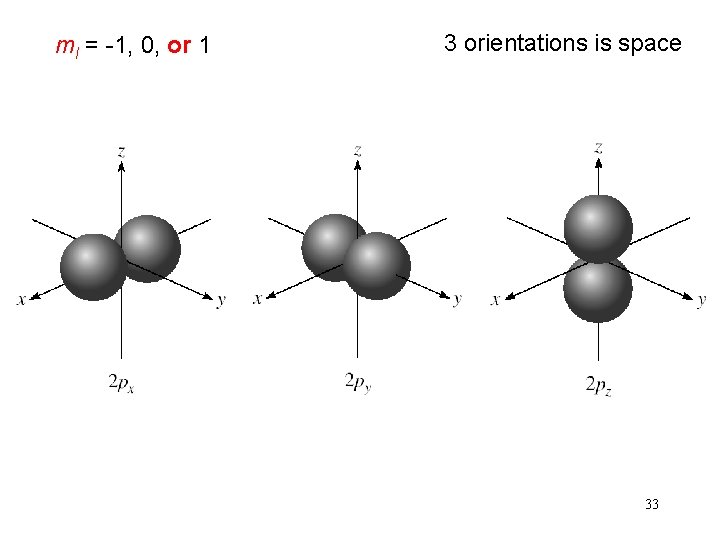
ml = -1, 0, or 1 3 orientations is space 33
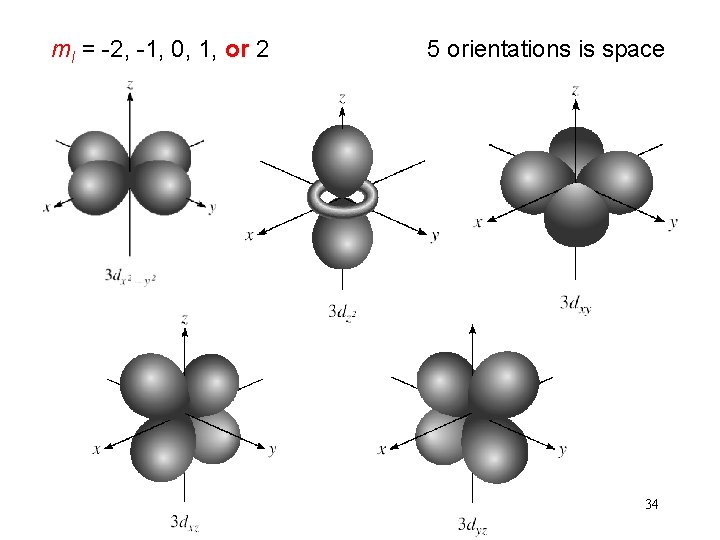
ml = -2, -1, 0, 1, or 2 5 orientations is space 34

Schrodinger Wave Equation (n, l, ms) spin quantum number ms ms = +½ or -½ ms = +½ ms = -½ 35

Schrodinger Wave Equation quantum numbers: (n, l, ms) Existence (and energy) of electron in atom is described by its unique wave function y. Pauli exclusion principle - no two electrons in an atom can have the same four quantum numbers. Each seat is uniquely identified (E, R 12, S 8) Each seat can hold only one individual at a time 36

Schrodinger Wave Equation quantum numbers: (n, l, ms) Shell – electrons with the same value of n Subshell – electrons with the same values of n and l Orbital – electrons with the same values of n, l, and ml How many electrons can an orbital hold? If n, l, and ml are fixed, then ms = ½ or - ½ y = (n, l, ml, ½) or y = (n, l, ml, -½) An orbital can hold 2 electrons 37
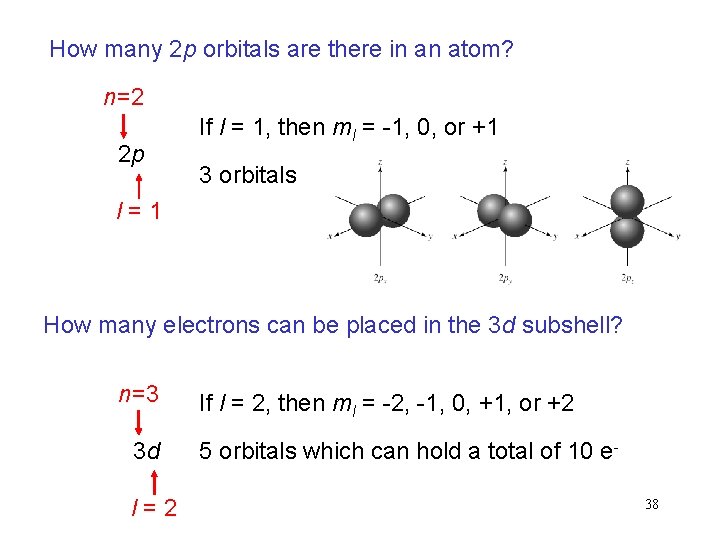
How many 2 p orbitals are there in an atom? n=2 2 p If l = 1, then ml = -1, 0, or +1 3 orbitals l=1 How many electrons can be placed in the 3 d subshell? n=3 3 d l=2 If l = 2, then ml = -2, -1, 0, +1, or +2 5 orbitals which can hold a total of 10 e 38
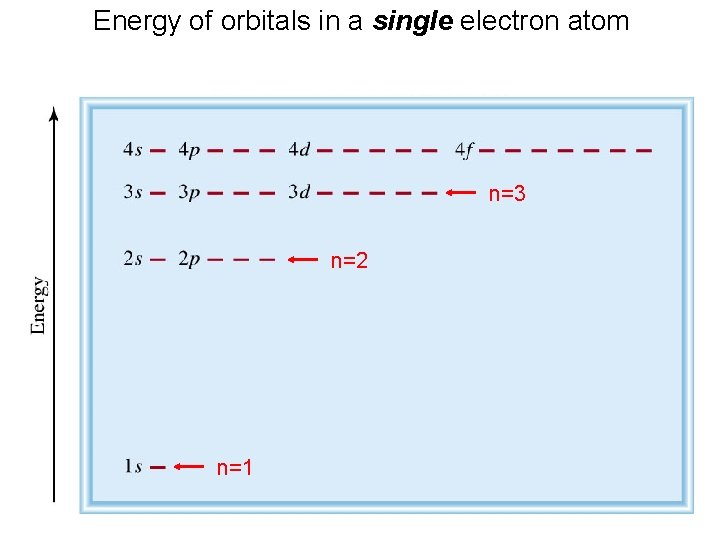
Energy of orbitals in a single electron atom n=3 n=2 n=1
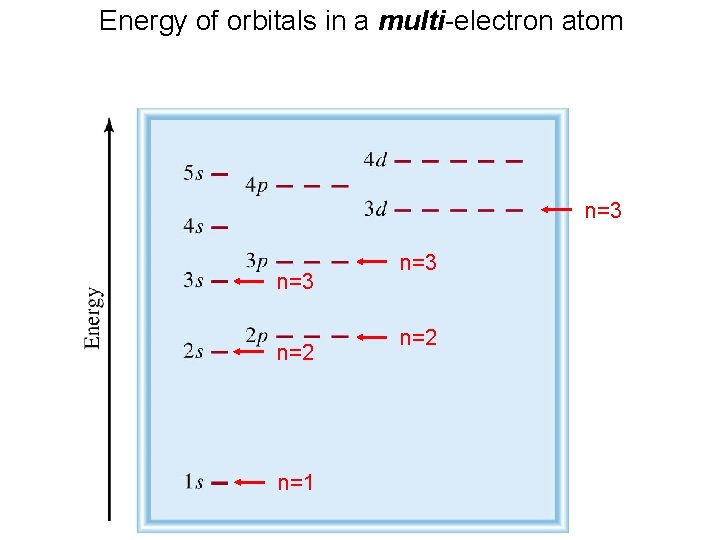
Energy of orbitals in a multi-electron atom n=3 n=2 n=1 n=3 n=2

Filling of Subshells and the Electron Energy Diagram • An atom is in its most stable state when its electrons are at the lowest possible energies. • Filling of an electron energy diagram must be done from the bottom (n=1) then upward. • When placing electrons in their respective orbitals one must take into consideration the Aufbau principle, the Pauli Exlusion Principle, and Hunds Rule.
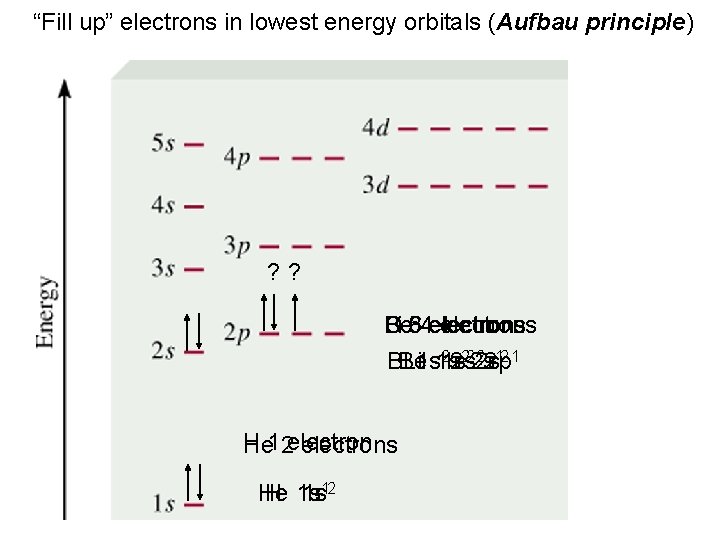
“Fill up” electrons in lowest energy orbitals (Aufbau principle) ? ? Be Li B 5 C 3 64 electrons 22 s 22 p 12 1 BBe Li 1 s 1 s 1 s 2 s H He 12 electrons He H 1 s 1 s 12
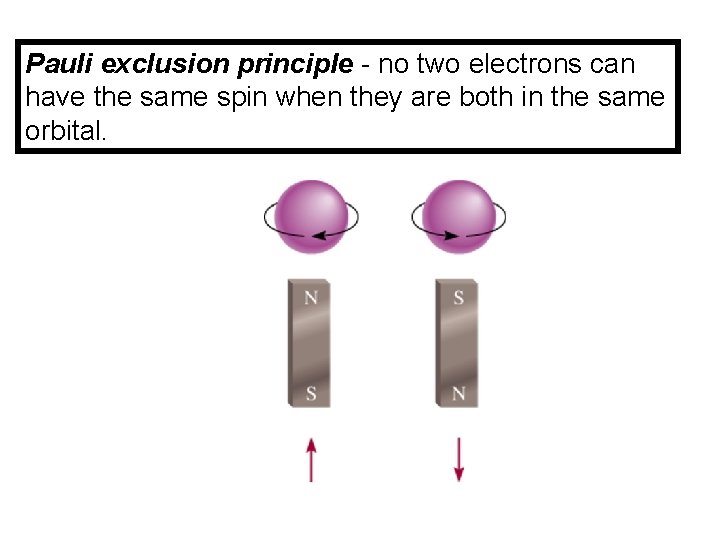
Pauli exclusion principle - no two electrons can have the same spin when they are both in the same orbital.
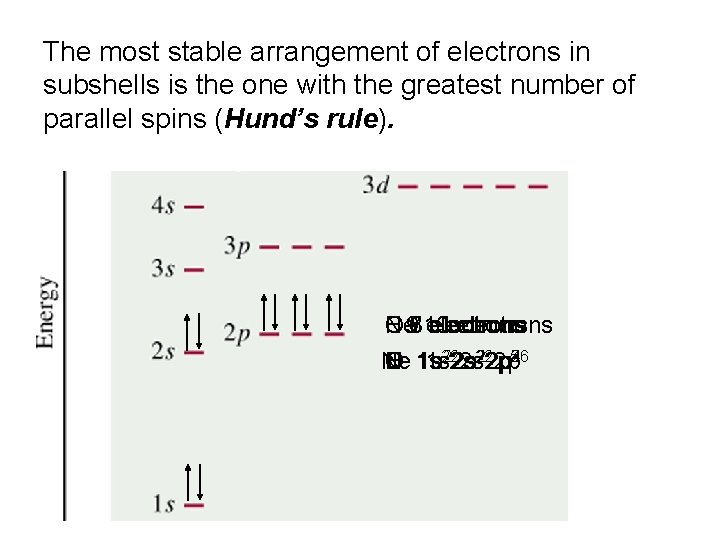
The most stable arrangement of electrons in subshells is the one with the greatest number of parallel spins (Hund’s rule). Ne 97 C N O F 6 810 electrons 222 p 22 p 5 246 3 Ne C N O F 1 s 1 s 222 s
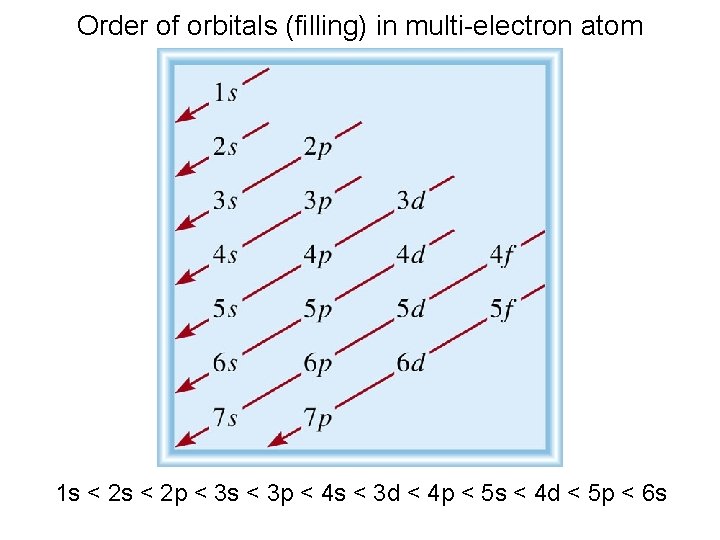
Order of orbitals (filling) in multi-electron atom 1 s < 2 p < 3 s < 3 p < 4 s < 3 d < 4 p < 5 s < 4 d < 5 p < 6 s
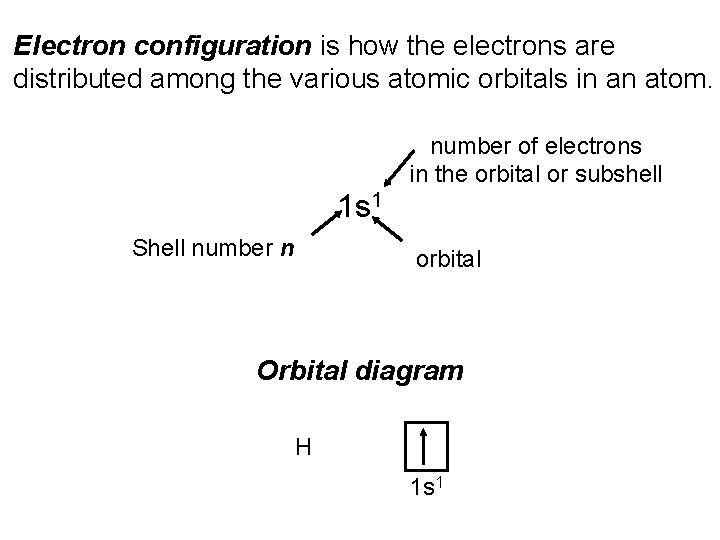
Electron configuration is how the electrons are distributed among the various atomic orbitals in an atom. number of electrons in the orbital or subshell 1 s 1 Shell number n orbital Orbital diagram H 1 s 1
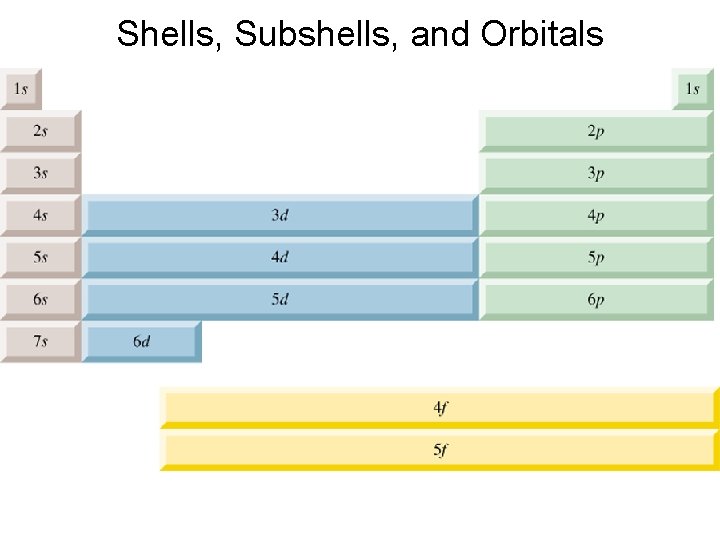
Shells, Subshells, and Orbitals

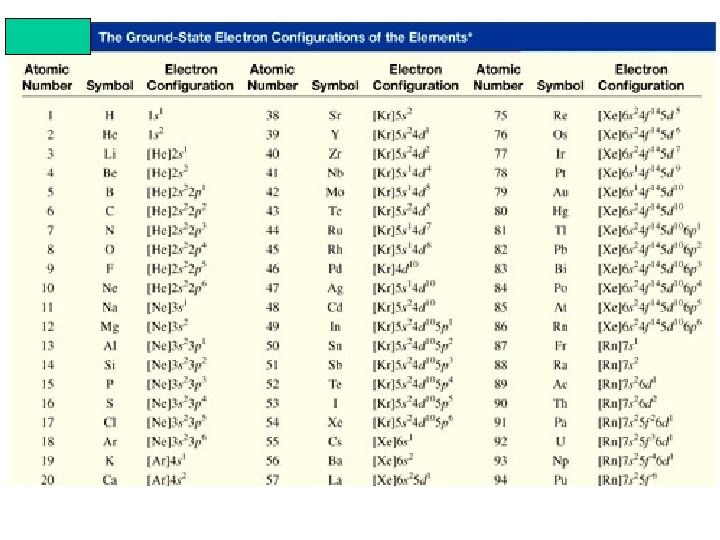
What is the electron configuration of Mg? Mg 12 electrons 1 s < 2 p < 3 s < 3 p < 4 s 1 s 22 p 63 s 2 2 + 6 + 2 = 12 electrons Abbreviated as [Ne]3 s 2 [Ne] 1 s 22 p 6 What is electron configuration of Cl? Cl 17 electrons 1 s 22 p 63 s 23 p 5 1 s < 2 p < 3 s < 3 p < 4 s 2 + 6 + 2 + 5 = 17 electrons Last electron added to 3 p orbital
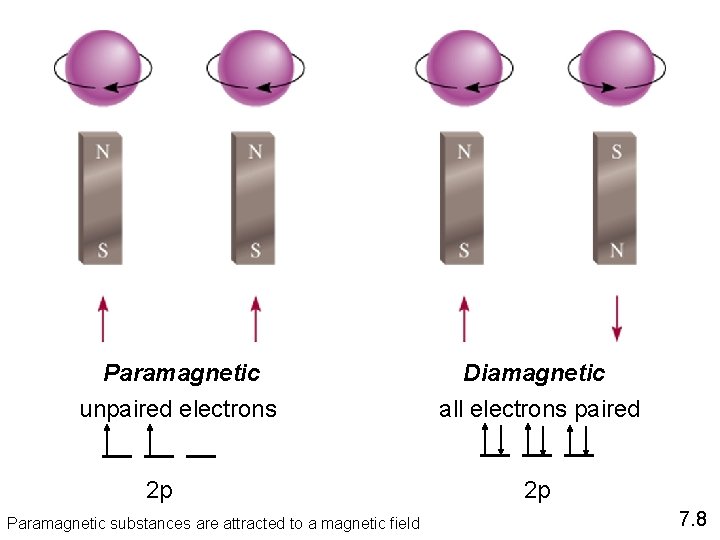
Paramagnetic unpaired electrons 2 p Paramagnetic substances are attracted to a magnetic field Diamagnetic all electrons paired 2 p 7. 8

THE END
- Slides: 51