The Periodic Table The periodic table is organized

The Periodic Table

The periodic table is organized like a big grid. Each element is placed in a specific location because of its atomic structure. As with any grid, the periodic table has rows (left to right) and columns (up and down). Each row and column has specific characteristics.

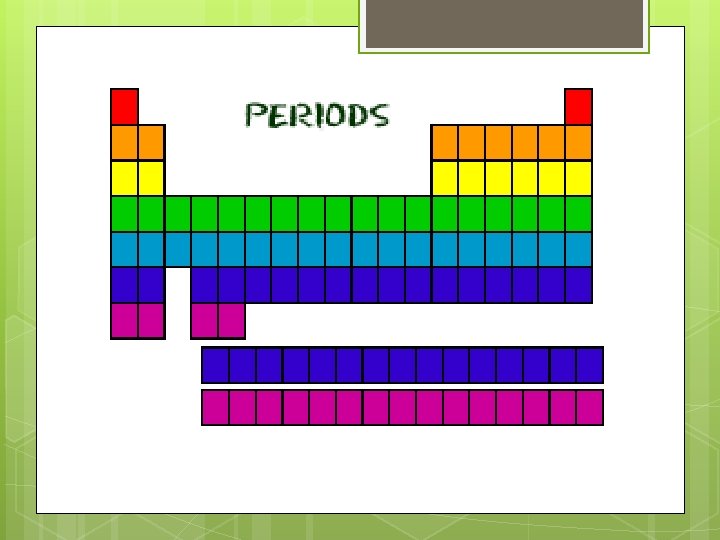
Periods (the rows) All of the elements in a period have the same number of atomic orbitals. Atomic orbitals are regions of space around the nucleus of an atom where an electron is likely to be found. Atomic orbitals allow atoms to make covalent bonds. For example, every element in the top row (the first period) has one orbital for its electrons. All of the elements in the second row (the second period) have two orbitals for their electrons. As you move down the table, every row adds an orbital. At this time, there is a maximum of seven electron orbitals.

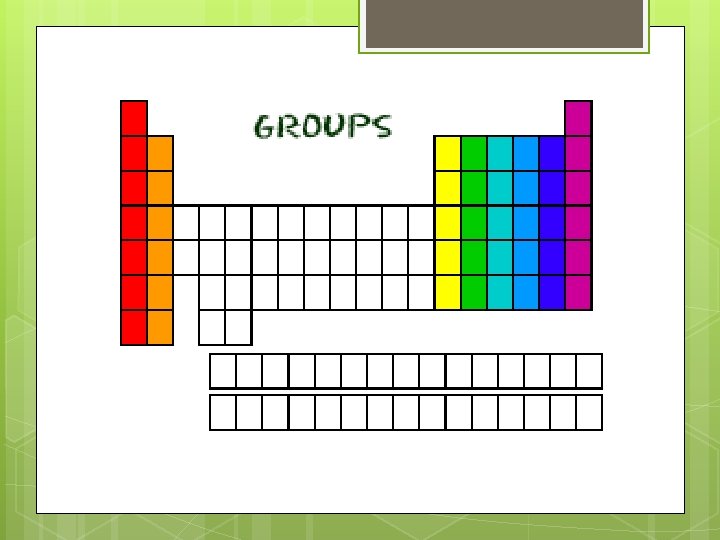
Groups (the columns) Each column is called a group. The elements in each group have the same number of electrons in the outer orbital. Those outer electrons are also called valence electrons. They are the electrons involved in chemical bonds with other elements. Every element in the first column (group one) has one electron in its outer shell. Every element in the second column (group two) has two electrons in the outer shell. As you keep counting the columns, you'll know how many electrons are in the outer shell.

Families There also other, less specific, groups of elements all over the table. Scientists group these groups, called families of elements, by their chemical properties. Each family reacts in a different way with the outside world. The columns of the periodic table are often used to define families.

Examples of Families - Alkali Metals - Alkaline Earth Metals - Transition Metals - Halogen Gases - Noble Gases
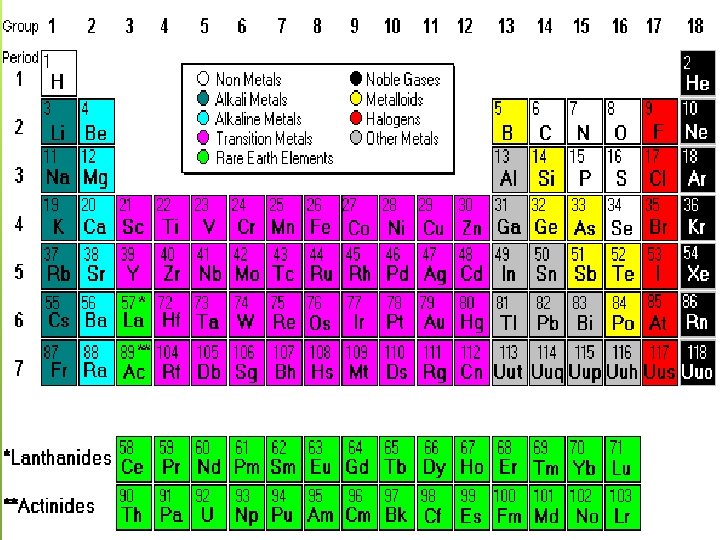

The Halogen Group In the second column from the right side of the periodic table, you will find Group Seventeen. This column is the home of the halogen family of elements. The elements included are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). Contain 7 electrons in their outer shell. They have the trait of combining with many different elements. They are very reactive. You will often find them bonding with metals and elements from Group One of the periodic table.

The Noble gases The noble gases are another family of elements, and all of them are located in the far right column of the periodic table (Group 18) The list includes helium, neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and radon (Rn). All of the noble gases (except Helium) have full outer shells with eight electrons. At the top of the noble gases is little helium (He), with a shell that is full with only two electrons. The fact that their outer shells are full means they are quite stable and don't need to react with other elements. In fact, they rarely combine with other elements.

Metals Most metals occur as solids at room temperature. Metals have metallic luster which makes them shiny. Metals are ductile, so they can be drawn into wires. Metals are malleable, so they can be hammered or rolled into sheets. Metals are good conductors of heat and electricity. Metals tend to give up electrons easily because they are not strongly held by the protons in the nucleus.

Nonmetals Most nonmetals are gases at room temperature or brittle solids. Solid nonmetals are not malleable or ductile, instead they are brittle and powdery They are poor conductors of heat and electricity because the electrons in nonmetals aren’t free to move as they do in metals. When bonded with other nonmetals, atoms of nonmetals usually share electrons to form covalent bonds. Ex: CO 2 and H 2 O

Ionic Bonding When metals combine with nonmetal, the atoms of the metals tend to lose electrons to the atoms of the nonmetals. The metal atoms become positive ions. The nonmetal atoms become negative ions. Both metals and nonmetals become more chemically stable when they form ions.
- Slides: 14