The Periodic Table Reading the Periodic Atomic Number

The Periodic Table
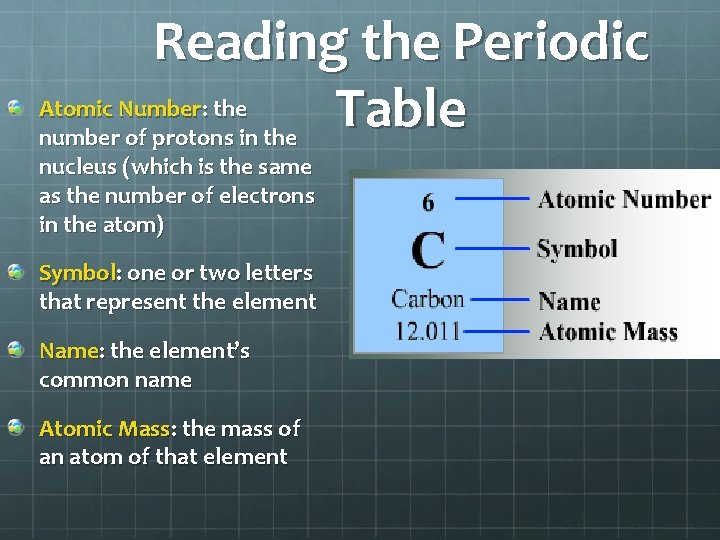
Reading the Periodic Atomic Number: the Table number of protons in the nucleus (which is the same as the number of electrons in the atom) Symbol: one or two letters that represent the element Name: the element’s common name Atomic Mass: the mass of an atom of that element

Periodic Table Grouping The periodic table is formed from 3 major groups of elements: Metals Nonmetals Metalloids or Semimetals
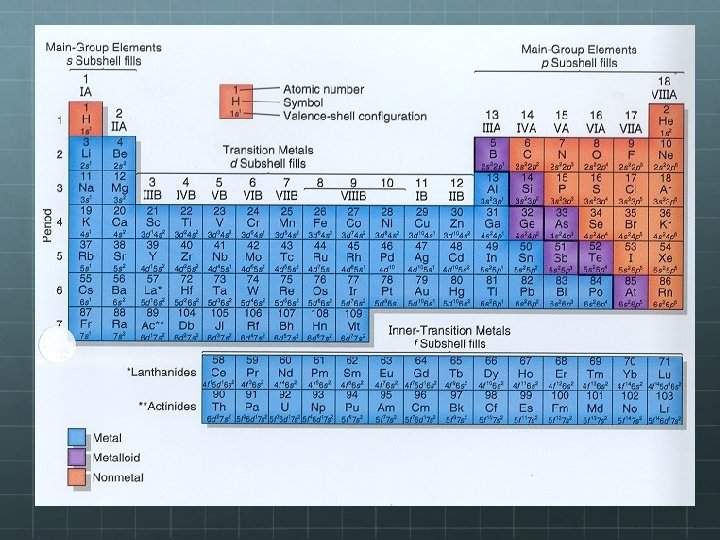

Properties of Metals are lustrous, also known as shiny, malleable, ductile, and are good conductors of heat and electricity. Mostly solids at room temperature. Can anyone think of an exception? ? ?

Properties of Nonmetals are dull and brittle. They are insulators which means they are nonconductive. Many of them are gases, with only some that are solids, and Br is a liquid.

Properties of Metalloids are also known as semi-metals Therefore, they have characteristics of both metals AND nonmetals For example, they are shiny and brittle Semiconductors

Periodic Arrangement Elements are placed in sequence of increasing atomic size. Grouped by similar properties: Groups/Families: Group 1 (Li - Fr): Alkali Metals Group 2 (Be - Ra): Alkali Earth Metals Group 18: Noble Gases

Periodic Trends The periodic table is also arranged according to trends such as: Atomic Size: all atoms have a different size Electronegativity: the ability of an atom of an element to attract electrons when the atom is in a compound Ionization Energies: the energy required to remove an electron from an atom Ionic Size: When metals and nonmetals react, metal atoms tend to lose electrons and therefore form cations. Nonmetal atoms tend to gain electrons and therefore form anions. This transfer of electrons affects the size of the ions.
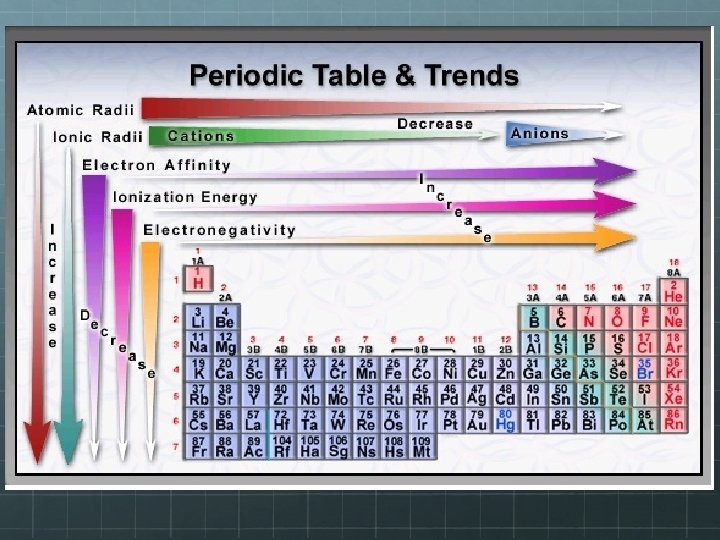

Periodic Trends Atomic Radius is an important trend and is affected by the atomic size. Radius is the distance from the center of the nucleus to the “edge” of the electron cloud

Atomic Radius

Ionization Energy The energy required to remove an electron from an atom is ionization energy. The larger the atom is, the easier it is to remove its electrons Ionization energy and atomic radius are inversely related An atom that has been “ionized” has become charged The number of protons and electrons are no longer equal

Active Chemistry Pg. 141, #2 X-axis: atomic numbers ranging 1 -36 with every other line being an increment of 1 Y-axis: ionization energies ranging 0 -130 with every line being an increment of 2 Title: Ionization Energies Legend: what colors are associated with your 1 st and 2 nd ion energies? Plot the first ionization energy data from the chart in one color, connecting them as you go along forming a line graph. Then plot the values for the second ionization energies in a different color

Active Chemistry Where are the ionization energies the largest? The smallest? What happens to the ionization energies as the atomic number increases? Describe how the 2 graphs are alike and/or different. What similarities or differences do you see?
- Slides: 16