The Practical Side of Nucleotide Metabolism November 29

The Practical Side of Nucleotide Metabolism November 29, 2001

The Plan for Today • Finish up Tuesday’s Leftovers • Brief Explanation of how d. UMP is converted to d. TMP • Some clinically relevant treatments based on these pathways that are used to combat: – Cancer – Viral Infections

Beyond AMP, GMP and UMP Purine Biosynthesis Pyrimidine Biosynthesis But other forms of these nucleotides are needed

Two Problems • These are monophosphates (i. e. GMP)- we need triphosphates (i. e. GTP) for both DNA and RNA synthesis • These are ribonucleotides- that’s fine for RNA but we also need to make DNA Synthesis of ribonucleotides first supports the RNA world theory

Specific Kinases Convert NMP to NDP Nucleoside Monophosphates Nucleoside Diphosphates Monophosphate Kinases • Monophosphate kinases are specific for the bases Adenylate Kinase AMP + ATP 2 ADP Guanylate Kinase GMP + ATP GDP + ADP

Conversion of Ribonucleotides to Deoxyribonucleotides BASE 5´ 1´ 4´ 3´ 2´ 4´ BASE 5´ 1´ 3´ 2´ Ribonucleotide Reductase Deoxyribonucleoside Ribonucleoside Somehow we need to get rid of this oxygen

Ribonucleotide Reductase • • • Catalyzes conversion of NDP to d. NDP Highly regulated enzyme Regulates the level of cellular d. NTPs Activated prior to DNA synthesis Controlled by feedback inhibition

d. NDP to d. NTP (the final step) • Once d. NDPs are generated by ribonucleotide reductase a general kinase (nucleoside diphosphate kinase) can phosphorylate to make the d. NTP’s Nucleoside diphosphate kinase ATP

Beyond d. GTP, d. ATP and d. UTP • So far we’ve made GTP, ATP, and UTP for incorporation into RNA • Also d. GTP and d. ATP for incorporation into DNA • We still need d. CTP for both RNA and DNA • We also need to generate d. TTP for DNA
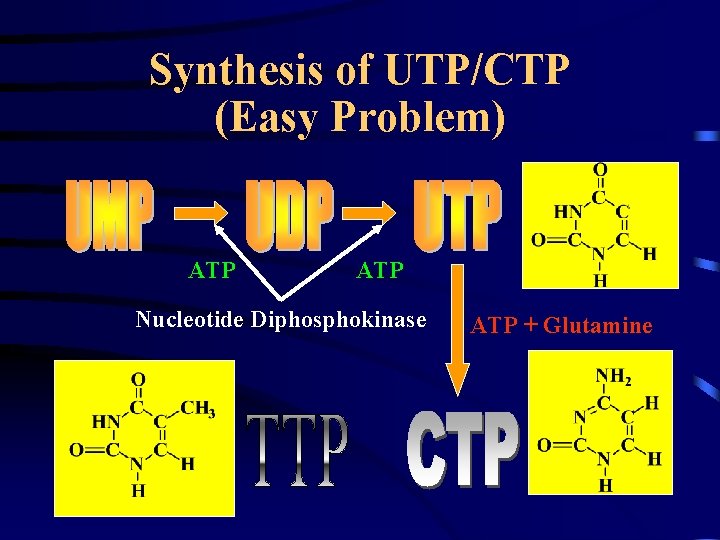
Synthesis of UTP/CTP (Easy Problem) ATP Nucleotide Diphosphokinase ATP + Glutamine
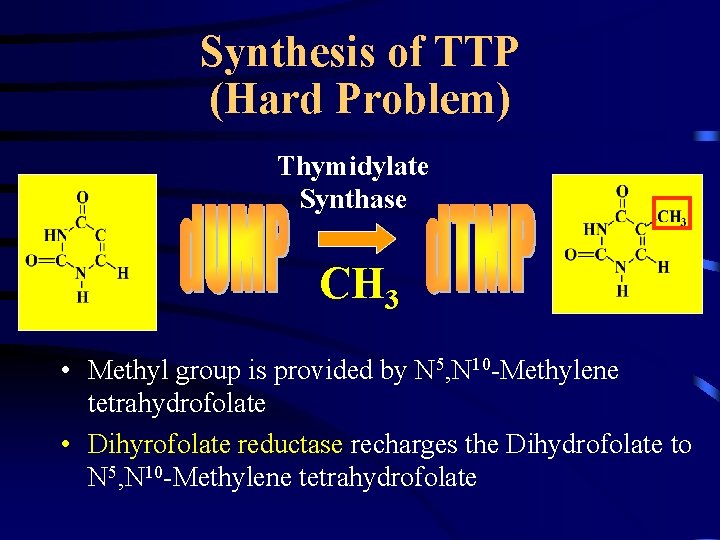
Synthesis of TTP (Hard Problem) Thymidylate Synthase CH 3 • Methyl group is provided by N 5, N 10 -Methylene tetrahydrofolate • Dihyrofolate reductase recharges the Dihydrofolate to N 5, N 10 -Methylene tetrahydrofolate
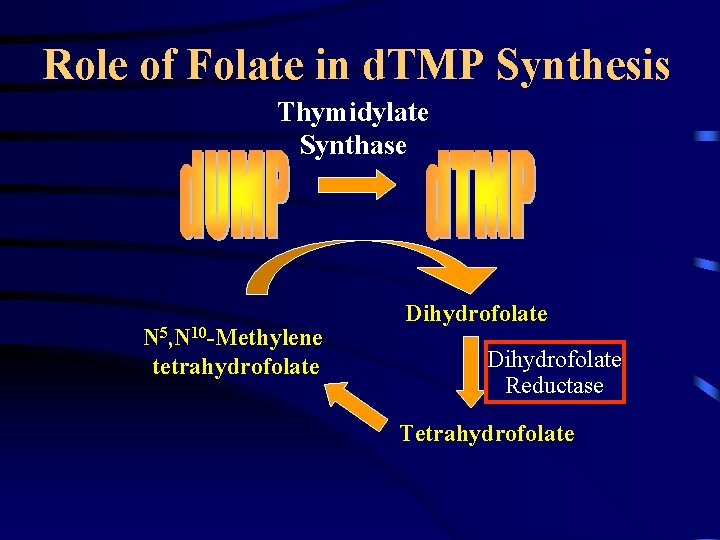
Role of Folate in d. TMP Synthesis Thymidylate Synthase N 5, N 10 -Methylene tetrahydrofolate Dihydrofolate Reductase Tetrahydrofolate

The Plan for Today • Finish up Yesterday’s Leftovers • Brief Explanation of how d. UMP is converted to d. TMP • Some clinically relevant treatments based on these pathways that are used to combat: – Cancer – Viral Infections

Antimetabolites • Often drugs that inhibit cell growth are used to combat cancer • Many of these compounds are analogues of purine and pyrimidine bases or nucleotides • Many of these drugs must be activated by cellular enzymes • They affect nucleic acid synthesis and tumor cells tend to be more susceptible since they are dividing more rapidly

6 -Mercaptopurine (6 -MP) • Purine Analogue • Used clinically to combat childhood leukemia • Since 1963 cure rate has increased from ~4% to greater than 80% Inhibitor of Committed Step in de novo Purine Biosynthesis PRPP + 6 -MP 6 -mercaptopurine ribonucleotide This reaction is more active in tumor cells
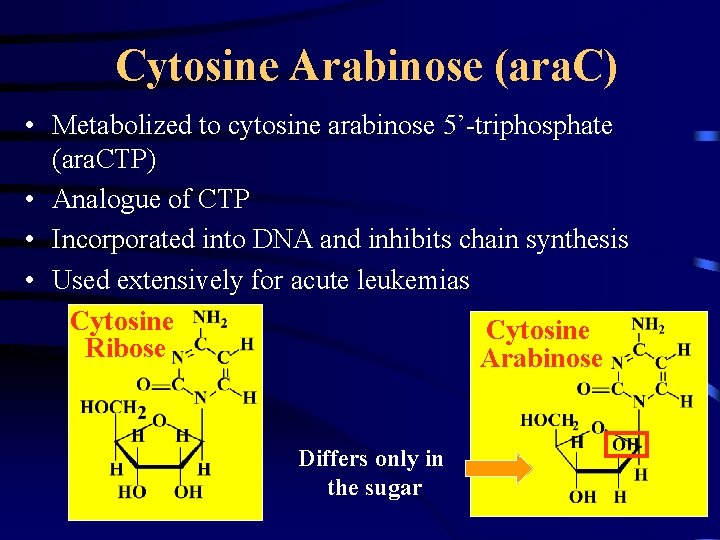
Cytosine Arabinose (ara. C) • Metabolized to cytosine arabinose 5’-triphosphate (ara. CTP) • Analogue of CTP • Incorporated into DNA and inhibits chain synthesis • Used extensively for acute leukemias Cytosine Ribose Arabinose Differs only in the sugar
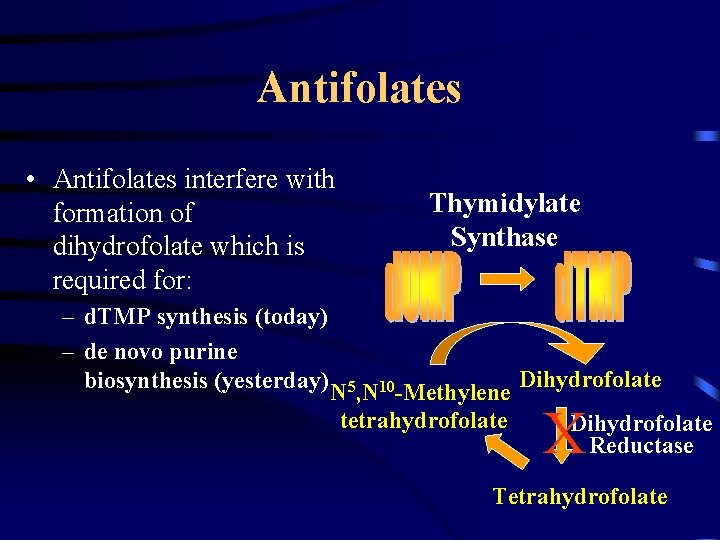
Antifolates • Antifolates interfere with formation of dihydrofolate which is required for: Thymidylate Synthase – d. TMP synthesis (today) – de novo purine biosynthesis (yesterday) N 5, N 10 -Methylene Dihydrofolate tetrahydrofolate X Dihydrofolate Reductase Tetrahydrofolate
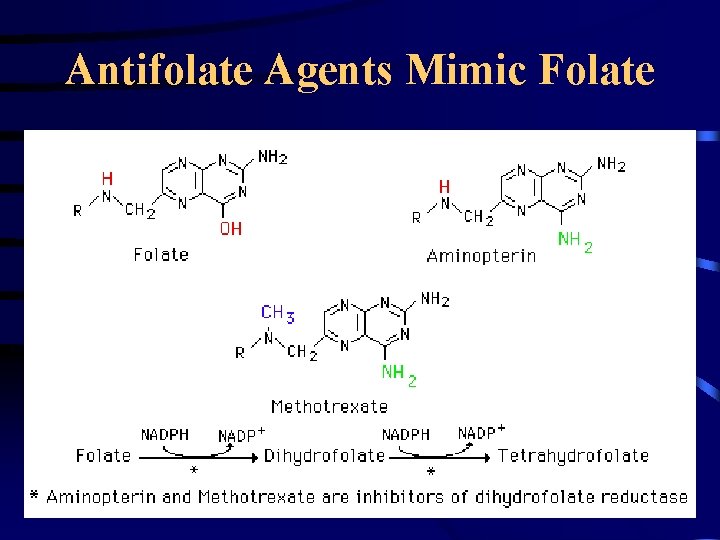
Antifolate Agents Mimic Folate
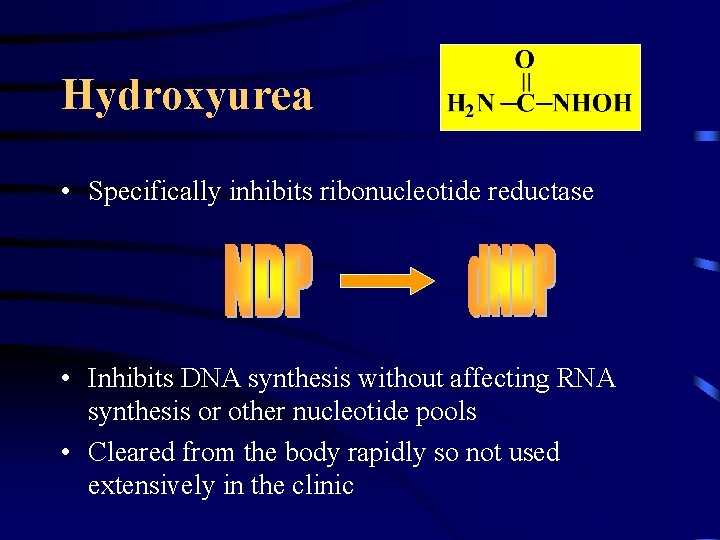
Hydroxyurea • Specifically inhibits ribonucleotide reductase • Inhibits DNA synthesis without affecting RNA synthesis or other nucleotide pools • Cleared from the body rapidly so not used extensively in the clinic

Practical Considerations • Most of these agents are used in combination therapies • Many need to be processed in cells to create the active compound • Often are not specific for tumor cells but rather for rapidly dividing tissues • Multiple modes of drug resistance can and do develop (Specific or General)

Example of Specific Drug Resistance: Methotrexate • Methotrexate works by inhibiting the function of dihydrolfolate reductase (DHFR) • Cells develop ways to avoid this block – Mutations in DHFR that make it bind less tightly to MTX – Amplication of the DHFR gene (more enzyme activity)

Anti-Viral Therapies • Target virally infected cells • Take advantage of aspects of viral metabolism that differ from normal cellular metabolism HIV- Human Immunodeficiency Virus HSV- Herpesvirus

AZT as an Anti-HIV Agent • • Azido-3’-deoxythymidine Pyrimidine Analogue HIV is a retrovirus RNA genome that is reverse -transcribed to DNA • Viral polymerase is inhibited by AZT
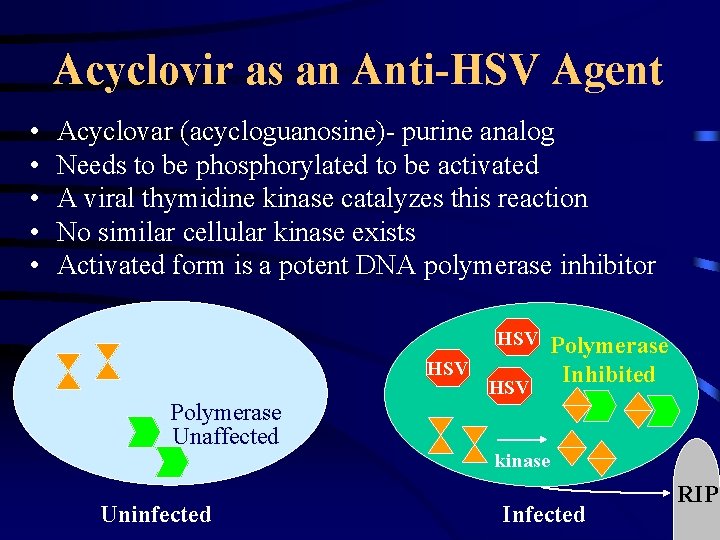
Acyclovir as an Anti-HSV Agent • • • Acyclovar (acycloguanosine)- purine analog Needs to be phosphorylated to be activated A viral thymidine kinase catalyzes this reaction No similar cellular kinase exists Activated form is a potent DNA polymerase inhibitor HSV Polymerase HSV Inhibited HSV Polymerase Unaffected Uninfected kinase Infected RIP

The BIG Picture • GMP, AMP, UMP on…. . • Generation of d. TMP • Common features of clinically relevant antimetabolites/antifolates • Antiviral agents- how are they specific for the virally infected cells?
- Slides: 26