The Periodic Table Valence Electrons and Bonding How

The Periodic Table, Valence Electrons and Bonding How Elements Form Compounds

• I CAN describe chemical bonding and determine an ionic chemical formula.

Chemical Bonding • Chemical Bonding is the joining of two or more atoms to form a compound. • Bonding occurs when atoms transfer electrons between them, or, in some cases, move close together and share electrons between them.

Valence Electrons • During bonding, the only electrons involved are those at the outer edge of the atom’s electron cloud. • These are called VALENCE ELECTRONS. • Atoms can have between 1 and 8 Valence Electrons [Ve-].

• When atoms come into close contact with each other, VALENCE ELECTRONS can be TRANSFERRED from one atom to another. • Typically, atoms on the LEFT SIDE of the periodic table [the METALS] LOSE electrons. • Atoms on the RIGHT SIDE of the periodic table [the NON-METALS] gain electrons.

• The number of electrons GAINED or LOST depends on the GROUP NUMBER the atom is in: • LOSE ELECTRONS GAIN ELECTRONS • Group 1 loses 1 e • Group 2 loses 2 e • Groups 13 loses 3 e- Group 15 gains 3 e. Group 16 gains 2 e. Groups 17 gains 1 e-

Atoms become IONS • When an atom gains or loses electrons and there is a change in its overall charge, the atom is transformed into another form called an ION

• POSITIVE IONS are called CATIONS and are formed from METALS. • NEGATIVE IONS are called ANIONS and are formed from NON-METALS.
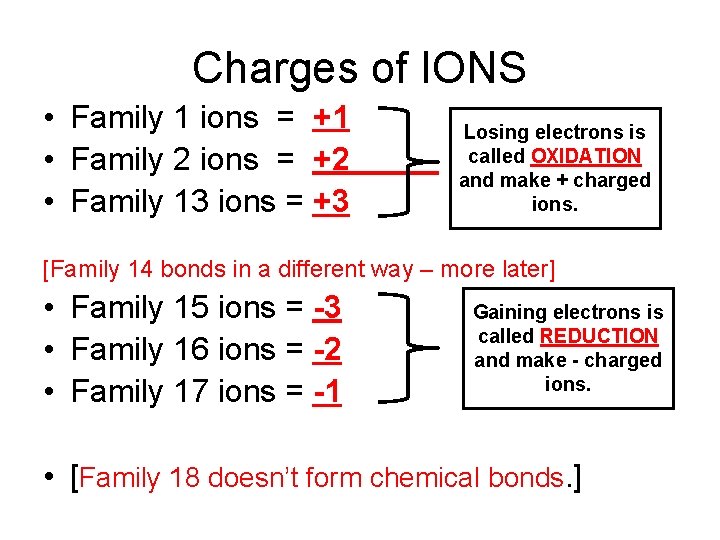
Charges of IONS • Family 1 ions = +1 • Family 2 ions = +2 • Family 13 ions = +3 Losing electrons is called OXIDATION and make + charged ions. [Family 14 bonds in a different way – more later] • Family 15 ions = -3 • Family 16 ions = -2 • Family 17 ions = -1 Gaining electrons is called REDUCTION and make - charged ions. • [Family 18 doesn’t form chemical bonds. ]

IONIC BONDING • IONIC BONDING occurs between two IONS with OPPOSITE CHARGES. • Remember OPPOSITES ATTRACT!

Characteristics of IONIC Compounds • Generally consist of a METAL CATION and a NON-METAL ANION. • High melting points • Almost always solid at room temperature • Brittle • Generally not very flammable • Dissolve in water [electrolytes]

Forming an IONIC Compound • The simplest IONIC COMPOUNDS are called BINARY IONIC compounds because they consist of only TWO DIFFERENT kinds of atoms…. . one a POSITIVE ION from the metal side of the PT and the other a NEGATIVE ION from the non-metal side of the PT. • Together their CHARGES add up to equal ZERO. • PROBLEM • What would be the formula for a compound resulting from ionic bonding between Potassium and Bromine? • STEPS • 1. Find the elements in the Periodic Table and write down their symbols. • 2. Use the element’s location on the Periodic Table to predict the charge of its ION. • 3. Criss-cross the charges (NO SIGNS) and multiply. • 4. Write the FORMULA.
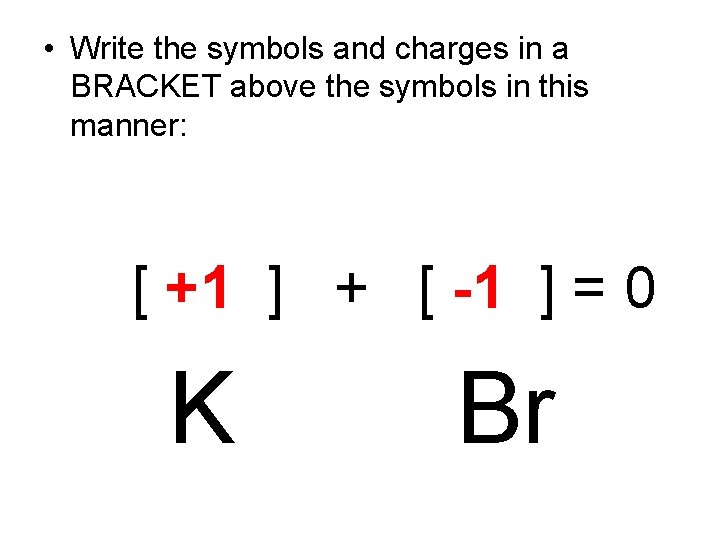
• Write the symbols and charges in a BRACKET above the symbols in this manner: [ +1 ] + [ -1 ] = 0 K Br
![• BALANCE the charge by criss-crossing the numbers [don’t worry about charges] and • BALANCE the charge by criss-crossing the numbers [don’t worry about charges] and](http://slidetodoc.com/presentation_image/98f11fc2cf5e0323f5c181b79b9ed263/image-14.jpg)
• BALANCE the charge by criss-crossing the numbers [don’t worry about charges] and multiplying each bracket. 1 [ +1 ] + 1 [ -1 ] = 0 K Br

• To write the FORMULA, • Write the symbol of the POSITIVE ION first, the NEGATIVE ION second. – Place the number you MULTIPLIED the bracket by AFTER and slightly BELOW the symbol. – This number is called a SUBSCRIPT. 1[ +1 ] + 1[ -1 ] = 0 K Br K 1 Br 1

• If the number is ONE, it is understood and you DO NOT have to write it. – Think of X in math…. 1 X = X • So the formula for a compound of Potassium and Bromine is: • KBr

• PROBLEM • What is the formula for an ionic compound of Magnesium and Chlorine?
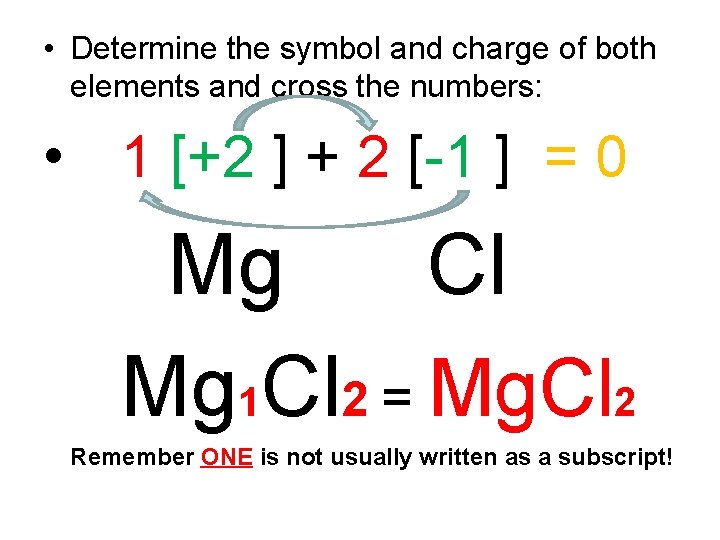
• Determine the symbol and charge of both elements and cross the numbers: • 1 [+2 ] + 2 [-1 ] = 0 • Mg Cl • Mg 1 Cl 2 = Mg. Cl 2 • Remember ONE is not usually written as a subscript!

Problem What is the formula of an ionic compound of Aluminum and Oxygen? • [ +3] + [ -2 ] • Al O
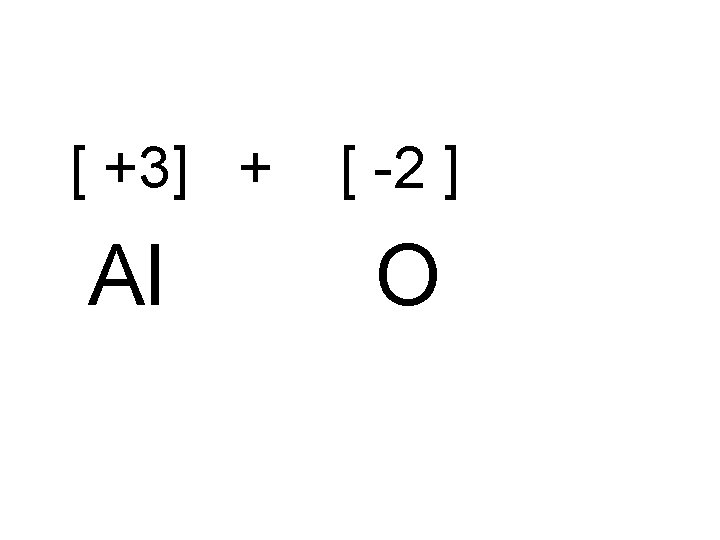
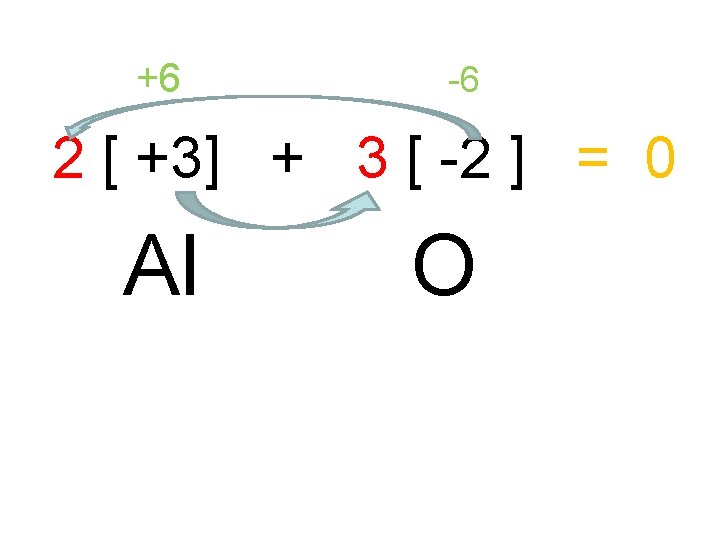

• So the formula becomes: • Al 2 O 3
- Slides: 22