The Periodic Table Topic 5 Click for song

The Periodic Table Topic 5 Click for song SMB, Periodic Table Notes 2011 1

Searching For an Organizing Principle Chlorine, bromine, and iodine have very similar chemical properties. SMB, Periodic Table Notes 2011 2

6. 1 Mendeleev’s Periodic Table z. Mendeleev’s Periodic Table (1869) y. How did Mendeleev organize his periodic table? y. The Abbreviated History of the Periodic Table for Regents Chemistry SMB, Periodic Table Notes 2011 3

Click on pix for history SMB, Periodic Table Notes 2011 4

I. HISTORY A. Dmitri Mendeleev (1869, Russian) z Organized elements by increasing ATOMIC MASS. q. Elements with similar chemical properties were grouped together. q. There were some discrepancies. SMB, Periodic Table Notes 2011 5

B. Henry Moseley q ORGANIZED ELEMENTS BY INCREASING ATOMIC NUMBER. q Resolved discrepancies in Mendeleev’s arrangement. SMB, Periodic Table Notes 2011 6

6. 1 The Periodic Law x. In the modern periodic table, elements are arranged in order of increasing atomic number. SMB, Periodic Table Notes 2011 7
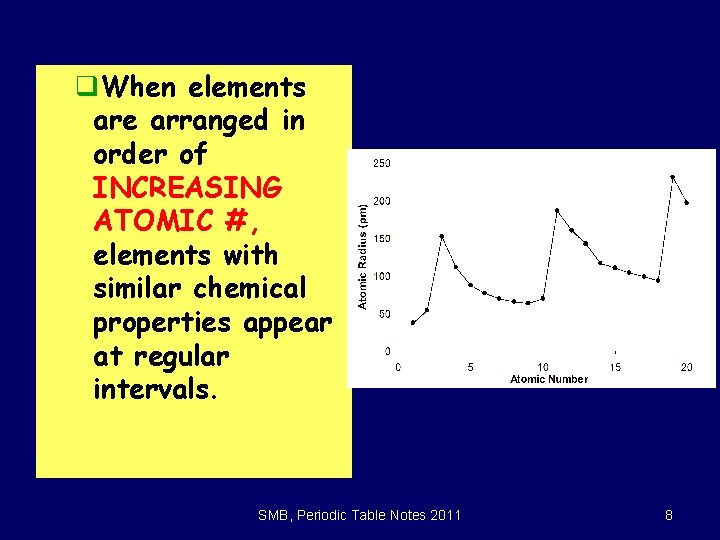
q. When elements are arranged in order of INCREASING ATOMIC #, elements with similar chemical properties appear at regular intervals. SMB, Periodic Table Notes 2011 8

6. 1 The Periodic Law x. The periodic law: When elements are arranged in order of increasing atomic number, there is a periodic repetition of their physical and chemical properties. • The properties of the elements within a period change as you move across a period from left to right. • The pattern of properties within a period repeats as you move from one period to the next. SMB, Periodic Table Notes 2011 9
Periodic Table of the Elements Intro Click on for intro SMB, Periodic Table Notes 2011 10
II. ORGANIZATION OF THE ELEMENTS A. Arrangement of Table 1. Horizontal rows q. Called PERIODS q. All elements in the same period have the same number of ENERGY LEVELS in their atomic structure Click on for intro SMB, Periodic Table Notes 2011 11

2. Vertical Columns a) Called GROUPS OR FAMILIES b) All elements in the same group have the same number of VALENCE ELECTRONS, therefore lose or gain the SAME number of electrons, form similar CHEMICAL FORMULAS and have similar CHEMICAL PROPERTIES ex. XCl 2 Group 2: y. Remember: When +2 Cl -1 = Be. Cl Be writing formulas, use 2 the criss-cross rule Mg +2 Cl -1 = Mg. Cl 2 to cancel out oxidation states SMB, Periodic Table Notes 2011 12
III. Comparing Metals, Nonmetals & Metalloids Elements on the Periodic Table are divided into three subgroups called METALS, NONMETALS and METALLOIDS (semimetals). Increase nonmetallic properties Decrease metallic properties SMB, Periodic Table Notes 2011 13

METALS: located on the LEFT SIDE of the periodic table (except H); MORE THAN 2/3 of all elements 1. Chemical properties q tend to LOSE ELECTRONS EASILY q have LOW IONIZATION ENERGY (energy needed to remove electrons) q Metallic character INCREASES as ionization energy decreases. q have LOW ELECTRON AFFINITY (attraction for electrons) q form POSITIVE IONS when combining with other atoms q FRANCIUM most reactive metal: See Table SMB, Periodic Table Notes 2011 14 J http: //castlelearning. com/review/reference/chem%20 table%20 j. htm

2. Metals Physical Properties q good conductors of heat and electricity q LUSTROUS - reflect light, shine when they are polished q MALLEABLE - can be rolled or hammered into sheets q DUCTILE - can be drawn into wires q are SOLIDS at room temperature except for MERCURY (liquid) SMB, Periodic Table Notes 2011 15

B. NONMETALS located on the right side of the periodic table 1. Chemical properties q tend to GAIN electrons to form NEGATIVE IONS q have high electron affinities (electronegativity) q produce COVALENT bonds by SHARING electrons with other nonmetals q FLUORINE most reactive nonmetal: see Table JSMB, Periodic Table Notes 2011 16

2. Nonmetals Physical Properties q exist as gases, molecular solids, or network solids at room temperature except BROMINE (liquid) q BRITTLE - (shatters when struck) q DULL - does not reflect light even when polished q POOR CONDUCTORS of heat and electricity q Allotropes: Different SHAPE & PROPERTIES forms from the same element. q. CARBON: coal; diamond, graphite q. OXYGEN: O 2; O 3 (OZONE) SMB, Periodic Table Notes 2011 17
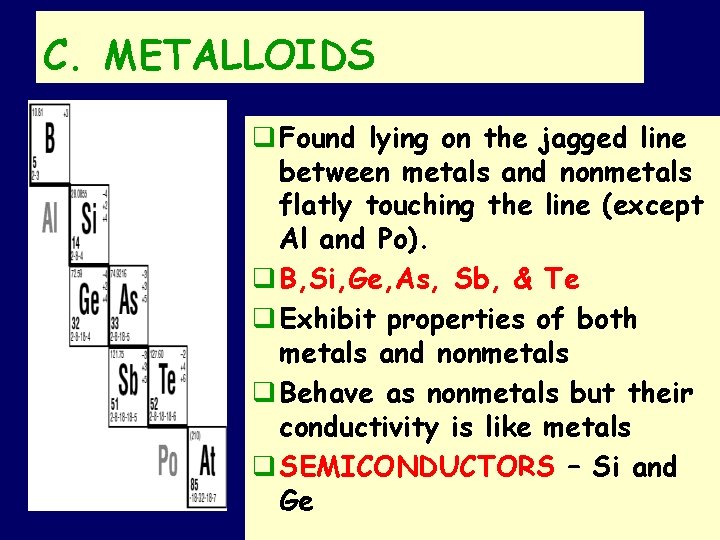
C. METALLOIDS q Found lying on the jagged line between metals and nonmetals flatly touching the line (except Al and Po). q B, Si, Ge, As, Sb, & Te q Exhibit properties of both metals and nonmetals q Behave as nonmetals but their conductivity is like metals q SEMICONDUCTORS – Si and Ge SMB, Periodic Table Notes 2011 18
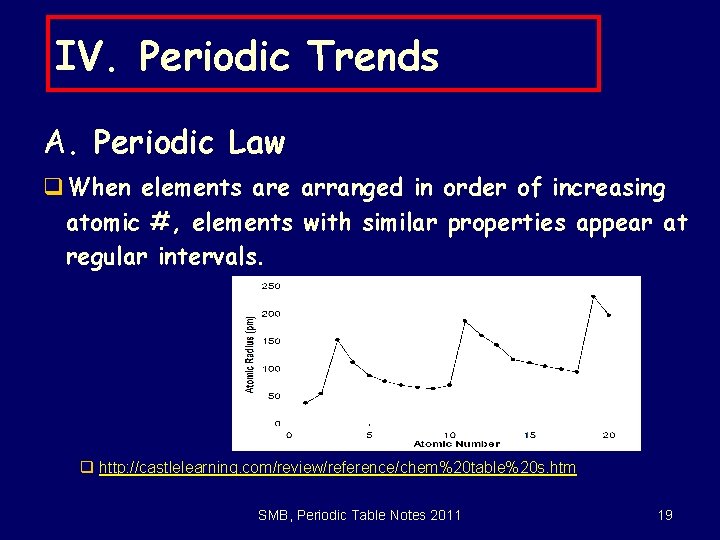
IV. Periodic Trends A. Periodic Law q When elements are arranged in order of increasing atomic #, elements with similar properties appear at regular intervals. q http: //castlelearning. com/review/reference/chem%20 table%20 s. htm SMB, Periodic Table Notes 2011 19

1) Ionization Energy z. Energy needed to remove the most loosely bound electron from a neutral gaseous atom z. X + energy X+ + e- SMB, Periodic Table Notes 2011 20
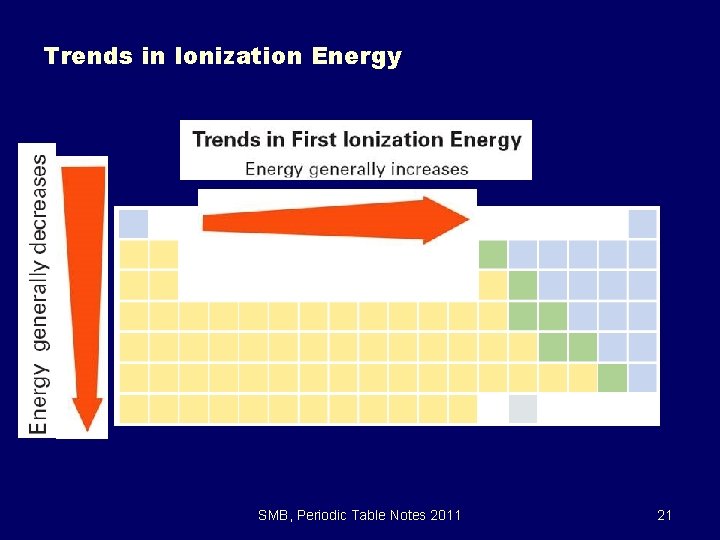
6. 3 Trends in Ionization Energy SMB, Periodic Table Notes 2011 21

Trends in Ionization Energy z. IE increases as you move across a period z. Why? z. The nuclear charge (atomic #) is increasing therefore greater attraction of the nucleus for electrons hence harder to remove an electron SMB, Periodic Table Notes 2011 22

Trends in Ionization Energy z. IE decreases as you move down a group z. Why? z. Atom size increases making the outermost electron farther away from the nucleus therefore making it easier to remove z. Shielding increases SMB, Periodic Table Notes 2011 23

Ionization Energy cont. q. Why opposite of atomic radius? üIn small atoms, e- are close to the nucleus where the attraction is stronger q. Why small jumps within each group? üStable e- configurations don’t want to lose e. SMB, Periodic Table Notes 2011 24
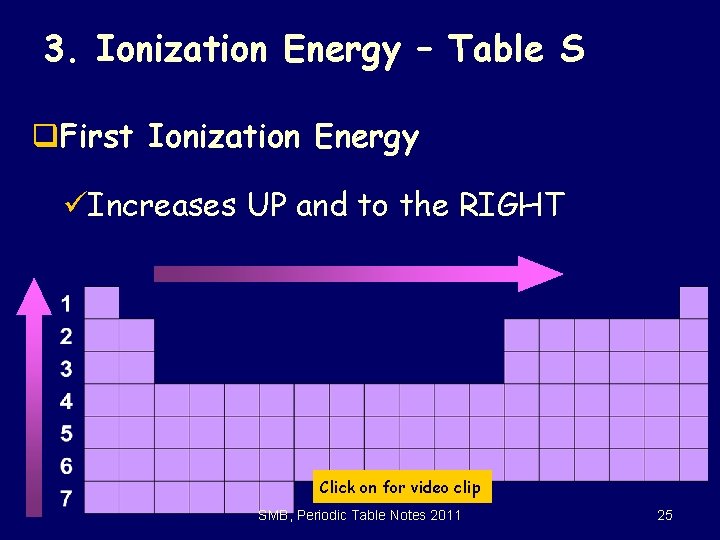
3. Ionization Energy – Table S q. First Ionization Energy üIncreases UP and to the RIGHT Click on for video clip SMB, Periodic Table Notes 2011 25

Ionization Energy cont. q. Successive Ionization Energies üLarge jump in I. E. occurs when a CORE e- is removed. § Mg Core e- 1 st I. E. 736 k. J 2 nd I. E. 1, 445 k. J 3 rd I. E. 7, 730 k. J SMB, Periodic Table Notes 2011 26
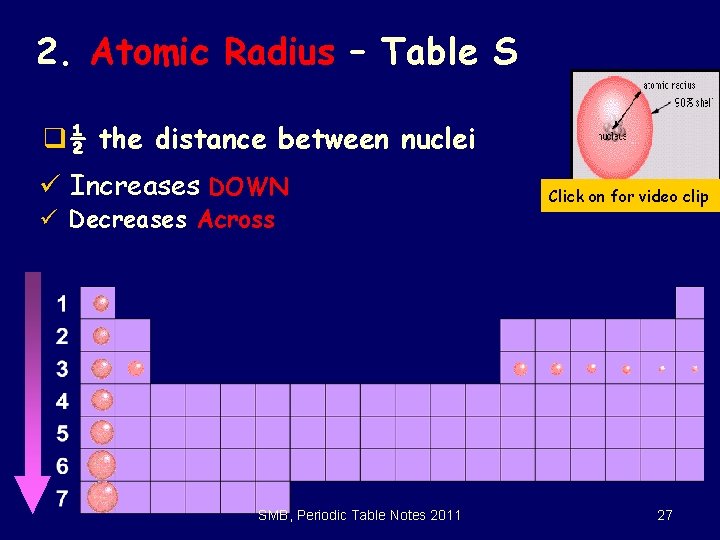
2. Atomic Radius – Table S q½ the distance between nuclei ü Increases DOWN ü Decreases Across SMB, Periodic Table Notes 2011 © 1998 LOGAL Click on for video clip 27

Atomic Radius cont. q. Why is it smaller to the right? üIncreased nuclear charge(# p+) without additional shielding pulls e- in tighter q. Why is it larger going down? üHigher energy levels have larger orbitals üShielding - core e- block the attraction between the nucleus and the valence e. SMB, Periodic Table Notes 2011 28
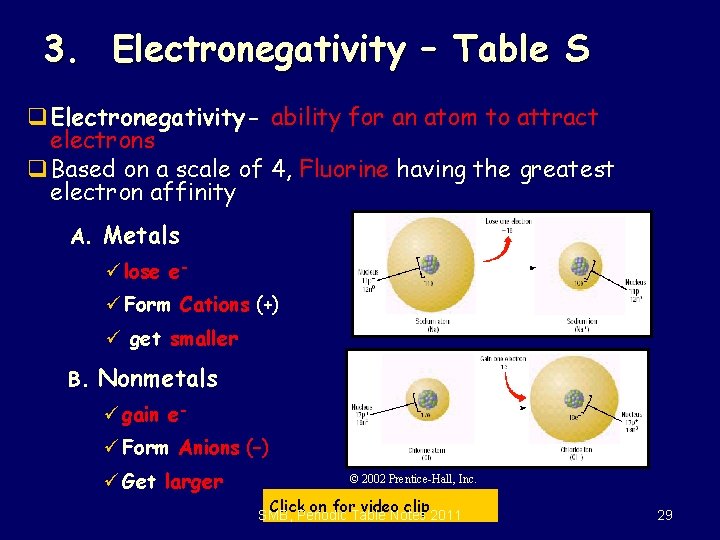
3. Electronegativity – Table S q Electronegativity- ability for an atom to attract electrons q Based on a scale of 4, Fluorine having the greatest electron affinity A. Metals ü lose eü Form Cations (+) ü get smaller B. Nonmetals ü gain eü Form Anions (–) ü Get larger © 2002 Prentice-Hall, Inc. Click on for video clip SMB, Periodic Table Notes 2011 29
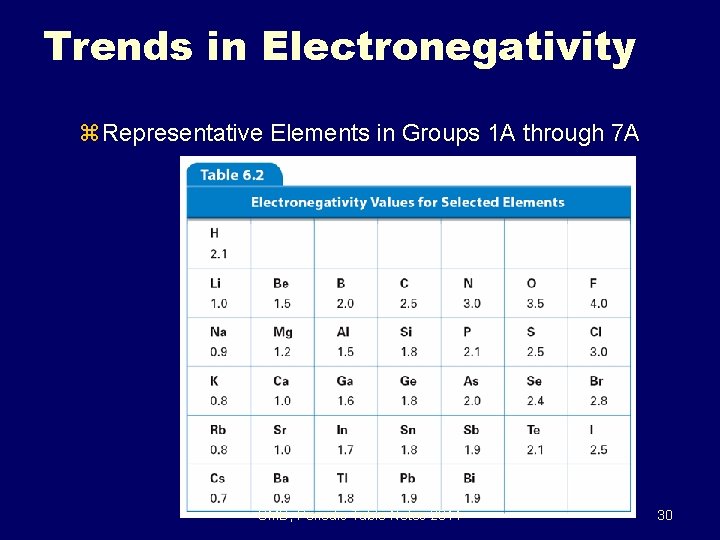
6. 3 Trends in Electronegativity z Representative Elements in Groups 1 A through 7 A SMB, Periodic Table Notes 2011 30
4. Melting/Boiling Point – Table S q. Melting/Boiling Point üHighest in the middle of a period. SMB, Periodic Table Notes 2011 31

Periodic Trends Summary (use reference Table S for data comparison) Trend Across a period Up a group Ionization energy increases Electronegativity increases Atomic radii decreases Metallic properties decreases SMB, Periodic Table Notes 2011 y. Click on for video clip 32
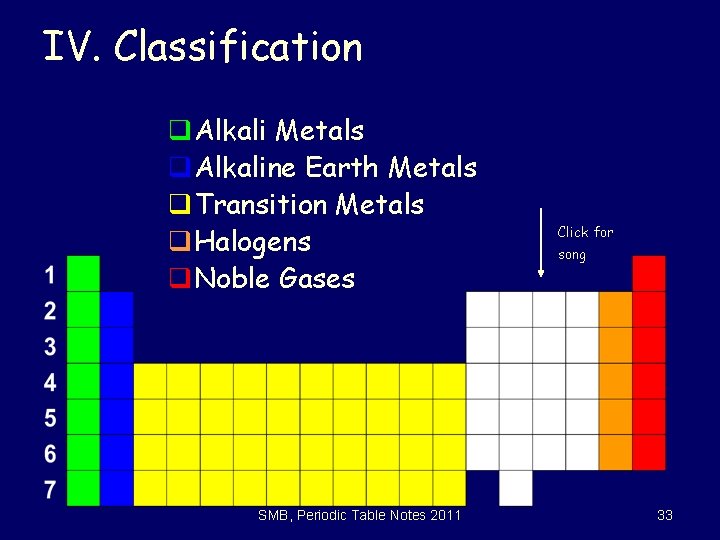
IV. Classification q. Alkali Metals q. Alkaline Earth Metals q. Transition Metals q. Halogens q. Noble Gases SMB, Periodic Table Notes 2011 Click for song 33

Group 1: Alkali Metals qextremely reactive (not found free in nature) q form stable ionic compounds qreact with water to form a base qreact with air to form oxides qreact with acids to form salts Click on for video clip SMB, Periodic Table Notes 2011 34

Group 2: Alkaline Earth Metals qreactive (not found free in nature) form stable ionic compounds qreact with water to form a base qreact with air to form oxides q react with acids to form salts Click on for video clip SMB, Periodic Table Notes 2011 35

Groups 3 -11: Transition Metals qmultiple positive oxidation states q. Lose electrons from two outermost energy levels q. Ions form colored solutions SMB, Periodic Table Notes 2011 36

Group 15 – unique features q. Members range from typical nonmetals (nitrogen and phosphorus) through metalloids (arsenic and antimony) to metals (bismuth) q Nitrogen q. Forms stable diatomic molecules with a triple bond q. Component of protein q. Forms some unstable compounds that are used as explosives q Phosphorus q. Component of nucleic acids (DNA, RNA) q. More reactive than nitrogen at room temperature SMB, Periodic Table Notes 2011 37

Group 16 – unique Features q. Members range from typical nonmetals (oxygen and sulfur) through metalloids (selenium and tellurium) to metals (polonium) q. Solids except oxygen q Oxygen can exist as O 2 and O 3 (it is an allotrope) q. Polonium is radioactive SMB, Periodic Table Notes 2011 38

Group 17: Halogens q very reactive nonmetals - high electronegativity q not found free in nature q form diatomic molecules when free q react with metals to form salts (halides) q Found in all three phases (s, l, g) due to differences in Van der Waals forces (these are weak) SMB, Periodic Table Notes 2011 39

Group 18: Noble Gases q. Have complete outer shells q. Almost inert (not reactive); stable q. Krypton, xenon, and radon form compounds with oxygen and fluorine q. Referred to as monatomic gases SMB, Periodic Table Notes 2011 40

TODAY……. . 1. 1. Assemble into your groups according to Take your notes and 5 questions with you. Using your notes, discuss & complete the Teachback WS questions pertaining to your group. § Use the post-it notes for any questions you may have THAT MAY NEED ANSWERING SMB, Periodic Table Notes 2011 41

3. BEGIN your teachback, this NOT a silent lesson – use your low voices to teach the other members. Ask your questions to ascertain clarity. At the end of this session, everyone in the group should have the Teachback WS completed. § Use the post-it notes for any questions you may have THAT NEED ANSWERING. . ØOnce finished, begin RB questions 46 -90. Cooperation and diligence is necessary…. . I will be watching and listening. SMB, Periodic Table Notes 2011 42
TEACHBACK PROJECT REVIEW q. Alkali Metals q. Alkaline Earth Metals q. Transition Metals q. Halogens q. Noble Gases SMB, Periodic Table Notes 2011 Click for song 43

GROUP 1 1. The name of this group is ALKALI METALS 2. Does this group contain metals or nonmetals? METALS 3. Alkali metals (lose or gain)l LOSE electrons becoming (positive or negative) ions? POSITIVE 4. What happens to the reactivity of the elements in this group as the atomic number increases. (increases or decreases) INCREASES 4. Are they (more or less) MORE reactive than all of the elements in Group 2 and why? THEY HAVE LOW IONIZATION ENERGIES SMB, Periodic Table Notes 2011 44

Group 1 continued: 5. Can these compounds be found in nature in the elemental or combined state? COMBINED STATE IN THE FORM OF A SALT 6. What type of compounds do they normally form (ionic or covalent)? IONIC (M + NM) 7. If element Y represents an alkali metal, what is it’s general formula for the reaction with a: Chloride: YCl Oxide: Y 2 O 8. What is the most reactive metal in this group? SMB, Periodic Table Notes 2011 45 FRANCIUM OR CESIUM

GROUP 2 1. The name of this group is ALKALINE EARTH METALS. 2. Does this group contain metals or nonmetals? METALS 3. They (lose or gain) LOSE electrons & form (positive or negative) ions POSITIVE ? 4. Describe the reactivity of the elements in this group as the atomic number increases (increases or decreases) INCREASES 5. Are they (more or less) LESS reactive than all of the elements in Group 1 and why? _____________________ SMB, Periodic Table Notes 2011 46

Group 2 continued: 5. Can these compounds be found in nature in the elemental or combined state? COMBINED STATE IN THE FORM OF A SALT 6. What type of compounds do they normally form (ionic or covalent)? IONIC (M + NM) 7. If element Z represents an alkaline earth metal, what is it’s general formula for the reaction with a: Chloride ZCl 2 SMB, Oxide: Periodic Table ZO Notes 2011 47

GROUP 15 1. What is this group referred to as? NITROGEN GROUP 2. Name the diatomic element in this group? NITROGEN 3. Classify each element in this group as a metal, non-metal or semi-metal (metalloid). metals: BISMUTH nonmetals: NITROGEN, PHOSPHORUS semi-metal: ARSENIC. ANTIMONY 4. What happens to the reactivity of a non-metal as the atomic number increases (increases or decrease) DECREASES. 5. Which is the most reactive non-metal in this group? PHOSPHORUS 6. Is nitrogen a (diatomic or monatomic) DIATOMIC molecule & what type of bond is found in nitrogen? TRIPLE COVALENT SMB, Periodic Table Notes 2011 48

GROUP 16 1. Classify each element in this group as a metal, non-metal or semi-metals: POLONIUM nonmetals: OXYGEN, SULFUR, SELENIUM semi-metal: TELLERIUM 2. Name the diatomic element in this group. OXYGEN 3. Define an allotrope? DIFFERENT FORMS OF AN ELEMENT IN THE SAME PHASE WITH HAVING DIFF CHEM & PHYSICAL PROPERTIES 4. Which element(s) in this group is an allotrope? PHOSPHORUS, SULFUR, OXYGEN (O 2, O 3) 5. What type of element is Polonium? RADIOACTIVE METAL SMB, Periodic Table Notes 2011 49

TRANSITION ELEMENTS (groups 3 B-12) 1. Which element is a liquid at room temperature? MERCURY (Hg) 2. What are the four main characteristic chemical properties of transition elements? § MULTIPLE POSITIVE OXIDATION STATES § IONS FORM COLORED SOLUTIONS § LOSE ELECTRONS FROM TWO OUTERMOST ENERGY LEVELS § UNFILLED D ORBITALS SMB, Periodic Table Notes 2011 50

GROUP 17 1. The name of this group is HALOGENS 2. Classify the elements in this group: NONMETALS 3. Halogens (lose or gain) GAIN electrons becoming (positive or negative) ions? NEGATIVE 4. Why is astatine not included much in these discussions? NOT ENOUGH AVAILABLE TO STUDY 5. What would the general formula of a Group 17 element (represented by X) combined with magnesium of group 2? Mg. X 2 SMB, Periodic Table Notes 2011 51

Group 17 continued: 6. What is the most reactive element in this group? FLUORINE 7. Can these compounds be found in nature in the elemental or combined state? COMBINED STATE AS SALTS 8. What type of salts do these elements form? HALIDES 9. For each state of matter, list the element(s) in this group. solid: IODINE liquid: BROMINE gas: CHLORINE, FLUORINE 10. What type of forces of attractions account for the different states of matter that exist and the high MP’s and BP’s as you go down the group? VAN DER WAALS FORCES (weak forces that get stronger as you go down the group ) SMB, Periodic Table Notes 2011 52

GROUP 18 z The name of this group is called NOBLE GASES. z What type of molecules do these gases form? (monatomic or diatomic) MONATOMIC z Describe the electron arrangement in the outermost energy level of all these elements. STABLE OCTECT – INERT GAS STRUCTURE z Which element has only two electrons? HELIUM z Describe the reactivity of the elements in this group. THEY ARE UNREACTIVE (Kr and Xe can be forced to react with F in lab settings) SMB, Periodic Table Notes 2011 53
- Slides: 53