The Periodic Table Periodic Trends Atomic Radius radius

The Periodic Table

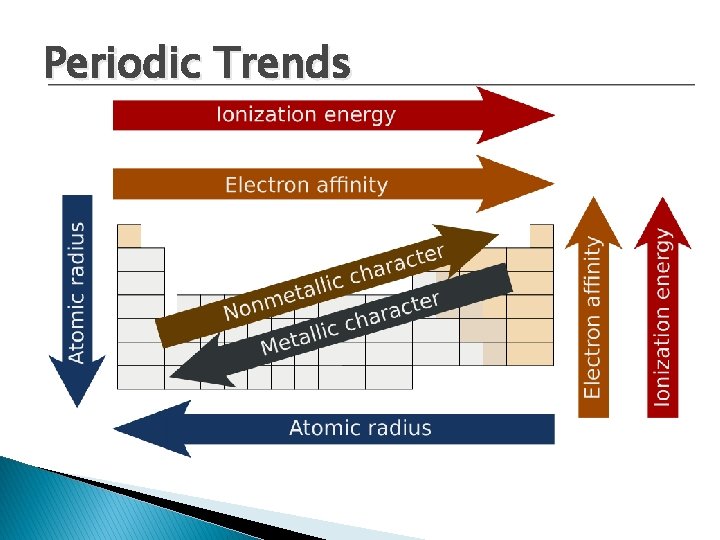
Periodic Trends � Atomic Radius (radius of an atom) ◦ atomic radius is measured (just like any other radius) ◦ the atomic radius of an atom increases each period and decreases from group to group ◦ The shielding effect is how the atomic radius of an atom decreases from left to right on the periodic table � Ionic Radius (radius of an ion) ◦ ions have a different radius than their non-ionic counterparts. � Example: H+ has a different radius than H because it has no electrons ◦ protons pull electrons towards the center, preventing them from escaping orbit ◦ electrons pull away from the center because they are in constant motion � cations are smaller than their neutral form because they have more protons than electrons (more protons means more force keeping electrons in) � anions are larger than their neutral form (more electrons means they can orbit further away)
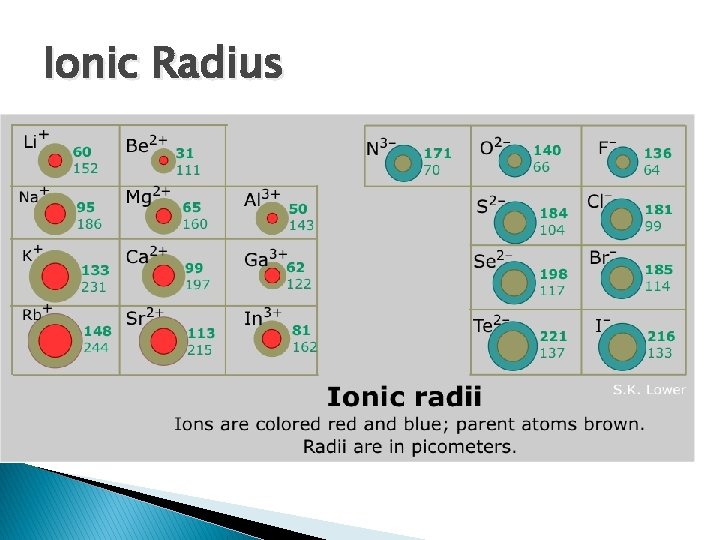
Ionic Radius

Periodic Trends Ionization Energy and Electronegativity � Ionization Energy is the amount of energy required to lose an electron from a regular atom (how easily it will give up an electron) � Electronegativity/Electron Affinity is an atom’s ability to draw electrons toward it when it has a chemical bond � ionization energy and electronegativity share the same patter on the periodic table ◦ They both increase as the group number increases, but decreases as the period number increases
Periodic Trends

Valence Electrons � Valence electrons are the electrons that occupy the highest energy level (1 -7) of the electron cloud. ◦ The highest energy level is equal to the period Example: ◦ Carbon has the electron configuration 1 s 2 2 p 2. ◦ Carbon belongs to period 2, so its highest occupied energy level is 2. ◦ Carbon’s 2 nd energy level has 2 electrons in sublevel s and 2 electrons in sublevel p, therefore carbon has 4 valence electrons

Valence Electrons � Group 1 has 1 valence electron � Group 2 has 2 valence electrons � Groups 3 -12 (transition metals) vary � Group 13 has 3 valence electrons � Group 14 has 4 valence electrons � Group 15 has 5 valence electrons � Group 16 has 6 valence electrons � Group 17 has 7 valence electrons � Group 18 has 8 valence electrons

Formation of Ions � Ionization – The formation of an ion � Groups 1 -14 elements make cations ◦ These ions ALWAYS have 0 valence electrons � Groups 14 -17 elements make anions ◦ These ions ALWAYS have 8 valence electrons � � Group 18 elements (noble gases) don’t make ions because they already have 8 valence electrons The charge of an ion can be determined from the number of valence electrons ◦ 1 valence e- makes a +1 charge because it had to lose 1 electron to equal zero ◦ 6 valence e- makes a -2 charge because it had to gain 2 electrons to equal 8 ◦ 4 valence e- makes either a +4 or a -4 charge, depending on the element
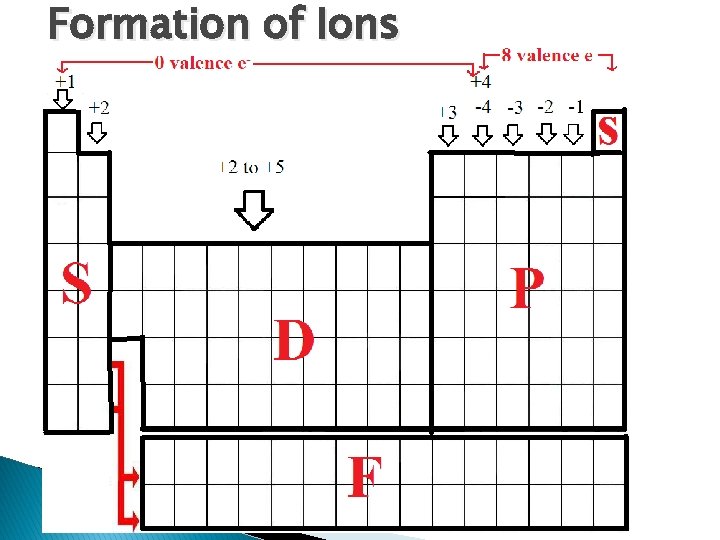
Formation of Ions

Properties of Groups � Group 1 Alkali Metals: Most metallic and reactive group ◦ form +1 cations � Group 2 Alkaline Earth metals: Very reactive group ◦ form +2 cations � Block D Transition Metals: Semi-reactive ◦ form cations with different charges � Block F Lanthanides and Actinides: Radioactive and extremely heavy metals � Group 16 Chalcogens: Very reactive and electronegative ◦ form -2 anions � Group 17 Halogens: Extremely reactive and electronegative group ◦ form -1 anions � Group 18 Noble Gases: Completely unreactive because of complete valence shell

Types of Elements � There are 3 types of elements: metals, metalloids and nonmetals ◦ 1) S, D and F block Metals: Malleable (can change shape without breaking), shiny, conducts electricity ◦ 2) P Block Metalloids: Show both metallic and non-metallic character. Metalloids touch the staircase of the periodic table ◦ 3) P Block Nonmetals: dull, brittle, and poor at conducting electricity because they hog electrons � Note: metallic character trend follows the same pattern as atomic radius

History � Mendeleev was the scientist responsible for seeing the pattern of properties of the elements in the periodic table ◦ he was the first to organize those elements into periods and groups. ◦ Elements within the same group have the same physical and chemical properties. ◦ When he organized his version of the table, there were holes in it because there were only 60 out of 110 known elements at the time. After he died, the holes were later filled by newly discovered elements, verifying what he knew to be correct: � you can predict how an atom will behave by knowing where its atomic number falls on the periodic table. � Periodic law – Both physical and chemical properties of the elements are functions of their atomic numbers.
- Slides: 12