The PolyzeneF Nano Coated Cobalt Chromium Alloy Stent

The Polyzene-F Nano Coated Cobalt Chromium Alloy Stent: Results from the ATLANTA Study by Alessio La Manna, MD

Background DES have shown to reduce post-stenting restenosis, but there are some issues: Ø Need for long-term dual antiplatelet therapy with related cost and possible risk of bleeding Ø Risk of early antiplatelet therapy discontinuation or drug resistance (comorbidities, patient compliance, drug intolerance) Ø Increased risk of stent thrombosis (event mortality rate ~ 40%) compared to BMS, particularly in the long term follow-up

Polyzene®-F Surface Modification • • • Anti-Inflammatory Bacterial Resistant Promotes Healthy Endothelial Cell Growth • • • Inorganic High Molecular Weight Ultra-Pure – Residual Chlorine < 0. 003% • • Can Coat Multiple Substrates Applied on Stent at 40 Nanometers

What is Polyzene-F? It is an inorganic, hydrophobic, highly biocompatible, nanothin polymer with some important properties: ØLow activation of platelets ØLow platelet adhesion ØLow GP IIB/IIIA receptor density ØLow activation of clotting and complement systems ØLow or no infiltration of inflammatory cells ØLow foreign body reaction

Polyzene®-F Surface Modification • Stained Platelets Appear Green • Polyzene®-F Does Not Activate Platelets Untreated Polyzene®-F Treated Mrowietz et al, Clinical Hemorheology and Microcirculation 32; (2005) 89– 103

7 Days after Stent Implantation Uncoated Coated
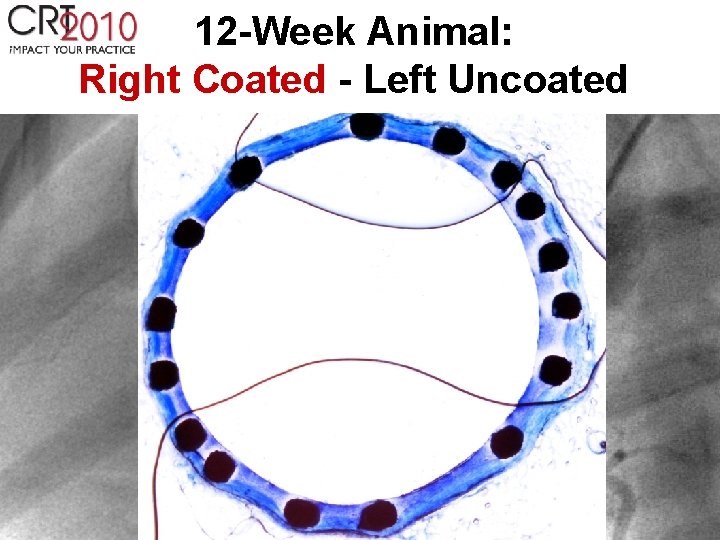
12 -Week Animal: Right Coated - Left Uncoated
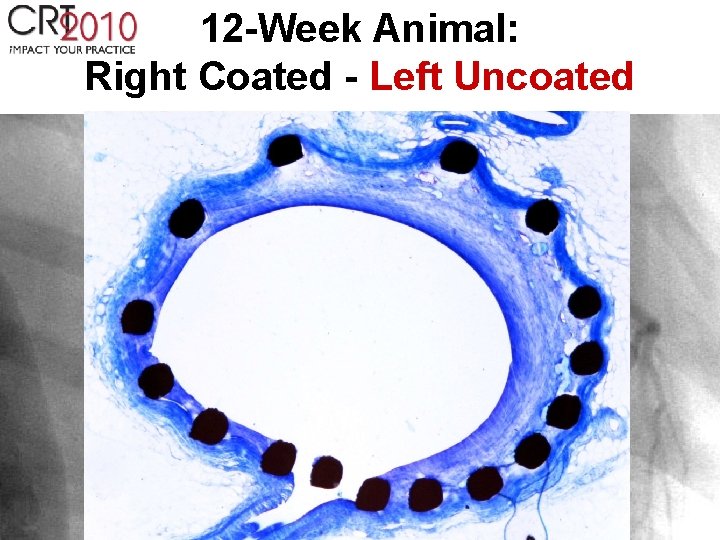
12 -Week Animal: Right Coated - Left Uncoated
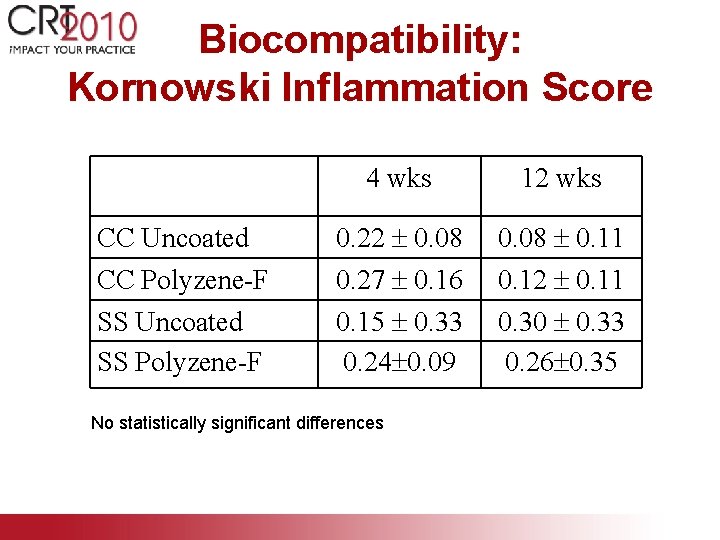
Biocompatibility: Kornowski Inflammation Score 4 wks 12 wks CC Uncoated CC Polyzene-F 0. 22 0. 08 0. 27 0. 16 0. 08 0. 11 0. 12 0. 11 SS Uncoated SS Polyzene-F 0. 15 0. 33 0. 24 0. 09 0. 30 0. 33 0. 26 0. 35 No statistically significant differences

CATANIA Stent Ø Ø Nanothin (~40 nm) Surface of Ultra-Pure Polyzene®-F Co-Cr Alloy Stent Modified Open Cell Design Small and Large Version Ø Strut Diameter 65 μm and 74 μm Ø Lengths from 8 -38 mm Ø Semi-Compliant Balloon Ø Balloon Diameter Ø 2. 0, 2. 25, 2. 75, 3. 0, 3. 25, 3. 50, 4. 0 Ø Maximum Guidewire 0. 014” (0. 356 mm)

The CATANIA™ Stent System “The CAT” Ormiston, TCT 2004 Polyzene-F is Applied ~ 40 Nanometers Drug-Eluting Polymers are Thick, Sticky, Crack & Break

Assessment of The LAtest Non-Thrombogenic Angioplasty Stent Sponsor: Principal Investigator: Co-Investigators: Study Monitor: Independent Core-Lab: Independent CEC: Ethical Committee: Celo. Nova Bio. Sciences Inc. , Newnan GA, USA C. Tamburino 1, MD, Ph. D, FESC, FSCAI A. La Manna 1, MD, M. E. Di Salvo 1, MD, G. Sacchetta 1 , MD T. Corcos 2, MD, FESC, FSCAI, FACC F. Prati 3, MD, FESC C. Vassanelli 4, MD, Ph. D, FESC Azienda Ospedaliera V. E. Ferrarotto, S. Bambino 1 Cardiology Department, Ferrarotto Hospital, University of Catania, Italy Catheterization and Interventional Laboratory, Clinique Turin, Paris, France 3 San Giovanni Hospital & Rome Heart Research (RHR), Rome, Italy 4 Cardiovasclar Department, University of Verona, Italy 2 Cardiac JACC Cardiovasc. Interv. 2009; 2: 197 -204

Aim and Endpoints SAFETY - EFFICACY: 55 pts with CAD and PCI Primary endpoints Secondary endpoints ØCardiac ØProcedural death ØIndex vessel related non fatal MI ØTVR non-TLR ØStent thrombosis (ARC) success ØOverall mortality ØBinary restenosis ØLLL ØNIHV JACC Cardiovasc. Interv. 2009; 2: 197 -204

Inclusion Criteria Ø Age > 30 < 70 years Ø Clinical or diagnostic documentation of stable or unstable myocardial ischemia Ø De novo type A and/or B 1 AHA/ACC lesion on a major coronary vessel Ø < 2 stent/patient (except bail-out) Ø 1 stent/lesion (except bail-out) Ø Lesion length < 20 mm Ø Vessel size > 2. 5 < 3. 5 mm JACC Cardiovasc. Interv. 2009; 2: 197 -204
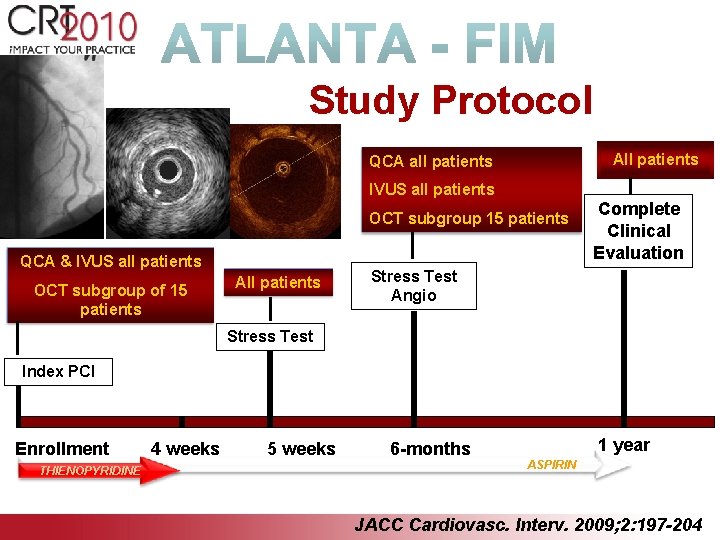
Study Protocol All patients QCA all patients IVUS all patients OCT subgroup 15 patients QCA & IVUS all patients OCT subgroup of 15 patients All patients Complete Clinical Evaluation Stress Test Angio Stress Test Index PCI Enrollment THIENOPYRIDINE 4 weeks 5 weeks 6 -months 1 year ASPIRIN JACC Cardiovasc. Interv. 2009; 2: 197 -204

Baseline and Angiographic Characteristics (55 pts) Ø Mean age 58 ± 9 yrs Ø Male gender 41 pts (75%) Ø Diabetes 19 patients (34%) (13 patients NIDD, 6 patients IDD) Ø Hypertension 33 patients (60%) Ø Unstable angina 35 patients (63%) Ø Recent MI (> 48 hours < 4 weeks) 6 patients (11%) Ø Mean LVEF 52 ± 7 % Ø Lesion type B 53 (70%) Ø Lesion type C 15 (20%) Ø Two vessels treated 24% JACC Cardiovasc. Interv. 2009; 2: 197 -204

Procedural Characteristics Ø Total stents implanted Ø Total lesions treated Ø Stents/patient Ø Lesions/patient Ø Stents/lesion Ø Mean stent length Ø Mean stent diameter Ø Mean stent length/patient Ø Mean stent length/lesion 89 76 1. 4 1. 2 17. 1 4 2. 9 0. 3 26. 6 14 19. 2 8 JACC Cardiovasc. Interv. 2009; 2: 197 -204
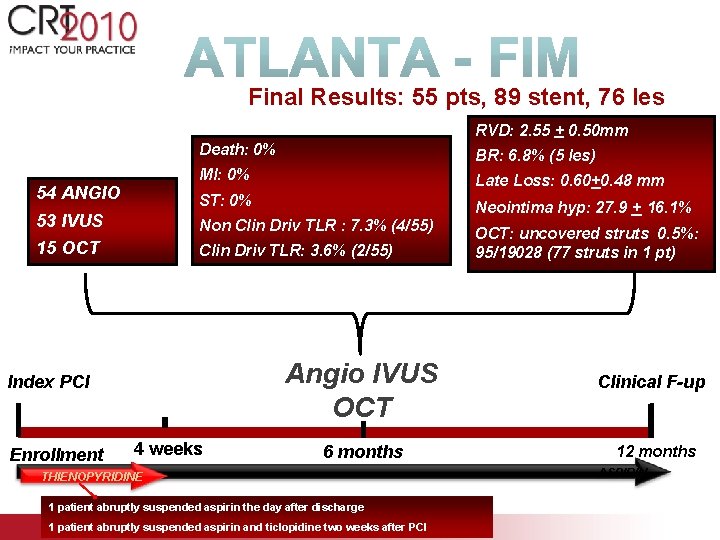
Final Results: 55 pts, 89 stent, 76 les RVD: 2. 55 + 0. 50 mm 54 ANGIO Death: 0% BR: 6. 8% (5 les) MI: 0% Late Loss: 0. 60+0. 48 mm ST: 0% Neointima hyp: 27. 9 + 16. 1% 53 IVUS Non Clin Driv TLR : 7. 3% (4/55) 15 OCT Clin Driv TLR: 3. 6% (2/55) Angio IVUS OCT Index PCI Enrollment 4 weeks 6 months THIENOPYRIDINE 1 patient abruptly suspended aspirin the day after discharge 1 patient abruptly suspended aspirin and ticlopidine two weeks after PCI OCT: uncovered struts 0. 5%: 95/19028 (77 struts in 1 pt) Clinical F-up 12 months ASPIRIN

FU OCT data We analysed • 15 stents • 1. 904 cross-sections • 19. 028 stent struts Am J Cardiol 2009; 103: 1551 -5

FU OCT data Stent struts coverage at OCT in 99, 5% of struts …………. only 0, 5% of struts were uncovered Am J Cardiol 2009; 103: 1551 -5

FU OCT data Complete apposition at OCT in 99, 85% of struts …………. only 0, 15% of struts were malapposed Am J Cardiol 2009; 103: 1551 -5
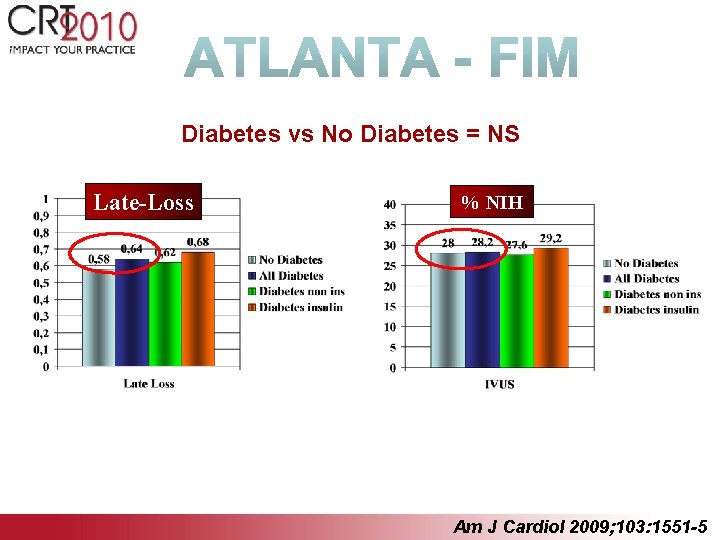
Diabetes vs No Diabetes = NS Late-Loss % NIH Am J Cardiol 2009; 103: 1551 -5

Conclusions Results of the trial are very promising: Ø 100% technical success Ø 0% death, MI, stent thrombosis, @ 12 months Ø Low binary restenosis and TLR Ø Full stent healing @ IVUS and OCT Ø Dual antiplatelet therapy ended after one month Ø No differences between diabetics and non diabetics Ø The absence of stent thrombosis confirms the strong preclinical evidence that a Polyzene-F surface treatment’s gives very positive effects
- Slides: 23