The Properties of Water primarily due to polarity













- Slides: 17

The Properties of Water primarily due to polarity

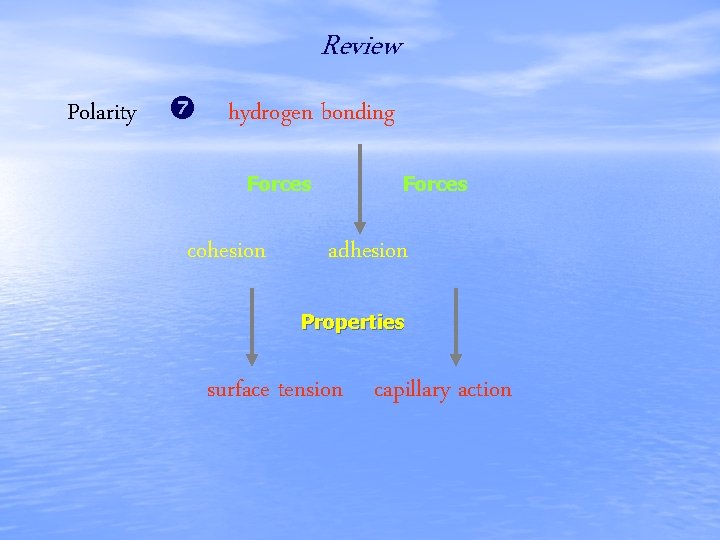
Terms to Know • • • polarity hydrogen bond cohesion adhesion surface tension capillary action

Water • Most abundant naturally occurring liquid • Liquid at most Earth temperatures • Unlike most liquids – it expands when frozen • The lower density of ice allows it to float (4°C most dense)

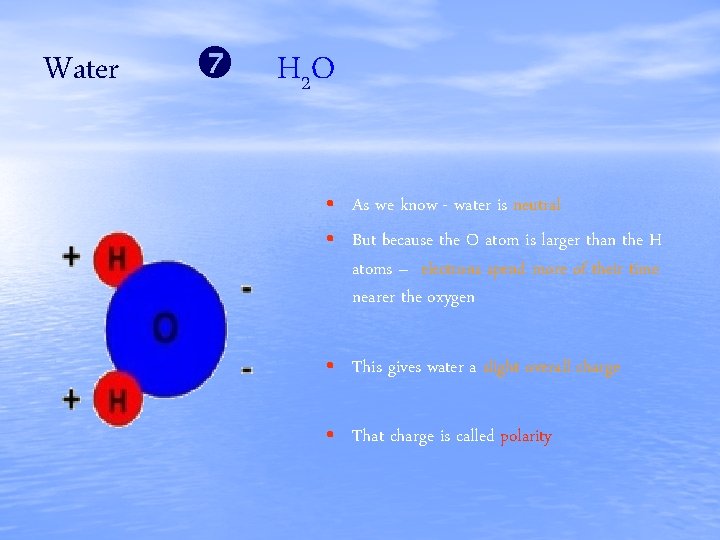
Water H 2 O • As we know - water is neutral • But because the O atom is larger than the H atoms – electrons spend more of their time nearer the oxygen • This gives water a slight overall charge • That charge is called polarity

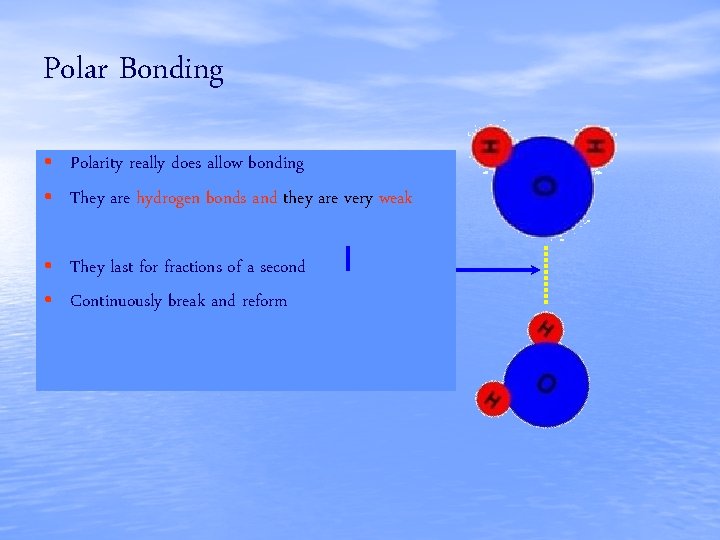
Polar Bonding • Polarity really does allow bonding • They are hydrogen bonds and they are very weak • They last for fractions of a second • Continuously break and reform

Forces due to polarity 1. Cohesion 2. Adhesion

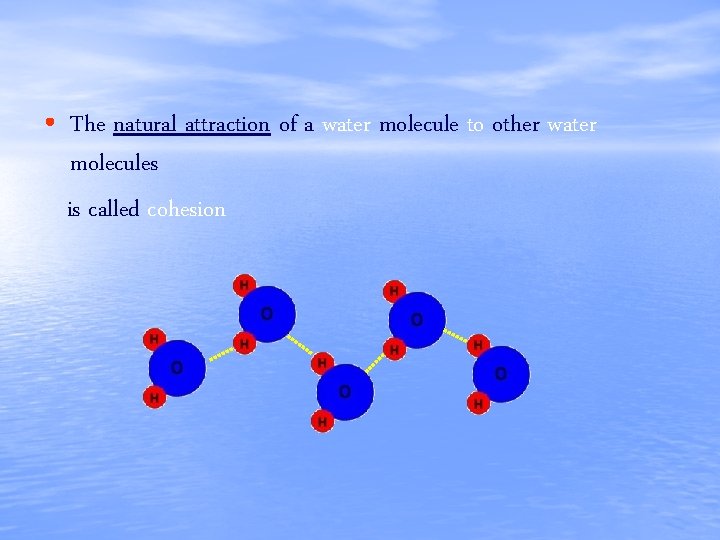
• The natural attraction of a water molecule to other water molecules is called cohesion

Cohesion • Can be seen as water droplets form

• The attraction of a water molecule to another polar molecule is adhesion • Molecules such as soil and clay (dust) • Surfaces like glass or paper straws • Certain clothing fibers and … animal hair

Adhesion • Can be seen as water droplets form on the spider web (another polar surface)

• Two simple properties associated with polarity are Capillary Action Surface Tension

Capillary Action 1. We know that gravity is ALWAYS pulling on objects with mass 2. Yet water can move up a paper towel with relative ease - How can this 3. 4. happen? Because the positive and negative charges in the paper attract the polar water molecules (adhesion) This property of adhesion is called capillary action


Surface Tension 1. 2. 3. 4. Inside a drop of water polar water molecules attract to each other in a random fashion At the surface of the drop, water does not attract to the air A unified layer of molecules at the surface creates surface tension There the water behaves like an flexible sheet allowing denser objects to “sit” on the surface

Surface Tension

Review Polarity hydrogen bonding Forces cohesion Forces adhesion Properties surface tension capillary action

The End polarity