The Periodic Table PrePeriodic Table Chemistry was a
The Periodic Table

Pre-Periodic Table Chemistry … …was a mess!!! No organization of elements. Difficult to find information. Chemistry didn’t make sense.

Dmitri Mendeleev Father of the Periodic Table WHAT HE DID… SOME PROBLEMS… Put elements in rows He left blank spaces Put elements in He broke some by increasing atomic weight columns by the way they reacted. for what he said were undiscovered elements. patterns

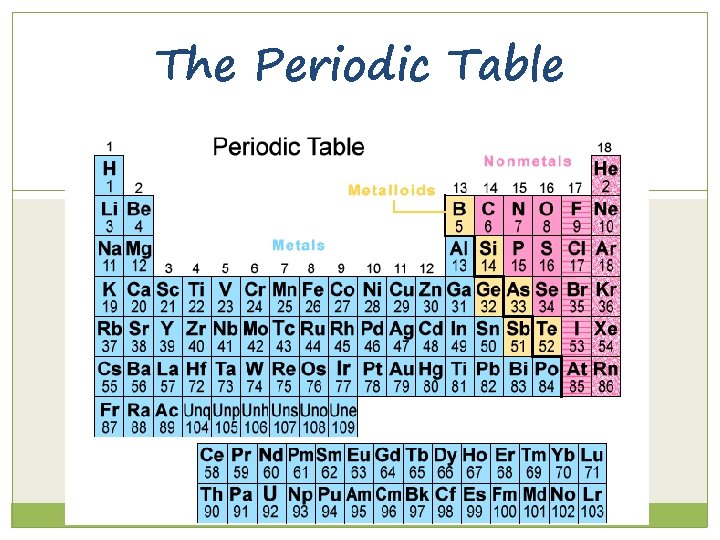
The Current Periodic Table Now the elements are put in rows by increasing atomic number The horizontal rows are called periods and are labeled from 1 to 7. The vertical columns are called groups or families are labeled from 1 to 18.

6. 1 The Periodic Law States that when elements are arranged in order of increasing atomic number, there is a periodic repetition of their physical and chemical properties. This allows us to put elements in order and make predictions about their behavior
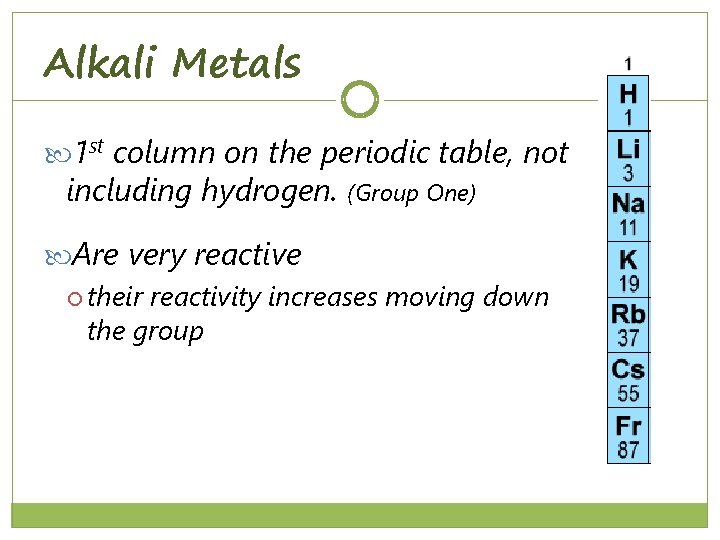
Alkali Metals 1 st column on the periodic table, not including hydrogen. (Group One) Are very reactive their reactivity increases moving down the group

Alkaline Earth Metals Second column on the periodic table (Group 2) Reactive metals Several of these elements are important mineral nutrients (such as Mg and Ca) All are solids
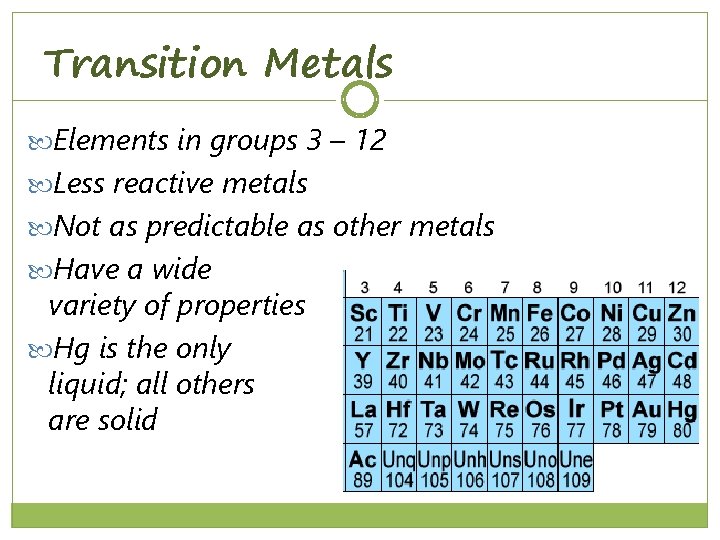
Transition Metals Elements in groups 3 – 12 Less reactive metals Not as predictable as other metals Have a wide variety of properties Hg is the only liquid; all others are solid

Halogens Elements in group 17 Very reactive nonmetals Fluorine is the most reactive this decreases as you move down the group F and Cl are gas; Br is liquid; I and At are solid

The Noble Gases Elements in group 18 Extremely low chemical reactivity or inert (very stable) Used in common items such as neon signs or balloons All are gas

Lanthanides & Actinides Lanthanides: atomic numbers 57 – 71 (top row) Actinides: atomic numbers 89 – 103 (bottom row) All solid
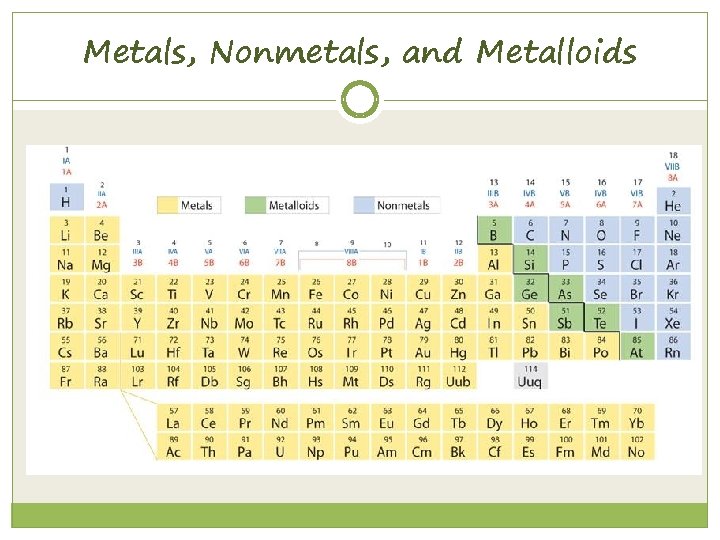
6. 1 Metals, Nonmetals, and Metalloids

Metals To the left of the staircase line Physical Properties: Ø Luster (shiny) Ø Good conductors Ø High melting point Ø Malleable Chemical Properties: Ø Easily lose electrons Ø Corrode easily (rusting or tarnishing)
Non-metals to the right of the staircase line Physical Properties: Ø Dull (not shiny) Ø Poor conductor of electricity Ø Brittle Ø Low Melting Point Chemical Properties: Ø Tend to gain electrons

Metalloids along the staircase line Solids Shiny or dull Malleable Conduct heat and electricity better than non metal but not as well as metals

Protons, Neutrons, Electrons Protons are positively Atomic Mass Number Neutrons are neutral # Protons = Atomic # charged particles in the nucleus particles also in the nucleus Electrons are negatively charged particles that surround the nucleus = # protons + # neutrons # protons = # electrons
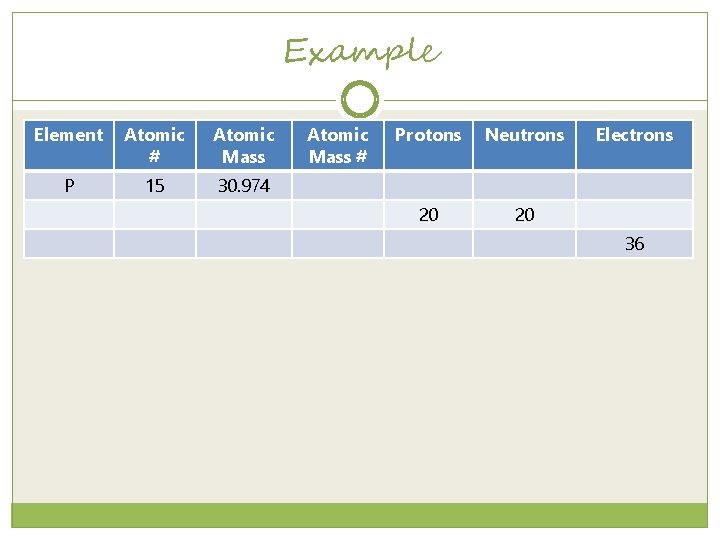
Example Element Atomic # Atomic Mass P 15 30. 974 Atomic Mass # Protons Neutrons 20 20 Electrons 36

Ions are charged particles that have either lost or gained electrons Non-metals gain electrons (- charge) Metals lose electrons (+ charge) Ex: Calcium ion: Ca 2+ (loses 2 electrons) Oxide ion: O 2 - (gains 2 electrons)
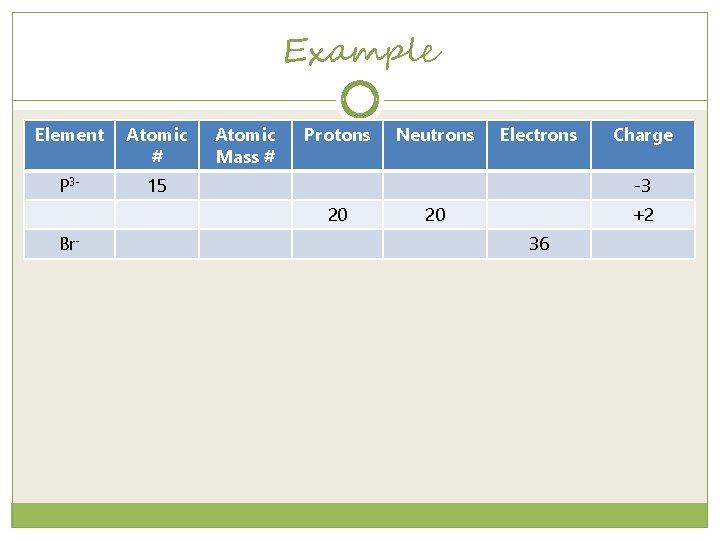
Example Element Atomic # P 3 - 15 Atomic Mass # Protons Electrons Charge -3 20 Br- Neutrons 20 +2 36
- Slides: 19