The Periodic Table The Periodic Table H History
The Periodic Table

The Periodic Table H History and Development Arrangement and Organization of Elements Finding Information

History of the Table H H H What was the table like before Demetri Mendeleev ? What did he do to improve it? Has it changed since Mendeleev?

Early Study of the Elements Beguyer de Chancourtois - 1862 geologist; arranged elements by atomic weights about a cylinder Meyer 1895 plotted atomic volume against molecular weight; found 2 short periods and 2 long periods on his graph Newlands - English 1864 - Law of Octaves - every 8 th element has similar properties

Stanislao Cannizzaro H H H Discovered a new way to calculate atomic mass Scientists everywhere agreed this was a very reliable method Provided a sense of order for the elements

The Periodic Law H H The physical and chemical properties of the elements are periodic functions o their atomic numbers. OR: when elements are arranged in order of increasing atomic number, elements with similar properties appear at regular intervals

Development of Periodic Table Periodic Law 1869 allowed Mendeleev to predict properties of unknown elements

Mendeleev’s Periodic Table H arranged elements according to: – increasing atomic weights – physical properties • metal/nonmetal • melting points, boiling points, density, reactivity H Many later discoveries support Mendeleev’s table
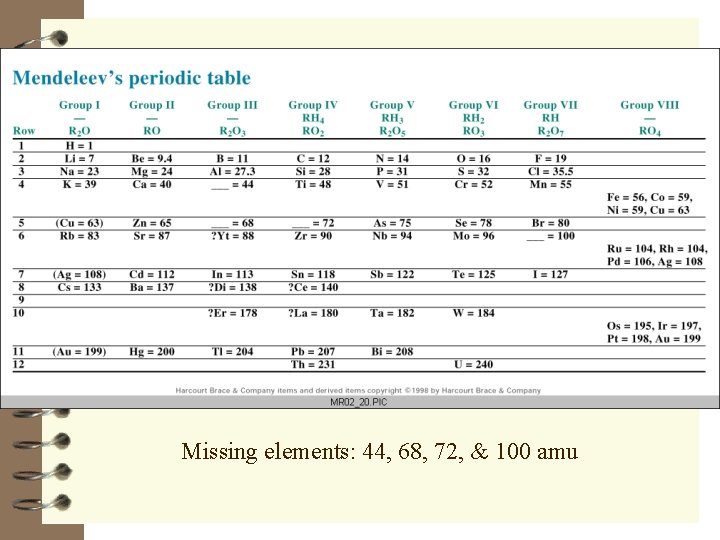
Missing elements: 44, 68, 72, & 100 amu

Mendeleev’s Table - Irregularities H H When arranged by similar properties, a few elements were out of place by atomic mass There were some empty spaces in the table

Moseley’s Contribution Moseley, Henry Gwyn Jeffreys 1887– 1915, English physicist. Studied the relations among bright-line spectra of different elements. Derived the ATOMIC NUMBERS from the frequencies of vibration of X-rays emitted by each element. Moseley concluded that the atomic number is equal to the charge on the nucleus. This work explained discrepancies in Mendeleev’s Periodic Table.

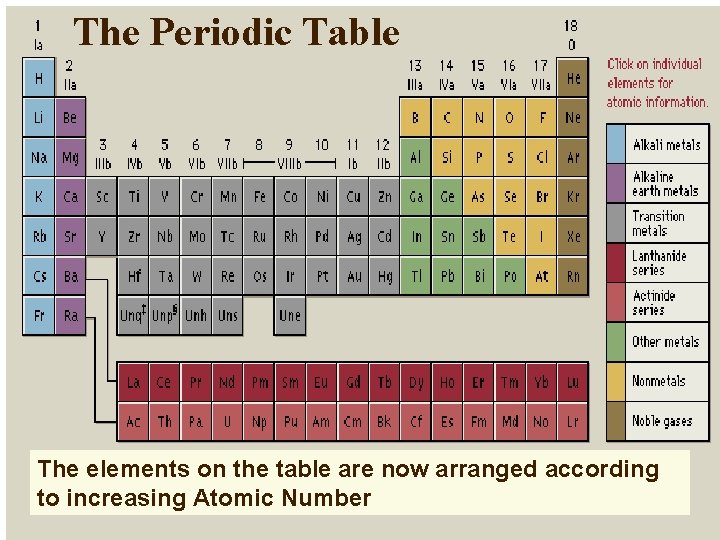
The Periodic Table The elements on the table are now arranged according to increasing Atomic Number

Organization of Periodic Table H H period group type of element physical properties – mp, bp, density, conductivity H chemical reactivity – ionization energy, electron affinity, electronegativity

Organization and Arrangement H H period - horizontal row group or family - vertical column

Types of Elements metals nonmetals metalloids - semimetals

Periodic Table of the Elements
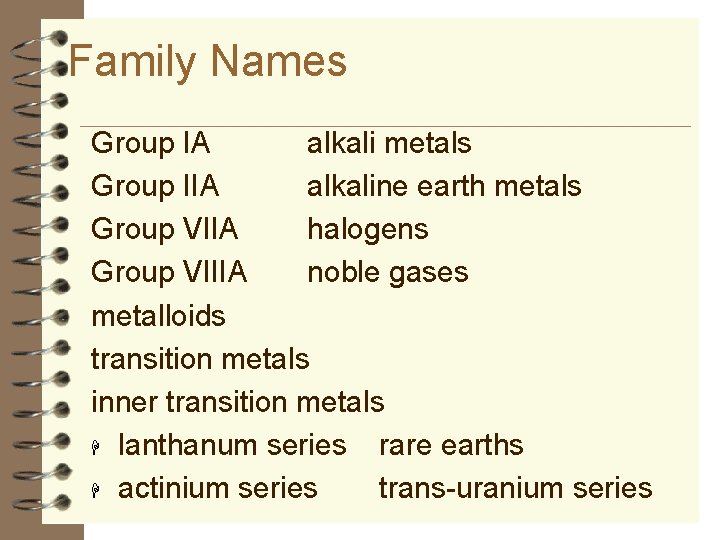
Family Names Group IA alkali metals Group IIA alkaline earth metals Group VIIA halogens Group VIIIA noble gases metalloids transition metals inner transition metals H lanthanum series rare earths H actinium series trans-uranium series
The Periodic Table Know what information you can find and where to find it

Finding Information A periodic table is always available to you. To be successful in chemistry, you must know: H the names and symbols H later many of the ions that form H how to find number of protons, neutrons, and electrons in an element H how to read the other information on the table.

Information found on the Periodic Table H H H H Chemical symbol Atomic number Atomic Mass Protons in atom Electrons in atom Neutrons in atom Metal / Nonmetal H H H H Chemical activity Oxidation number Energy levels Electron configuration Chemical Families Natural / Manmade Physical state at room temperature
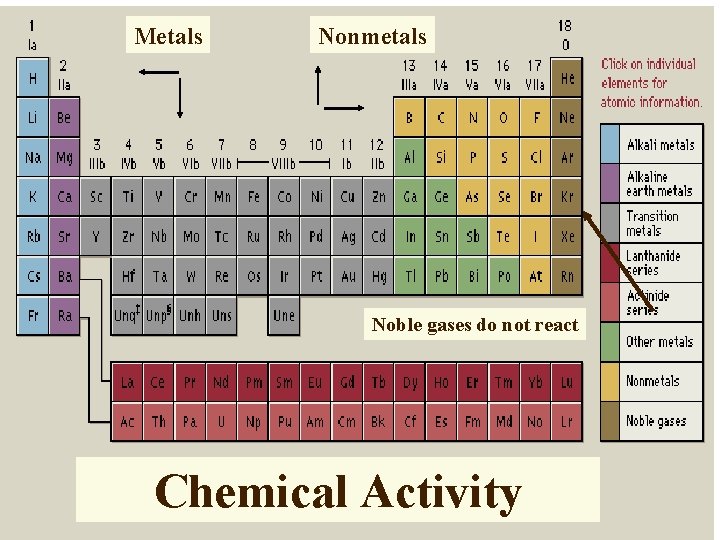
Metals Nonmetals Noble gases do not react Chemical Activity

+1 0 +2 +3 4 -3 -2 -1 M o s t a r e +1 o r +2 Oxidation Number
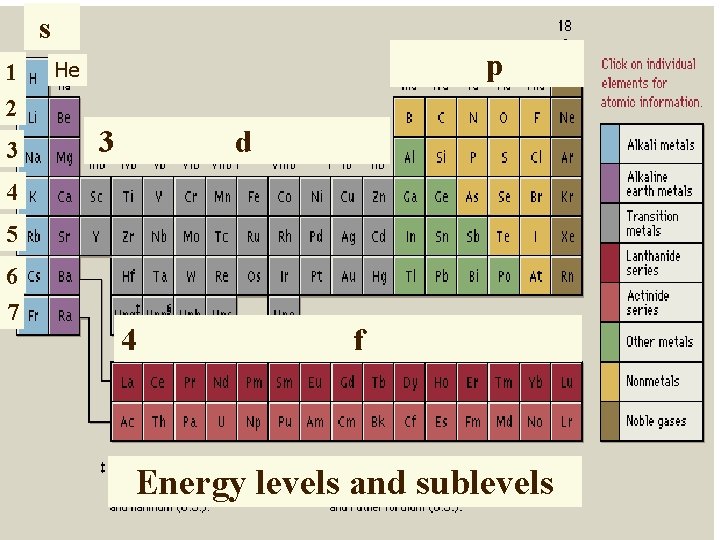
s 1 2 3 p He 3 d 4 5 6 7 4 f Energy levels and sublevels
The End
- Slides: 25