The Pi XL Club The Pi XL Club

The Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL Club T Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL Club The P Club The Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL Club The Pi. XL C The Pi. XL Club The Pi. XL Club The Pi. XL Club T Pi. XL Club The Pi. XL Club The Pi. XL Club The P Club The Pi. XL Club The Pi. XL Club The Pi. XL C The Pi. XL Club The Pi. XL Club T Pi. XL Club The Pi. XL Club The P Club The Pi. XL Club The Pi. XL C The Pi. XL Club The Pi. XL Club T Pi. XL Club The Pi. XLThis Club The Pi. XL Club Pi. XL for Clubthe Theuse Pi. XLof Club The Pi. XLschools Club The for Pi. XLas Club Theas Pi. XLthey Clubremain The Pi. XLmembers Club The Pi. XLClub The Pi. XL Club The P resource is The strictly member long of Club The Pi. XL Club. It. The Pi. XL Club The Pi. XL C may not be copied, sold nor transferred to a third party or used by the school after membership ceases. The Pi. XL Club The Until Pi. XL Club The Pi. XL Club T such time it may be freely used within the member school. Pi. XL Club The Pi. XL Club The P Club The Pi. XL Club The Pi. XL C and. Club contributions of the authors. The contents this are The not. Pi. XL connected with. Club The Pi. XL Club The All Pi. XLopinions Club The Pi. XL Clubare Thethose Pi. XL Club The of Pi. XL Clubresource The Pi. XL Club The Pi. XL nor endorsed by any other company, organisation or institution. Pi. XL Club The Pi. XL Club The Pi. XL Club The P Club The Pi. XL Club The Pi. XL Club The Pi. XL C The Pi. XL Club The Pi. XL Club The Pi. XL Club T Pi. XL Club The Pi. XL Clubwww. pixl. org. uk The Pi. XL Club The Pi. XL Club Thenumber Pi. XL Club 07321607 The Pi. XL Club The P Pi. XLClub. The Ltd, Pi. XL Company Club The Pi. XL Club The Pi. XL C The Pi. XL Club The Pi. XL Club T Required Practical Guide Chemistry Neutralisation by titration This document contains: • • • Links to You. Tube clips showing the practical procedure Information from examination boards AQA, OCR, Edexcel Potential examination questions and answers Commissioned by The Pi. XL Club Ltd. November 2016 © Copyright The Pi. XL Club Ltd, 2016

Required practical – exam boards • AQA
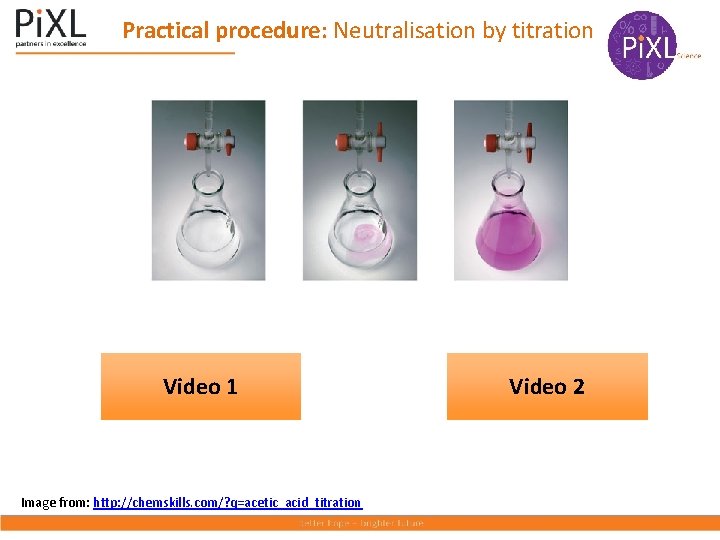
Practical procedure: Neutralisation by titration Video 1 Image from: http: //chemskills. com/? q=acetic_acid_titration Video 2

Possible examination style questions 1. Name the equipment used to measure the volume of acid used to neutralise an alkali. 2. What does the term ‘concordant results’ mean? 3. Name an indicator used when titrating and explain why universal indicator cannot be used. 4. 0. 5 M hydrochloric acid was titrated against 25 cm 3 of an unknown concentration of sodium hydroxide. Use the data below to calculate the mean titre. 5. Calculate the concentration of the sodium hydroxide. HCl + Na. OH H 2 O + Na. Cl Rough trial/ cm 3 First trial/ cm 3 Second trial/ cm 3 Third trial/ cm 3 Fourth trial/ cm 3 28. 0 27. 9 27. 6 27. 7 27. 6

Possible examination style questions answers 1. Name the equipment used to measure the volume of acid used to neutralise an alkali. ANS: • Burette

Possible examination style questions answers 2. What does the term ‘concordant results’ mean? ANS: • Results that agree or are consistent

Possible examination style questions answers 3. Name an indicator used when titrating and explain why universal indicator cannot be used. ANS: • Phenolphthalein • Methyl orange • Universal indicator does not have a clear endpoint

Possible examination style questions answers 4. 0. 5 M hydrochloric acid was titrated against 25 cm 3 of an unknown concentration of sodium hydroxide. Use the data below to calculate the mean titre. ANS: • Find the mean of the two concordant results • 27. 6 + 27. 6 = 27. 6 cm 3 2

Possible examination style questions answers 5. Calculate the concentration of the sodium hydroxide. HCl + Na. OH H 2 O + Na. Cl ANS: • Using the mean titre from previous question = 27. 6 cm 3 = 0. 0276 dm 3 • Calculate the moles of HCl used = volume x conc. • 0. 0276 x 0. 5 = 0. 0138 moles • This is equal to the moles of Na. OH = 0. 0138 • Calculate the concentration of Na. OH = moles ÷ volume = 0. 0138 ÷ 0. 025 dm 3 = 0. 55 M or 0. 6 M

Key questions: • What safety precautions should you take when carrying out this experiment and why? • Why is a burette used to measure the volume of one of the solutions instead of a measuring cylinder? • Why is it necessary to rinse the burette and pipette with the solutions they will contain before filling them? • Why is it necessary to use an indicator in the titration? • What are the colours of methyl orange in acid, neutral and alkaline solutions? • What are the colours of phenolphthalein in acid, neutral and alkaline solutions? • Why is universal indicator not suitable to use in a titration? http: //qualifications. pearson. com/content/dam/pdf/GCSE/Science/2016/teaching-and-learning-materials/GCSE-9 -1 -Sciences-core-practical-guide. pdf

Key questions: • Why is a white tile used? • Why is it important to swirl the conical flask during the titration? • What is meant by the end point? • Why do you repeat the titration? • What are concordant results? • Which results are included in the calculation when you determine the mean or average titre? http: //qualifications. pearson. com/content/dam/pdf/GCSE/Science/2016/teaching-and-learning-materials/GCSE-9 -1 -Sciences-core-practical-guide. pdf

Summary document A summary document is also available on Huddle which contains all the relevant information about this practical from the different examination boards. This document includes practical methods and other potential examination questions
- Slides: 12