The Physical Behavior of Matter What is Matter

The Physical Behavior of Matter

What is Matter? Anything that takes up space & has mass ¡ Substances – variety of matter that has the same composition and properties throughout. ¡ ¡ ¡ Two types are elements (Na) and compounds (Na. Cl). Mixtures – two or more substances mixed together (not united). Two types are homogeneous (salt water) and heterogeneous (sand sugar).

Substances ¡ ¡ Elements Cannot be decomposed (broken down) Ex: hydrogen (H 2), Oxygen (O 2) and nitrogen (N 2) Compounds ¡ ¡ ¡ Can be decomposed by a chemical change Chemically united Definite proportions Different properties Ex: H 2 O, Na. Cl, CO 2 A Binary Compound is a compound that only has two elements (Na. Cl)
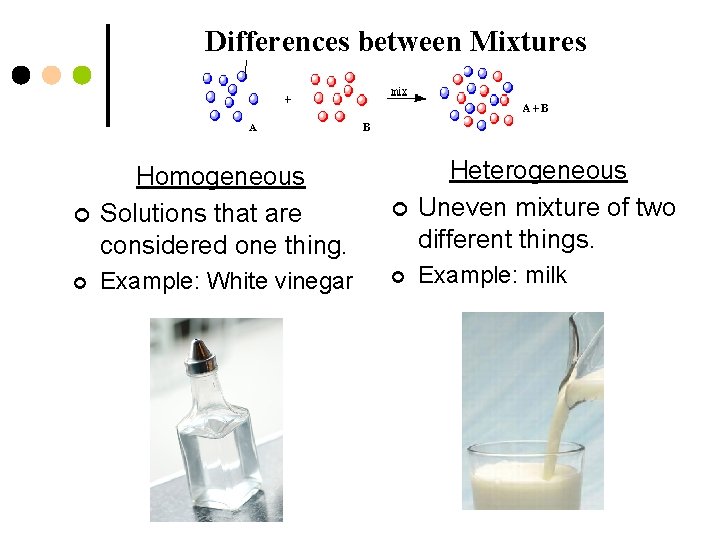
Differences between Mixtures ¢ Homogeneous Solutions that are considered one thing. ¢ Heterogeneous Uneven mixture of two different things. ¢ Example: White vinegar ¢ Example: milk

Ways To Separate Mixtures (1) § Distillation- a mixture of liquids can be separated by their boiling points. § Examples: distillation of salt water. distillation of petroleum liquids.

Ways to Separate Mixtures (2) • Filtration- separates the solid and liquid parts of a mixture. • Example: coffee filter which separates the coffee grounds from the brewed coffee.

Ways of Separating Mixtures (3) n Chromatography- way of separating different molecules in a mixture. n Example: separating components of chlorophyll.

Ways of Separating Mixtures (4) ¡ ¡ Centrifuge- a spinning machine that pushes the most dense particles to the bottom of the tube. Example: separate isotopes such as separating uranium hexafluoride, and uranium-235.

Ways to Separate Mixtures (5)

Properties Physical Properties Chemical Properties Can be found without changing the substance to something else. l Ex. Color, hardness, phase, solubility, odder, density, mass, volume l l Are found by making a substance react, and form a new substance. l Ex. Burning, reaction w/ water or acid, changing to a new substance.

Energy o o The ability to do work. Exothermic- energy given off in a chemical reaction. Endothermic- energy absorbed in a chemical reaction. You measure energy in joules (J).
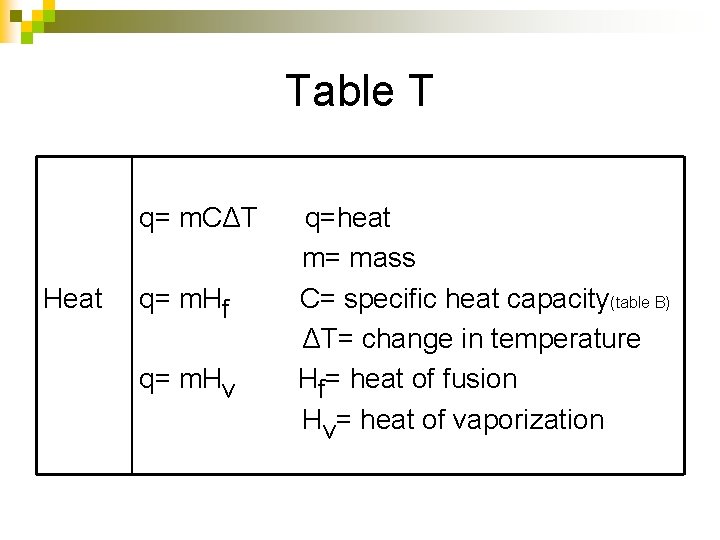
Table T q= m. CΔT Heat q= m. Hf q= m. Hv q=heat m= mass C= specific heat capacity(table B) ΔT= change in temperature Hf= heat of fusion Hv= heat of vaporization

Sample Problem: How much heat energy in joules if absorbed by 100 g of water when it is heated from 20ºC to 30ºC? q= m· C · ΔT q= 100 g x 4. 18 Joules/gºC x (30 – 20) ºC q= 100 g x 4. 18 J/gºC x 10ºC q= 4, 180 joules

Heating Curves l Heat of Fusion- is the amount of heat needed to change a solid into a liquid at a constant temperature. l Heat of Vaporization- is the amount of heat needed to change a liquid into a gas at a constant temperature. Table B Physical Constants for Water Heat of Fusion Heat of Vaporization 334 J/g 2260 J/g Specific heat capacity of H 2 O(l) 4. 18 J/gºC

Temperature § The measure of the average kinetic energy of the molecules. § The higher the temperature the more kinetic energy it has. § Heat flows from a higher temperature to a lower temperature until they are the same temperature. § Measured with a thermometer.

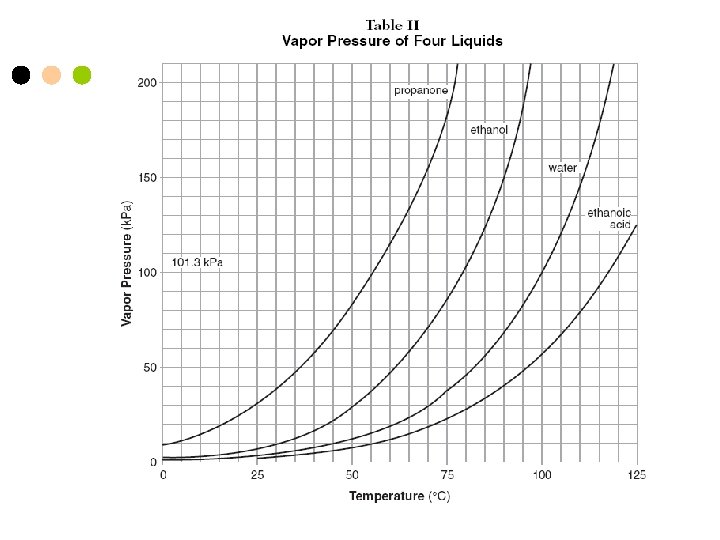
Temperature continued n Boiling Point- when the vapor pressure equals the atmospheric pressure. n Freezing Point- the temperature at which a liquid solidifies under a specified pressure. n n Absolute Zero= -273 o. C or 0 K Kelvin- K= Kelvin K = o. C + 273 ºC = degrees Celsius

Changes Physical Change -change in appearance, but no new substance is produced Ex- tearing a piece of paper, heating ice Chemical Change - Produces a new substance with different properties Ex- burning magnesium

Solids n n n Definite shape, definite volume, and crystalline structure, geometric pattern. Closely packed particles that vibrate but don’t change position. Melting point- temperature when a solid changes into a liquid. n Sublimation- the change from a solid directly to a gas. Ex. Dry ice (CO 2) & Iodine

Liquids n n n Definite volume, takes shape of its container. Particles are close together and move : water. Evaporation- when liquid changes into a gas. Ex: water vapor. Vapor Pressure- the pressure that the vapor exerts on the sides of the container.

Gases l l No definite volume or shape. Particles are far apart, and can expand anywhere. * When there is a phase change from solid to liquid to gas entropy increases
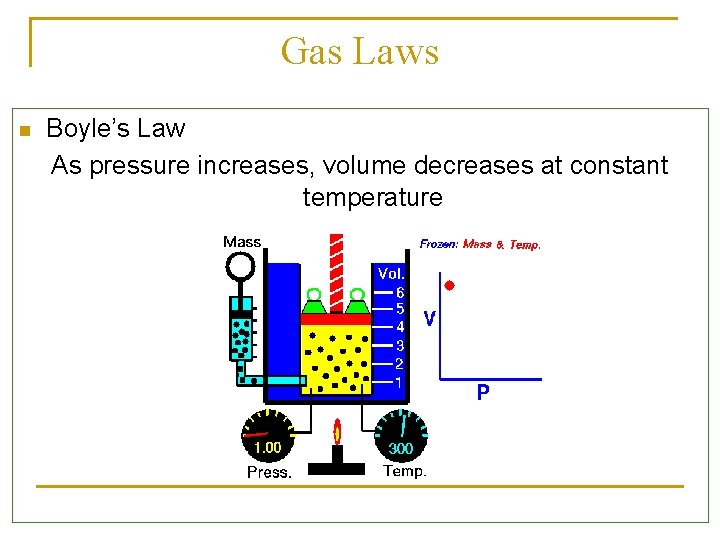
Gas Laws n Boyle’s Law As pressure increases, volume decreases at constant temperature
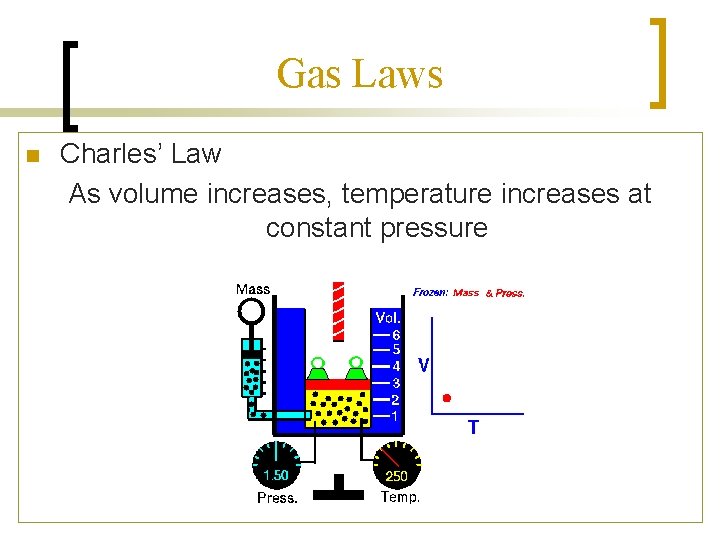
Gas Laws n Charles’ Law As volume increases, temperature increases at constant pressure

What is STP? o Standard Temperature and Pressure o When you have a combined gas law at STP, use 273 K as your temperature and 101. 3 k. Pa as the pressure.

Kinetic Molecular Theory (Ideal Gas Law) A model that tells how gasses should behave • Ideal gas- perfect gas that agrees with John Daltons 5 assumptions • • • Tiny particles Elastic collisions Gases are in constant motion No force of attraction Temperature is related to speed

Ideal Gas Real Gas • Particles have no • Particles have volume • No attractive forces • Attractive • Examples: H 2, He • Examples: Cl 2, H 2 O(g)

How do the Gas Laws relate to the Kinetic Molecular Theory? Boyle’s Law Charles’ Law • Boyle’s law states that • Charles’ Law states pressure and volume that as temperature are inversely increases, volume proportional. increases. • If you have a million • If you heat the air in a molecules in a container balloon, there will be and you decrease that more pressure on the container the molecules sides. This makes the will hit twice as often, balloon bigger in therefore twice the volume. pressure.

Combined Gas Law P 1 x V 1 T 1 = P 2 x V 2 T 2

THE END
- Slides: 29