The Purdue University Institutional Review Board An Overview

The Purdue University Institutional Review Board: An Overview and Update March 1 -2, 2012 Elizabeth Kipp Campbell, Ph. D. , CIP

Topics of Discussion • Why do we care about human research protection? • What is the IRB anyway? • Background on the Purdue IRB • Reorganization of the Purdue HRPP Staff • Metrics of IRB activity • Faculty Advisory Committee

Topics of Discussion Electronic submission and review Accreditation process Education for researchers Advanced Notice of Proposed Rulemaking: Changes to the federal regulations? • How the IRB can help you • Questions? • •

Why is Human Subjects Protection Important? • Promote the safety and well-being of human participants • Adhere to the ethical values and principles underlying research • Implement ethical and scientifically valid research • Allay concerns by the general public about the responsible conduct of research

What is the IRB? • The Institutional Review Board is a diverse group of individuals appointed by the institution responsible for overseeing the welfare of human subjects in research by applying ethical principles, the federal regulations, state statutes, and institutional policies – INDEPENDENT and Free from Undue Influence – Broad Composition • Must have at least one layperson and one non-affiliated member

The Purdue HRPP Staff • There are currently 6 staff members • These include: Director, IRB Administrator, Senior Protocol Analyst, 2 Protocol Analysts, and the IRB Administrative Assistant. • All receive initial and ongoing education and training in the protection of human subjects. • Several positions require certification as an IRB Professional (CIP)

The Purdue IRB Committees • There are 3 full boards of the IRB. – Social Science – Biomedical – Community Health • There approximately 15 members on each. • There is a Chair and Assistant Chair for each Committee. • Additionally, Chairs and Assistant Chairs serve as reviewers of Exempt and Expedited IRB applications. • All members receive initial and continuing education and training in the protection of human subjects.

Reorganization of the Purdue HRPP Staff • Due to increased volume, desire for better customer service and turnaround, and to attract high quality staff and remain competitive with peers, a new staffing configuration was proposed and approved by the Vice President for Research in the summer of 2011.
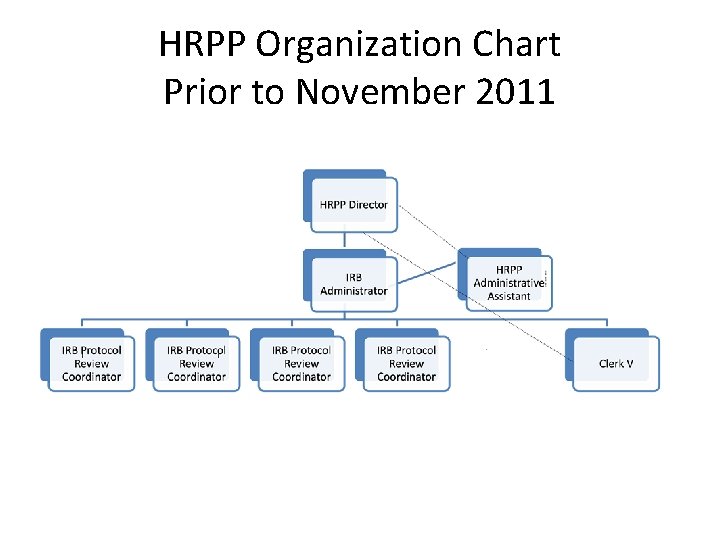
HRPP Organization Chart Prior to November 2011
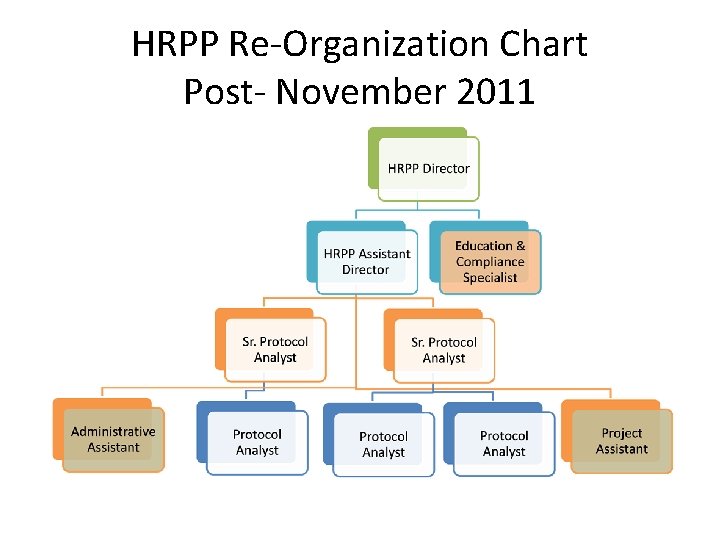
HRPP Re-Organization Chart Post- November 2011

Purdue IRB Metrics • Increased Volume of new studies over the past 6 years • Amendments and Continuing reviews remain relatively stable • The largest portion of reviews is Exempt Applications with very few studies needing Full Review
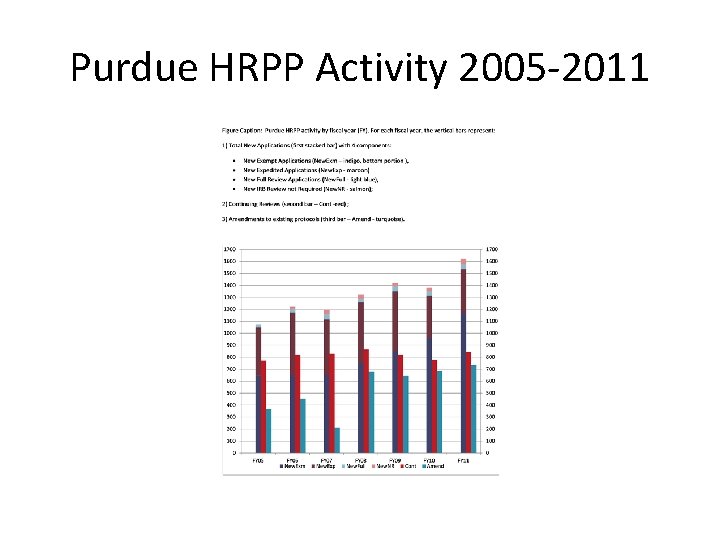
Purdue HRPP Activity 2005 -2011

Faculty Advisory Committee • Developed in the Fall of 2011 • IRB staff idea in order to increase input from and satisfaction of researchers • Also important to get researcher buy-in prior to accreditation visit • Composed of one member from each college and Purdue campus appointed by that college/campus. • Provides new ideas for better service, collegiality, and problem resolution.

Electronic Submission and Review • Coeus. Lite: Data management system used in SPS pre-awards and IACUC – IACUC recently rolled out to faculty – Currently struggling with system response time and support issues and working with responsible parties to resolve those issues – Pilot groups have been identified for IRB • IPFW has begun this process through the research office

Electronic Submission and Review • Will be a totally paperless process once fully implemented • Exempt and Expedited review will begin fully electronically as soon as rolled out • Full committee review will be phased in more slowly as resources for this implementation are sought and training for members completed

Accreditation Process • The University has been working toward accreditation by AAHRPP for many years. • Only recently have there been sufficient staff and resources to actually begin the process. • Purdue is the only Big 10 Institution not accredited • Pre-work is currently being done to ensure consistency and cohesiveness of the HRPP components before the application is submitted.

What is AAHRPP? AAHRPP is the Association for the Accreditation of Human Research Protection Programs. Developed by human research protection professionals in response to calls for the government and others to further regulate IRBs.

The Benefits of Accreditation • High standards and protections. • An assurance of quality. • Improved efficiency, effectiveness. • A competitive edge. • Government recognition. • Public trust, confidence.

Accreditation Principles • There are nine principles for accreditation. • What AAHRPP expects from Organization – Protecting the rights and welfare of research participants must be 1 st priority – Protecting research participants is the responsibility of everyone in the Organization and is not limited to the IRB – Striving to exceed the federal requirements and continually seeking new safeguards for protecting research participants while advancing scientific progress must be integrated into an Organization’s mission

AAHRP Accreditation Standards • The Standards are organized by Domain. There are three Domains. Under each Standard are specific elements that must be met. – Domain I: Organization – Domain II: Institutional Review Board – Domain III: Researcher and research staff

Domain I: Organization • This Domain describes the structural characteristics of the entity that assumes responsibility for the HRPP and applies for accreditation. • The Organization applies its HRPP to all research regardless of funding source, type of research, or place of conduct of research. • There are eight specific standards in this domain.

Domain II: Institutional Review Board • Within an HRPP, responsibilities must be delegated for providing ethical review and oversight of research. • The HRPP must have mechanisms in place to ensure the independence of its ethics review and oversight functions from other units within the organization, particularly with respect to decision -making regarding the ethics of research involving human participants. • There are five standards in this domain.

Domain III: Researcher and Research Staff • Competent, informed, conscientious, compassionate and responsible Researchers and Research Staff provide the best possible protection for human research participants. • As part of its HRPP, an organization can improve its protection of research participants if it has arrangements ascertaining and enhancing the competence of Researcher and Research Staff. • There are two standards in this domain.

Education for Researchers • Nuts and Bolts education in now available online • CITI Human Subject Protection Training is also available online • Staff are available to make general IRB presentations or specific topic presentations • A new position of IRB Education Specialist will be added to the HRPP Staff • Staff are available for phone consulations

Advanced Notice of Proposed Rulemaking (ANPRM) • Advanced Notice of Potential Changes in the Federal Regulations for the Protection of Human Subjects was published in the Federal Register on July 26, 2011 • Comments were submitted in response by over 1000 academic and research institutions and professional organizations • Director of OHRP noted at a professional conference that many misunderstood the intent of the proposed rules • Unclear if an official notice of changes will be published in the future.

ANPRM Summary of Changes

ANPRM Summary of Changes

ANPRM Summary of Changes

ANPRM Summary of Changes

ANPRM Summary of Changes

ANPRM Summary of Changes

Common Problems in the IRB Application Process • Two main areas of problems – Missing components – Incomplete or inadequate documentation • Either of these WILL delay study review and approval • The most common reason for delay is inadequate consent

Common Application Problems by Area • • • Research Team Qualifications Minimizing Risks Recruitment Mismatch amongst documents Informed Consent Process

Research Team Qualifications • Lack of documentation of Human Subjects Protection Training • Unclear that members of the team have the skill to conduct the kind of study they want to do

Minimizing Risks • Application materials not clear on how risks to subjects will be minimized • Missing or inadequate data safety monitoring plan (if required)

Recruitment • Lack of clarity regarding from where and by whom subjects will be recruited • Legitimate access to subjects/data • Advertisements or recruitment materials (e. g. , letters, flyers, postcards, etc. ) are missing or inflate monetary reward

Incongruent Documents • All study documents MUST coincide. • The application, narrative, consent, assent, grant, etc. must all be consistent with one another.

Informed Consent Process • Description of what will be said to subjects includes closed-ended questions and is inadequate • Written consent forms are inadequate – Language is too complex/not understandable – Elements are missing – Forms contain exculpatory language – Study specific language is not included

Readability Tips Declarative sentences at 8 th grade level Second person “you” not “I” Use paragraph headings and illustrations Describe quantities in lay terms Ask a non-researcher to read a final draft Software readability programs help, but don’t assess complexity • Replace technical language/jargon with lay terms • • •

How Can the IRB Help You? • We want to help you do the best research you can while protecting human subjects. • Start Early! • IRB can provide advice and assistance to complete your application. – (765) 494 -5942 – irb@purdue. edu – www. irb. purdue. edu – Walk-In Hours or by appointment
- Slides: 40