The Periodic Table The History Mendeleev Dmitri Mendeleev
The Periodic Table The History

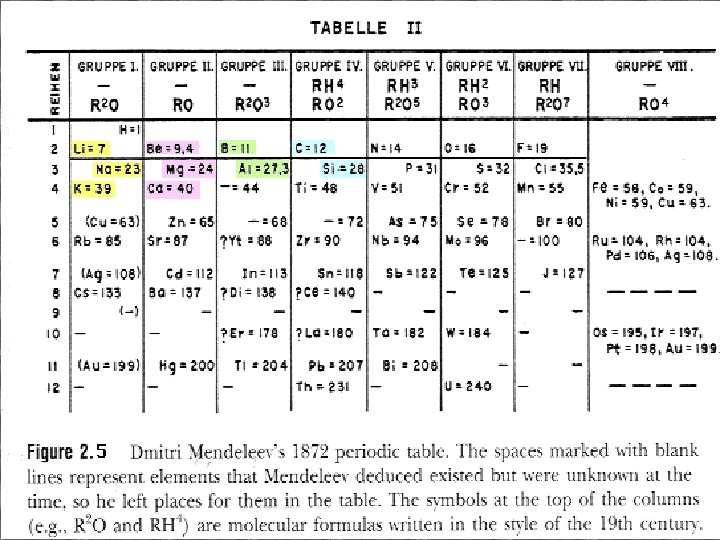
Mendeleev • Dmitri Mendeleev (1869, Russian) – Organized elements by increasing atomic mass. – Elements with similar properties were grouped together. – There were some discrepancies.

Mendeleev • Dmitri Mendeleev (1869, Russian) – Predicted properties of undiscovered elements.

Moseley • Henry Moseley (1913, British) – Organized elements by increasing atomic number. – Resolved discrepancies in Mendeleev’s arrangement.
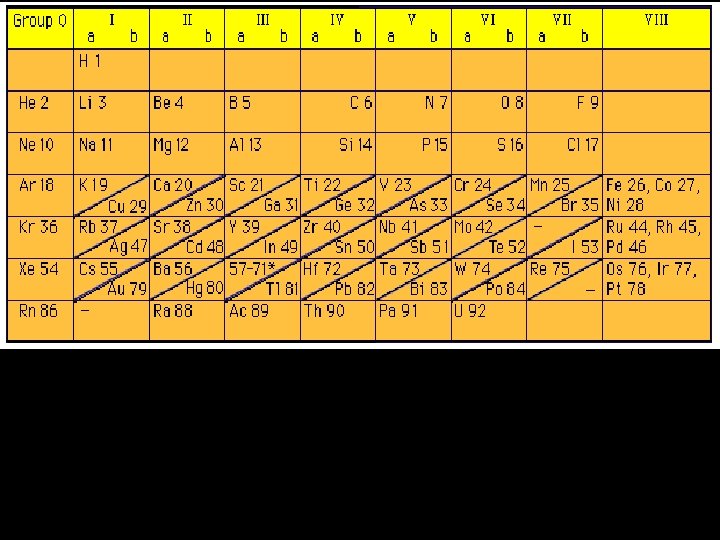

Modern Russian Table

Chinese Periodic Table
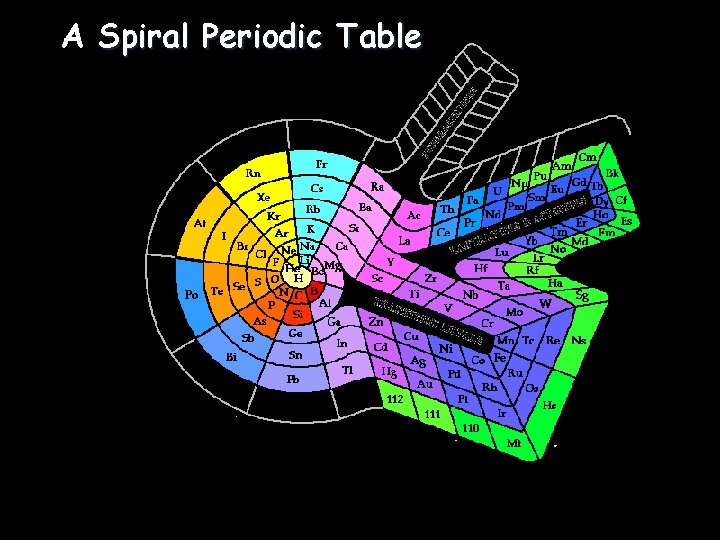
A Spiral Periodic Table
Triangular Periodic Table

Outcome C - The Periodic Table Organization of the Elements

Metallic Character • Metals • Nonmetals • Metalloids
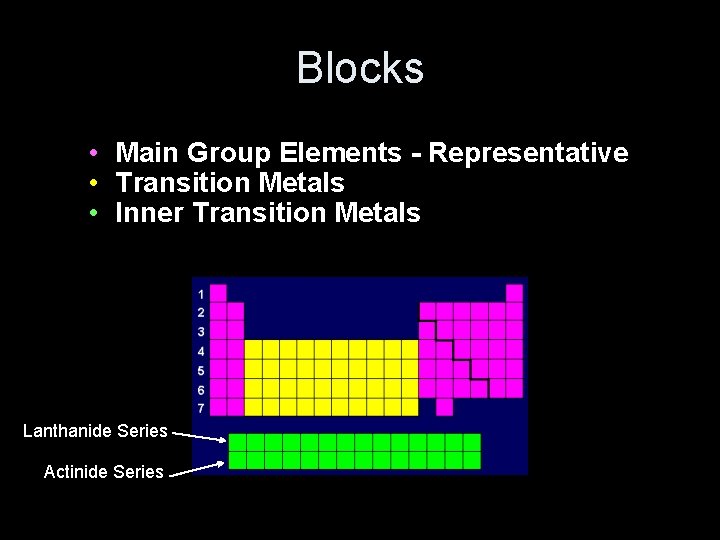
Blocks • Main Group Elements - Representative • Transition Metals • Inner Transition Metals Lanthanide Series Actinide Series
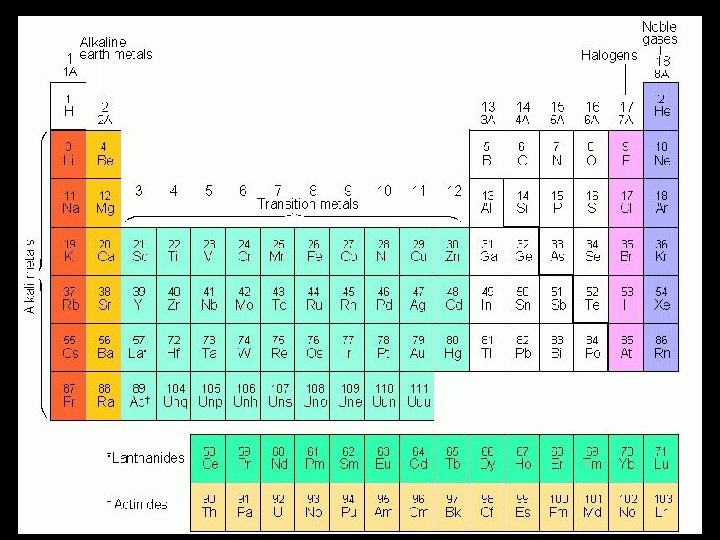
Periodic Table with Group Names
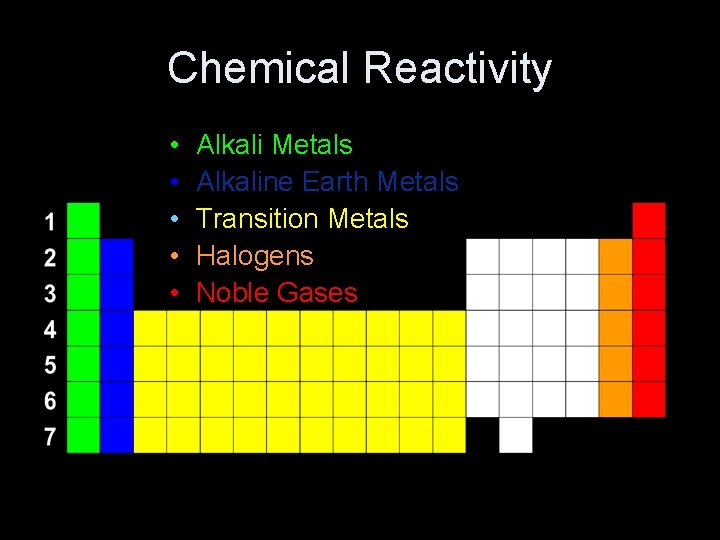
Chemical Reactivity • • • Alkali Metals Alkaline Earth Metals Transition Metals Halogens Noble Gases
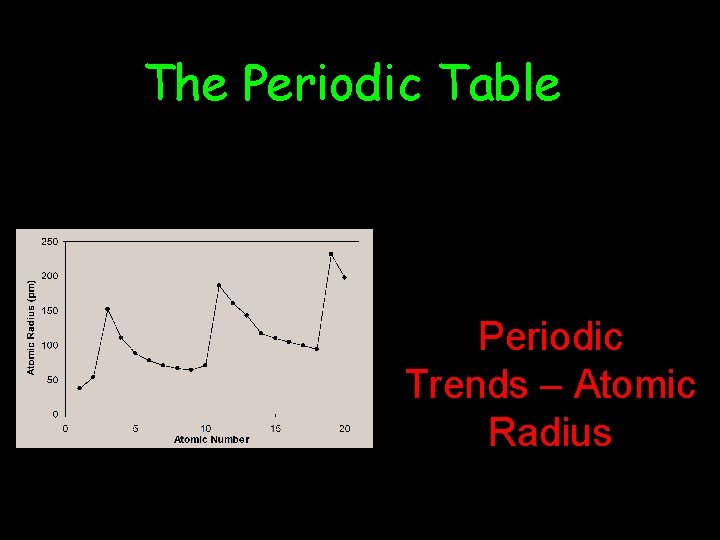
The Periodic Table Periodic Trends – Atomic Radius
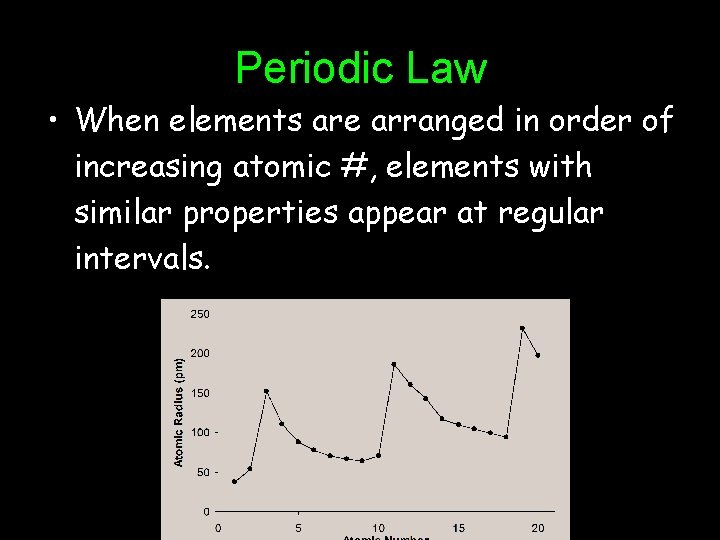
Periodic Law • When elements are arranged in order of increasing atomic #, elements with similar properties appear at regular intervals.
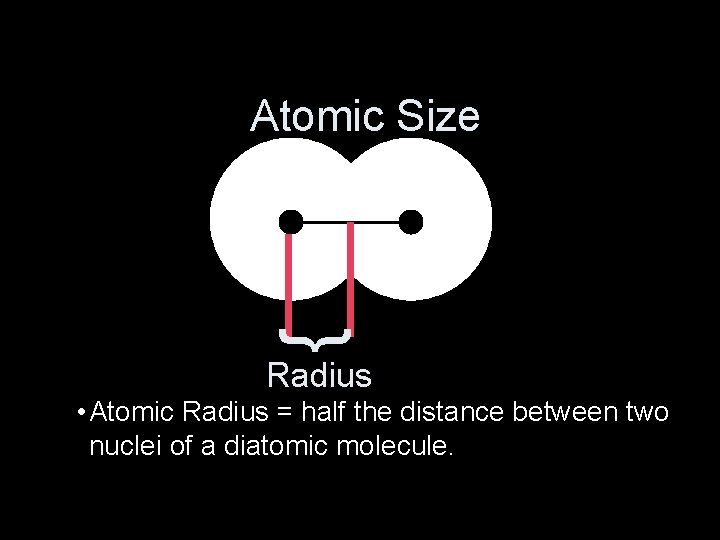
Atomic Size } Radius • Atomic Radius = half the distance between two nuclei of a diatomic molecule.

Trends in Atomic Size • • • Influenced by two factors. Energy Level Higher energy level is further away. Charge on nucleus More charge pulls electrons in closer.
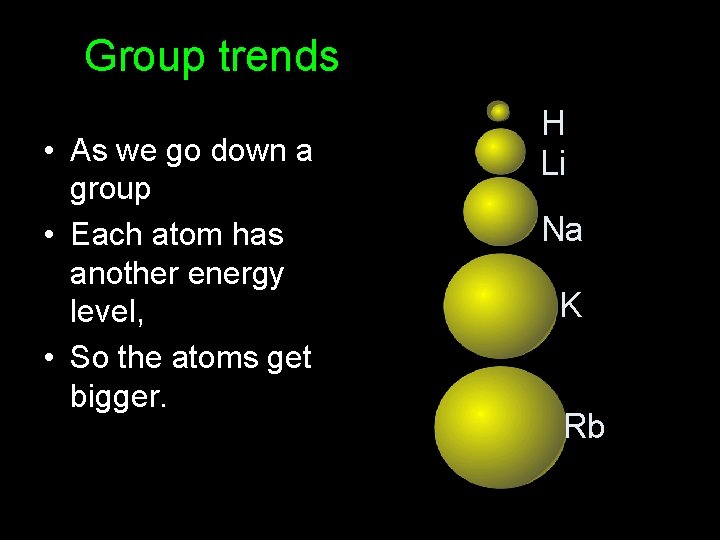
Group trends • As we go down a group • Each atom has another energy level, • So the atoms get bigger. H Li Na K Rb

Periodic Trends • As you go across a period the radius gets smaller. • Same energy level. • More nuclear charge. • Outermost electrons are closer. Na Mg Al Si P S Cl Ar
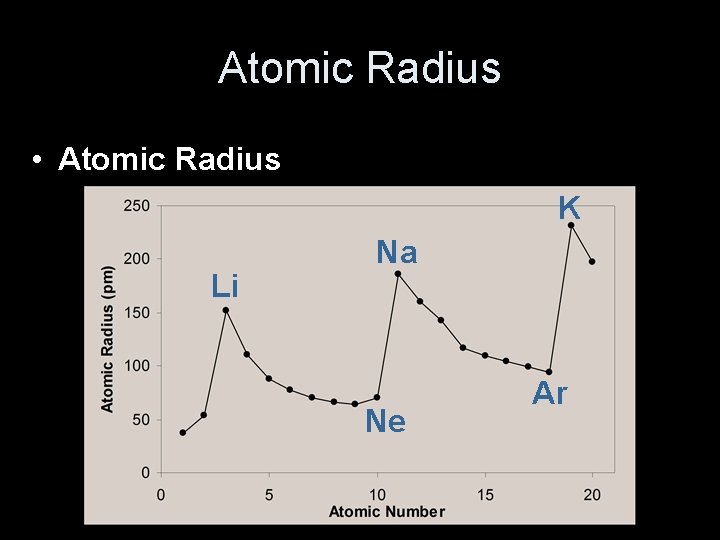
Atomic Radius • Atomic Radius K Li Na Ne Ar
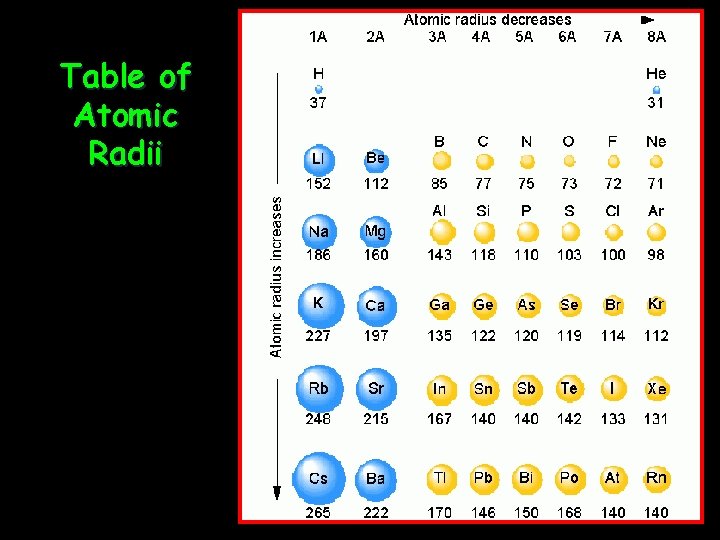
Table of Atomic Radii
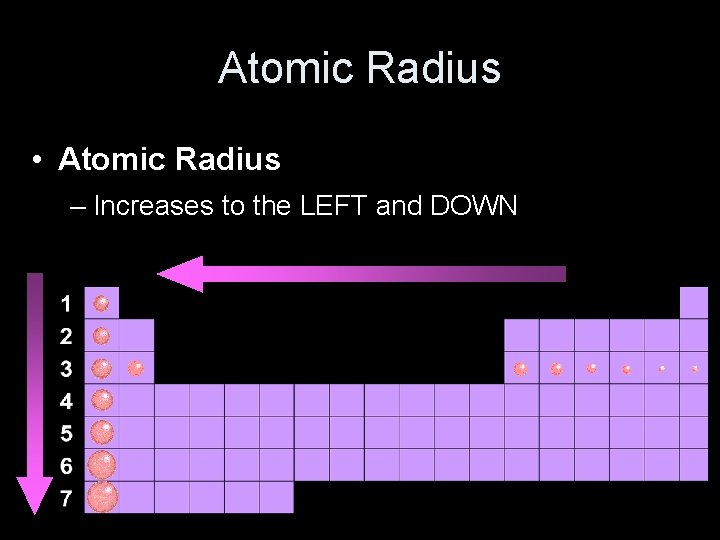
Atomic Radius • Atomic Radius – Increases to the LEFT and DOWN

Atomic Radius • Why larger going down? – Higher energy levels have larger orbitals – Shielding - core e- block the attraction between the nucleus and the valence e • Why smaller to the right? – Increased nuclear charge without additional shielding pulls e- in tighter
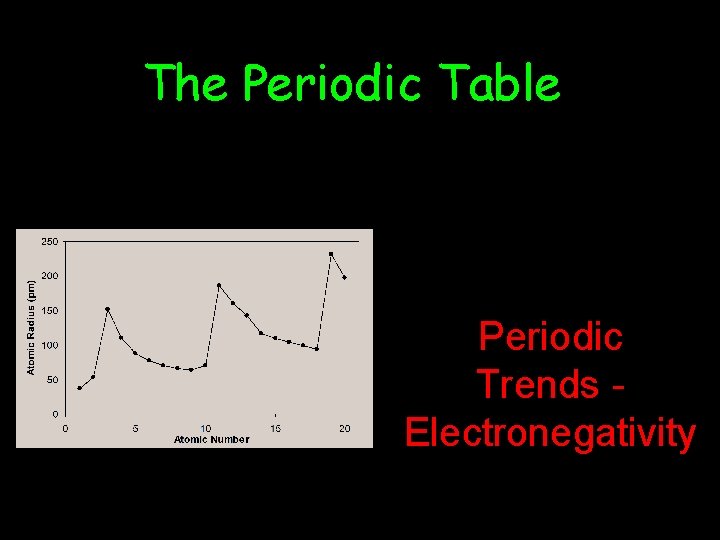
The Periodic Table Periodic Trends Electronegativity

Electronegativity • The tendency for an atom to attract electrons to itself when it is chemically combined with another element. • How fair it shares. • Big electronegativity means it pulls the electron toward it.

• Electronegativity – Attraction an atom has for a shared pair of electrons. – higher e-neg atom – lower e-neg atom +
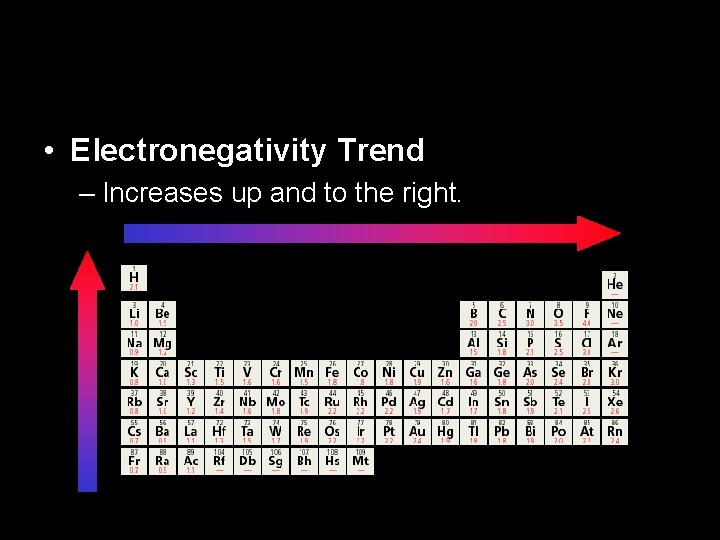
• Electronegativity Trend – Increases up and to the right.

Group Trend • The further down a group the farther the electron is away and the more electrons an atom has. • More willing to share. • Low electronegativity.

Periodic Trend • Metals are at the left end (larger atoms) • Electrons are farther from nucleus • Low electronegativity • At the right end are the nonmetals (smaller atoms) • Electrons are closer to nucleus. • High electronegativity.
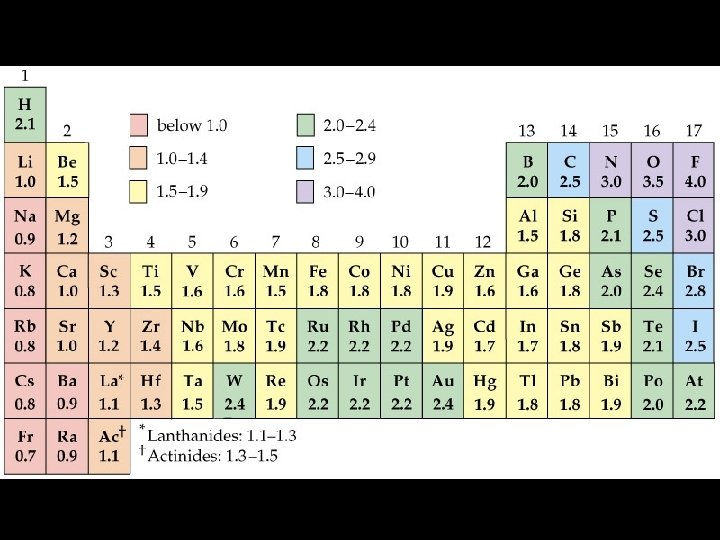
Periodic Table of Electronegativities

IONIZATION ENERGY • The amount of energy required to completely remove an electron from an atom. • Removing one electron makes a +1 ion. • The energy required to remove one electron is called the first ionization energy.

What determines IE • The size of the atom. • Distance of the electron from the nucleus • In larger atoms the valance electrons are farther from the nucleus
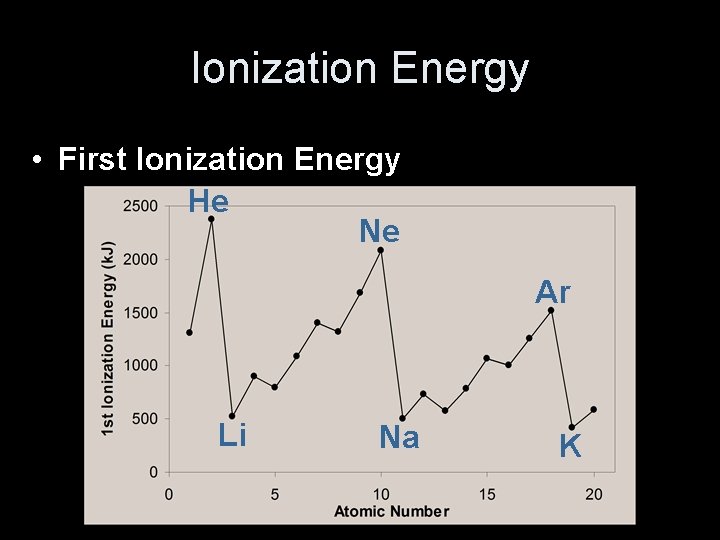
Ionization Energy • First Ionization Energy He Ne Ar Li Na K
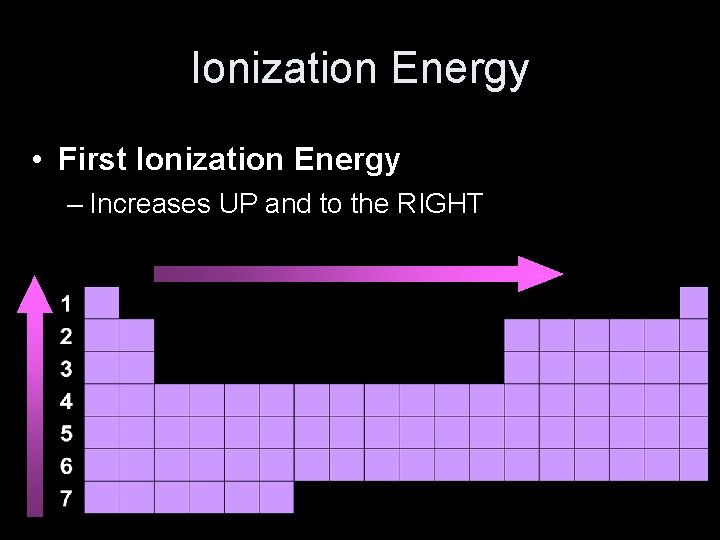
Ionization Energy • First Ionization Energy – Increases UP and to the RIGHT

Ionization Energy • Why opposite of atomic radius? – In small atoms, e- are close to the nucleus where the attraction is stronger • Why small jumps within each group? – Stable e- configurations don’t want to lose e- © 1998 LOGAL

Ionization Energy • The second ionization energy is the energy required to remove the second electron. • Always greater than first IE. • The third IE is the energy required to remove a third electron. • Greater than 1 st of 2 nd IE.

Ionization Energy • Successive Ionization Energies –Large jump in I. E. occurs when a CORE e- is removed. – Mg Core e- 1 st I. E. 736 k. J 2 nd I. E. 1, 445 k. J 3 rd I. E. 7, 730 k. J

Ionization Energy • Successive Ionization Energies – Large jump in I. E. occurs when a CORE e- is removed. – Al Core e- 1 st I. E. 577 k. J 2 nd I. E. 1, 815 k. J 3 rd I. E. 2, 740 k. J 4 th I. E. 11, 600 k. J
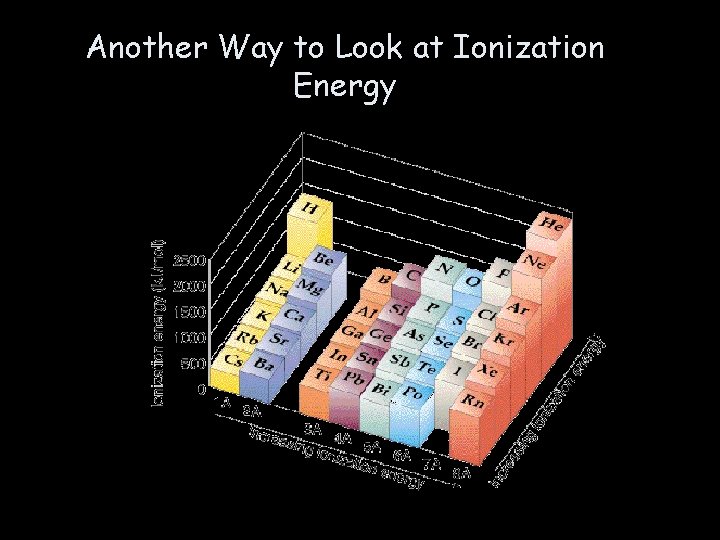
Another Way to Look at Ionization Energy
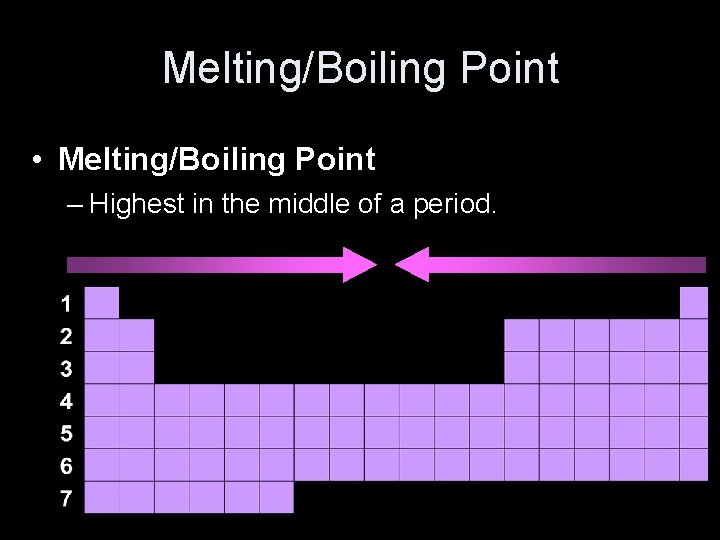
Melting/Boiling Point • Melting/Boiling Point – Highest in the middle of a period.
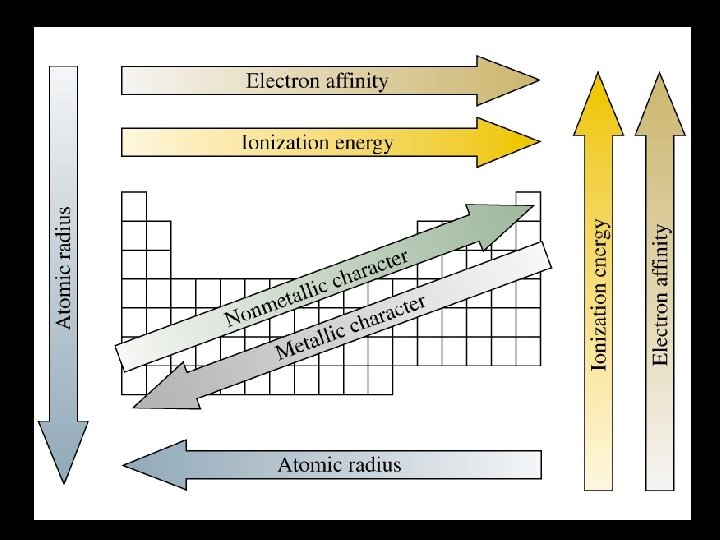
Summation of Periodic Trends
- Slides: 44