The Periodic Table Trends Pg 335 336 Bonds

The Periodic Table Trends - Pg 335 -336 Bonds – Pg 334

Periodic Table • Is a chart that lists atoms by their atomic number and their electron configurations (arrangement of electrons) • Periodic – pattern keeps repeating • Each box contains 4 things: – Its name – Its symbol – Its atomic number (# of protons/electrons) – Its atomic mass (At the bottom)

Going from up to down • Each row is called a period • Each element has another orbital filled to capacity (1 st – 2 e-, 2 nd – 8 e-, 3 rd – 8 e- ) • As orbits are filled, the atomic radius (size) also increases • Elements are less reactive as we go down the rows

Going from left to right • Each column is called a group • A proton and electron is added to every element. • Ex: H – 1 proton, He – 2 protons • Li – 3 protons Be– 4 protons/electrons • So the atomic number (protons) increases. • The number of valence (outermost) electrons also increases

In each group (column) • There are the same number of valence electrons in the group • Every element in the group has similar properties because of the same number of valence electrons. • Ex: H – 1 valence electron Li – 1 valence electron Na – 1 valence electron

Group 1 – Alkali Metals • Properties: – Have an extra valence electron and are very reactive – Very soft or brittle – Not found freely in nature because of their high reactivity Ex: Li, Na. . . s-orbitals are filled first

Group II – Alkali Earth Metals • Properties – Two extra valence electrons that are easily lost, but not as easily as Alkali Metals – Less reactive than the Alkali Metals – Not very soft – S-orbitals are filled

Group VII Halogens • Properties – These are non-metals – They easily gain an electron to form a negative ion (Cl-) – Very reactive, reacts rapidly with Groups I and II – P - orbitals are filled Ex: F, Cl, Br, I

Group VIII – Noble Gases • Properties – Non-metals – Completely filled valence electrons – Because of completely filled valence electrons, they are highly NON-reactive. – P-orbitals are filled

Bonds • Pg 334
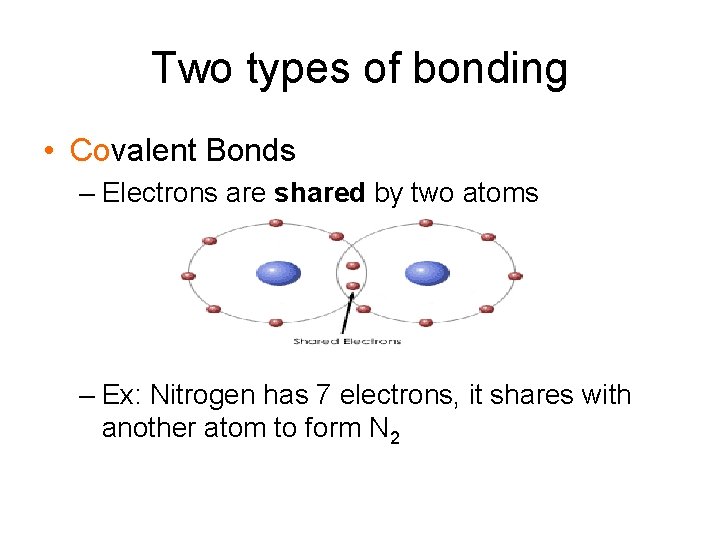
Two types of bonding • Covalent Bonds – Electrons are shared by two atoms – Ex: Nitrogen has 7 electrons, it shares with another atom to form N 2

Ionic bonds • In this type of bond, electrons from one atom are removed and attached to another atom, resulting in positive or negative ions • Generally involves 1 metallic atom and 1 non-metallic atom or 1 atom from the left side and 1 from the right of the table • Ex: Na. Cl
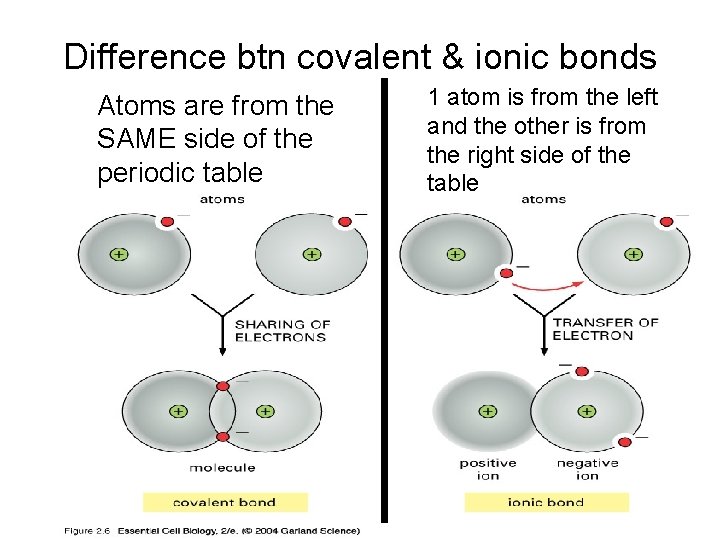
Difference btn covalent & ionic bonds Atoms are from the SAME side of the periodic table 1 atom is from the left and the other is from the right side of the table

Review • Periodic table is an arrangement of atoms based on their atomic number • Difference between Alkali Metals and Alkali Earth metals • Halogen and Noble gas groups • Difference between covalent and ionic bonds
- Slides: 14