The Periodic Table Phases of Matter Solids Atoms

The Periodic Table

Phases of Matter Solids • Atoms tightly bound • Fixed volume and shape (does not conform to container) • A chemical is denoted as solid by labeling it with (s) S(s)
Phases of Matter Liquids • Atoms less tightly bound than solids • Has a definite volume, but not definite shape (assumes the shape of its container) • Denoted by (L) H 2 O (L)

Phases of Matter Gases • Free atoms • No shape, no definite volume • Can be expanded or compressed (like engine piston) • Denoted by (g) ; ex. O 2 (g)

Elemental Classifications • Elements can be metals, nonmetals, or semiconductors (intermediates between metals and nonmetals) Physical Characteristics of Metal • Solids at room temperature (except Mercury) • Malleable and ductile • Conductive of electricity and heat • Have luster and shine • Very High Melting Points

Elemental Classifications Physical Characteristics of Nonmetals • Non conductive • Most are gases, but some are solid. The solids tend to be brittle and powdery • Low melting points He (g) S(s)
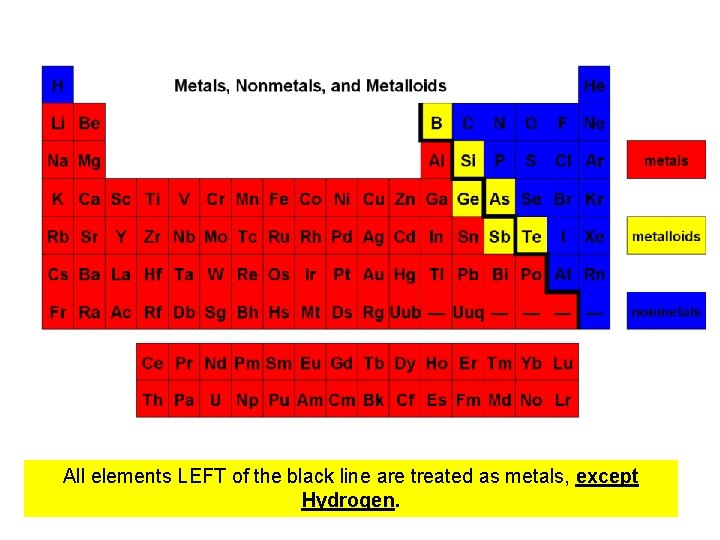
All elements LEFT of the black line are treated as metals, except Hydrogen.

Chemical Groups and Diatomic Molecules • Certain elements are unstable, and hence, do not commonly exist as individual neutral species, but rather as diatomic molecules • These include H, O, N, and all of the halogens (group 17) H H 2 (Hydrogen gas) O O 2 (Oxygen gas) N N 2 (Nitrogen gas) F, Cl, Br, I F 2, Cl 2, Br 2, I 2 (Fluorine (g), chlorine (g), bromine (L) and iodine (s))

Nomenclature: Ionic Compounds • Molecules are formed by atomic bonding. For purposes of naming, we consider two types of molecules: IONIC and COVALENT. Special rules exist for naming molecules of each type. • Ionic “bonds” form between metal ions and nonmetal ions • To name an ionic compound, you do the following 1. Write the name of the metal 2. Follow it with the ionic name of the nonmetal ion Example • KF Potassium Fluoride

Nomenclature: Covalent Compounds • Covalent bonds form between nonmetals • To name an covalent compound: 1. Write the name of the first nonmetal. For non-unity subscripts, use greek prefixes (shown on right) 2. Follow that with the ionic name of the second nonmetal. Again, include greek prefixes. Only use mono- for oxygen containing molecules. Examples CO carbon monoxide N 2 S dinitrogen sulfide CO 2 carbon dioxide P 4 Se 10 tetraphosphorus decaselenide

Nomenclature: The Strange Rules of Hydrogen 1. Hydrogen chalcogenides and halides are named according to ionic rules, if the hydrogen is listed first. Ex. H 2 S (g) = hydrogen sulfide; HF (g) = Hydrogen Fluoride 2. Hydrogen halides dissolved in water (anything dissolved in water is called an “aqueous solution”, abbreviated “aq”) are acids and are named as such. • We drop –gen and end the second nonmetal with the suffix “– ic acid” HCl (aq) = hydrochloric acid ; HF (aq) = hydrofluoric acid 3. If hydrogen is listed last, it is a hydride anion (H-). The name is ionic or covalent depending on what it is paired with. Ex. Mg. H 2 = magnesium hydride (ionic); Te. H 2 = tellerium dihydride (covalent)

Conservation of Mass • All chemical reactions obey the law of conservation of mass. Matter can not created or destroyed. • Therefore, in a chemical reaction, atoms are merely rearranged to create new molecules. The total mass of matter present at the end of a reaction must equal the mass present at the beginning. • You can not create elements via chemical reaction!

Chemical Reactions • As you can see from the chemical equation shown to the left, products typically exhibit vastly different characteristics the reactants • Also recall our discussion on the law of conservation of mass. Based on this law, can you find a problem with the equation written shown?

Balanced Reactions • Mass can not be created or destroyed. This means that every element contained in the reactants must be accounted for in the product(s) • There are two chlorine atoms on the reactant side, and only one chlorine atom one the product side. To balance the chlorine atoms, we add a coefficient of 2 to the product side • We now add a coefficient of 2 to the Na (s). The reaction is now balanced.

Coefficients And Subscripts • The balanced equation above says that two Na atoms react with one chlorine gas molecule to produce two molecules of Na. Cl • The coefficient of 2 means that there are two separate Na atoms • The subscript of 2 indicates two Cl atoms bonded together in a single molecule Na Na Cl Cl Na. Cl • Do not confuse coefficients and subscripts. Do not alter subscripts when balancing.

Tips For Balancing Reactions • Before carrying out any calculations, it is imperative that you first confirm that a given chemical equation is balanced. • The rules for balancing a chemical equation are provided below. 1. First, balance those elements that appear only once on each side of the equation 2. Balance the other elements as needed. Pay attention to subscripts. 3. Include phases

Tips For Balancing Reactions • Let’s balance the equation below using the rules from the previous slide. C 3 H 8 (s) + O 2 (g) CO 2 (g) + H 2 O (L) • We’ll balance C first. C 3 H 8 (s) + O 2 (g) 3 CO 2 (g) + H 2 O (L) • Now balance H. C 3 H 8 (s) + O 2 (g) 3 CO 2 (g) + 4 H 2 O (L) • Now balance O. C 3 H 8 (s) + 5 O 2 (g) 3 CO 2 (g) + 4 H 2 O (L)

Group Work • Balance the following reactions: Sulfur trioxide (g) Sulfur dioxide (g) + Oxygen (g) Dichlorine heptaoxide (g) + Water (L) Hydrochloric acid (aq) + Oxygen (g) Propane (g) + Oxygen (g) Carbon Dioxide (g) + Water (g)

Chemical Groups And Periodicity • As scientists first began to discover and classify the elements, patterns and similarities were observed in chemical behaviors of certain groups of elements. • Consider the three metals Li, Na, and K – All 3 metals have similar appearance, melting points, and densities – The most interesting feature is that all 3 metals react with the same elements in a nearly identical manner • As you see in the periodic table, these elements are all listed in the same group, or vertical column.

Chemical Groups And Periodicity • Dmitri Mendeleev created the periodic table in in 1869 by arranging the elements from left to right in order of increasing atomic number, and vertically according to their behavior (groups) • In doing so, he observed repetitive patterns in chemical behavior across periods (horizontal rows) • This periodicity is described in the next slide.
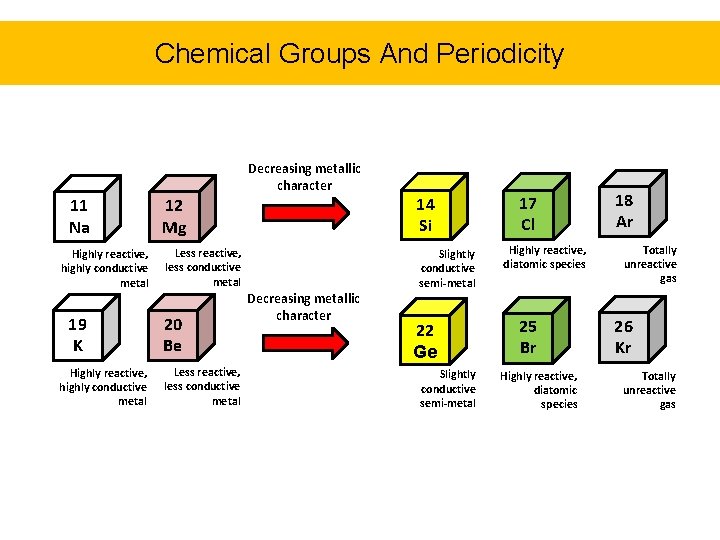
Chemical Groups And Periodicity Decreasing metallic character 11 Na Highly reactive, highly conductive metal 19 K Highly reactive, highly conductive metal 12 Mg 14 Si Less reactive, less conductive metal Slightly conductive semi-metal 20 Be Less reactive, less conductive metal Decreasing metallic character 22 Ge Slightly conductive semi-metal 17 Cl Highly reactive, diatomic species 25 Br Highly reactive, diatomic species 18 Ar Totally unreactive gas 26 Kr Totally unreactive gas

WHY? • We must now answer many questions about chemical reactivity. – Why is it that some atoms join together and form molecules, while others can’t? – Why is there such wide variation in the reactivity and physical properties of elements? – Why is there periodic repetition (periodicity) of the chemical/physical properties of elements as we move across the periodic table?

Explanation Of Elemental Groups • The existence of periodicity proves a very important point: The number of protons in the nucleus has no effect on chemical behavior. If it were so, chemical behaviors would never repeat given that no two elements have the same atomic number. The chemical behavior of an element is dictated by the arrangement of its electrons.

Ionization Energy • A direct indication of the arrangement of electrons about a nucleus is given by the ionization energies of the atom • Ionization energy (IE) is the minimum energy needed to remove an electron (form a cation) completely from a gaseous atom – Ionizations are successive. It becomes increasingly harder to remove additional electrons due to increased attraction to the remaining protons in the nucleus. . IE 1 < IE 2 < IE 3 ……. IEn 1 st Ionization Energy 2 nd Ionization Energy

Ionization Energy • By measuring the energy required to remove electrons from an element, you can gain an idea of: – how “willing” an atom is to lose an electron, and relate this to its reactivity – where the electrons are positioned
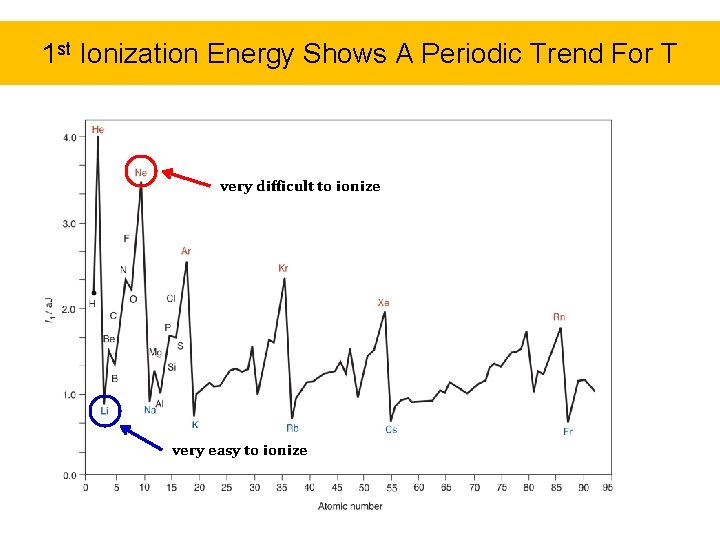
1 st Ionization Energy Shows A Periodic Trend For T very difficult to ionize very easy to ionize
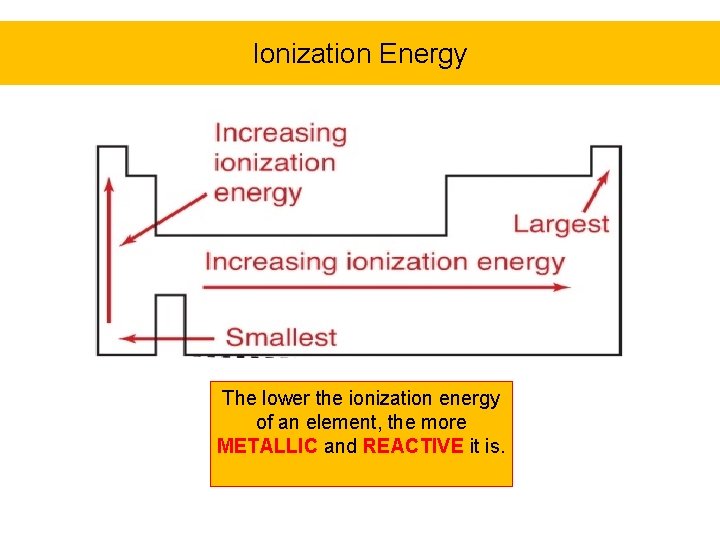
Ionization Energy The lower the ionization energy of an element, the more METALLIC and REACTIVE it is.
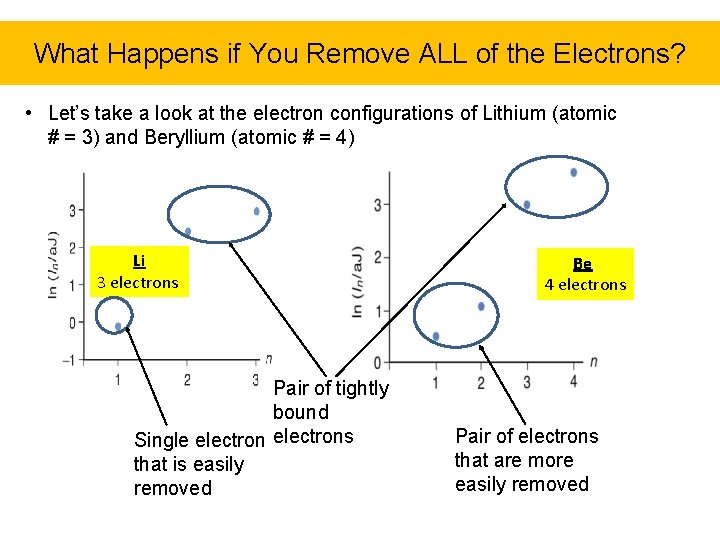
What Happens if You Remove ALL of the Electrons? • Let’s take a look at the electron configurations of Lithium (atomic # = 3) and Beryllium (atomic # = 4) Li 3 electrons Pair of tightly bound Single electrons that is easily removed Be 4 electrons Pair of electrons that are more easily removed
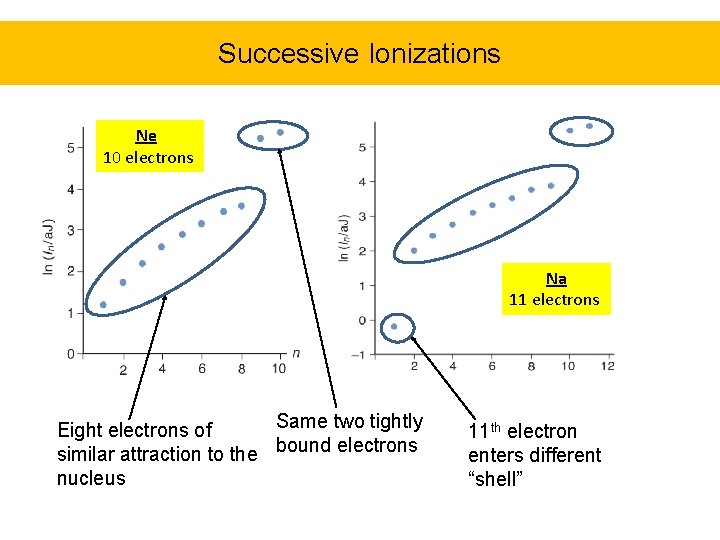
Successive Ionizations Ne 10 electrons Na 11 electrons Same two tightly Eight electrons of similar attraction to the bound electrons nucleus 11 th electron enters different “shell”
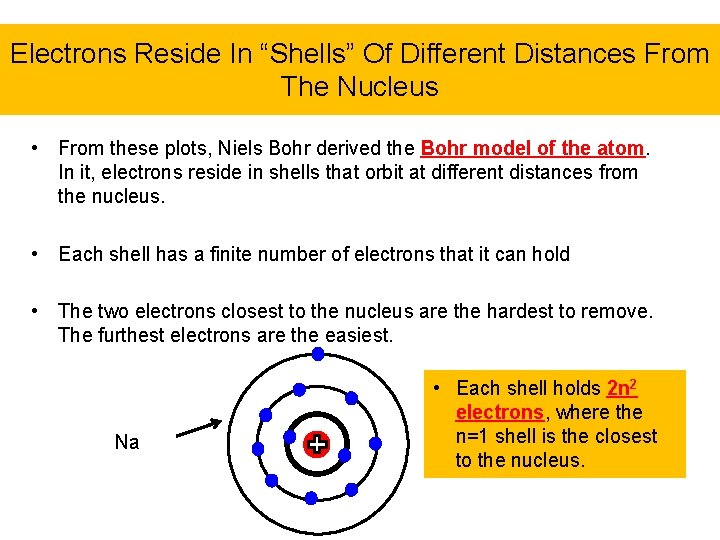
Electrons Reside In “Shells” Of Different Distances From The Nucleus • From these plots, Niels Bohr derived the Bohr model of the atom. In it, electrons reside in shells that orbit at different distances from the nucleus. • Each shell has a finite number of electrons that it can hold • The two electrons closest to the nucleus are the hardest to remove. The furthest electrons are the easiest. Na • Each shell holds 2 n 2 electrons, where the n=1 shell is the closest to the nucleus.

Noble Gas Configurations • Only the outermost electrons are involved in reactions. These are called valence electrons. • The inner-most electrons of an element comprise what is known as a noble gas core. They do not react. – Noble gases are chemically inactive because they have completely filled shells. • Lithium, for example, has a two electron core, which we call a Helium core, and one valence electron. Sodium has a 10 -electron, Neon core, and one valence electron; and so on.

Electron Configurations • Electrons in a shell exist in different “orbitals”. An orbital is a enclosed volume of space around the nucleus that an electron can move throughout. More detail will be provided in CHEM 105. • There are four types of orbitals: – s orbitals (max capacity: 2 electrons) – p orbitals (max capacity: 6 electrons) – d orbitals (max capacity: 10 electrons) – f orbitals (max capacity: 14 electrons) • S-orbitals exist in all shells. P-orbitals exist in all shells where n>2, dorbitals exist in all shells n>3, and f-orbitals exist in all shells n>4.
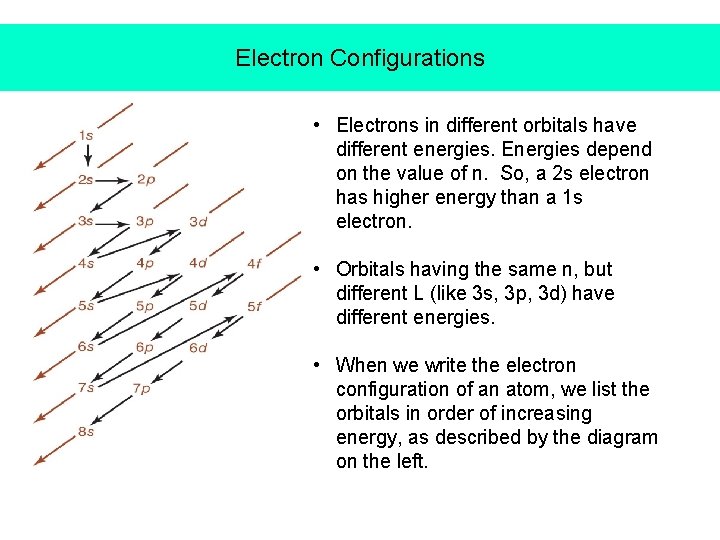
Electron Configurations • Electrons in different orbitals have different energies. Energies depend on the value of n. So, a 2 s electron has higher energy than a 1 s electron. • Orbitals having the same n, but different L (like 3 s, 3 p, 3 d) have different energies. • When we write the electron configuration of an atom, we list the orbitals in order of increasing energy, as described by the diagram on the left.
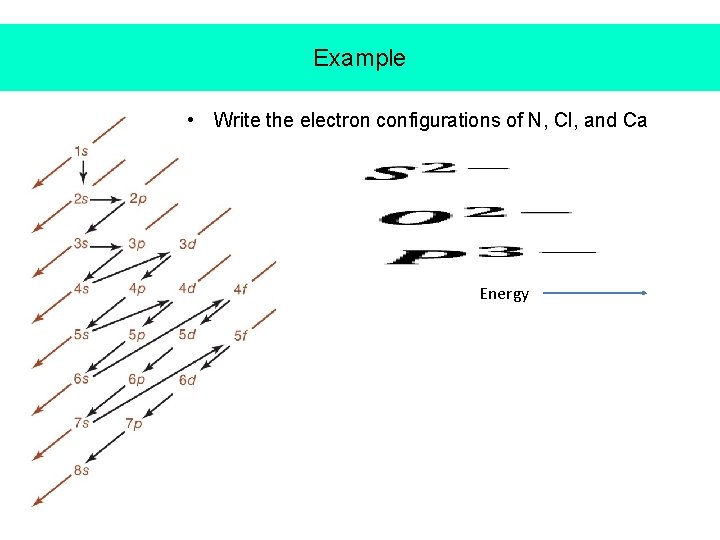
Example • Write the electron configurations of N, Cl, and Ca Energy

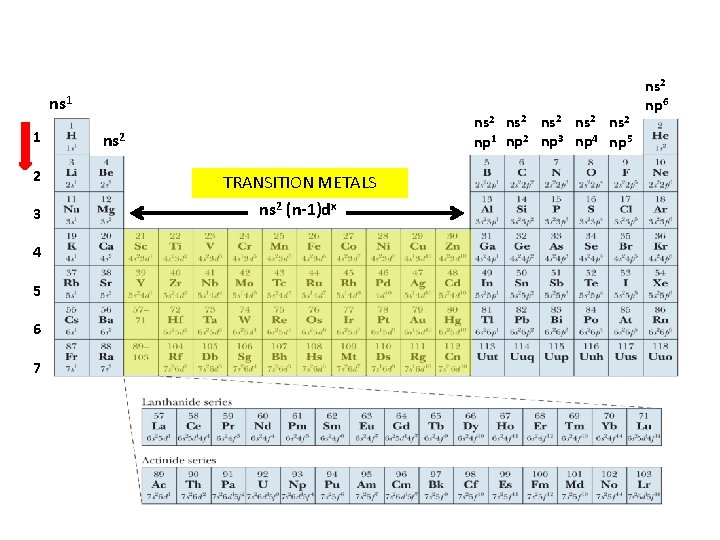
Noble Gas Configurations • Now that we can assign orbitals to electrons, we can write proper valence electron configurations.
ns 1 1 2 3 4 5 6 7 ns 2 ns 2 np 1 np 2 np 3 np 4 np 5 ns 2 TRANSITION METALS ns 2 (n-1)dx ns 2 np 6
- Slides: 36