THE PERIODIC TABLE OUTLINE History of Periodic Table
THE PERIODIC TABLE

OUTLINE �History of Periodic Table �Periodic Table Organization �Period �Group/Family �Representative Elements �Transition Elements �Inner Transition Elements �Types of Elements �Metals �Nonmetals �Metalloids

HISTORY. . . �DMITRI MENDELEEV (1834 -1907) �Wrote out elements in order of increasing ATOMIC MASS, and ended up with a table! �Now organized according to: �ATOMIC NUMBER �NUMBER OF ELECTRONS �Why “PERIODIC”? �Periodic means repeating patterns and properties.

HISTORY OF PERIODIC TABLE �By the Mid-1800 s, there were 65 known elements. �Scientists began to recognize patterns after recording information such as: �chemical reactivity (ex: bonding patterns) �physical properties (ex: state of matter, mass)

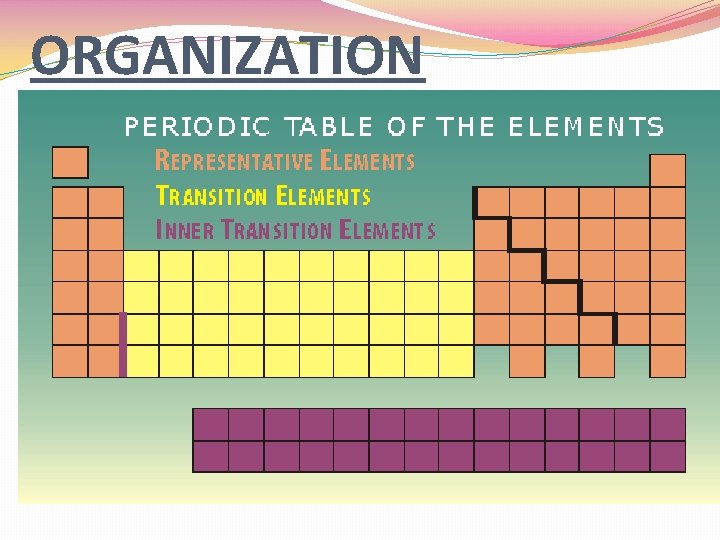
PERIODIC TABLE ORGANIZATION �The periodic table is arranged in rows and columns. �PERIOD �Horizontal rows on table � 7 in total �Atomic Mass and Atomic Number increase from LEFT TO RIGHT

ORGANIZATION. . . �GROUP �Also known as FAMILY �Vertical columns on table �Elements of the same group have similar but not identical properties. �There are two number systems for groups: �CURRENT: �OLD: 1 -18 ROMAN NUMERALS IA – VIIIA IB - VIIIB


�Diatomic Elements – (7 -up) �nitrogen, oxygen, fluorine, chlorine, bromine, iodine and hydrogen �These elements can NOT survive in nature by themselves – have to bond to become stable. � 2 MORE – sulfur and phosphorus

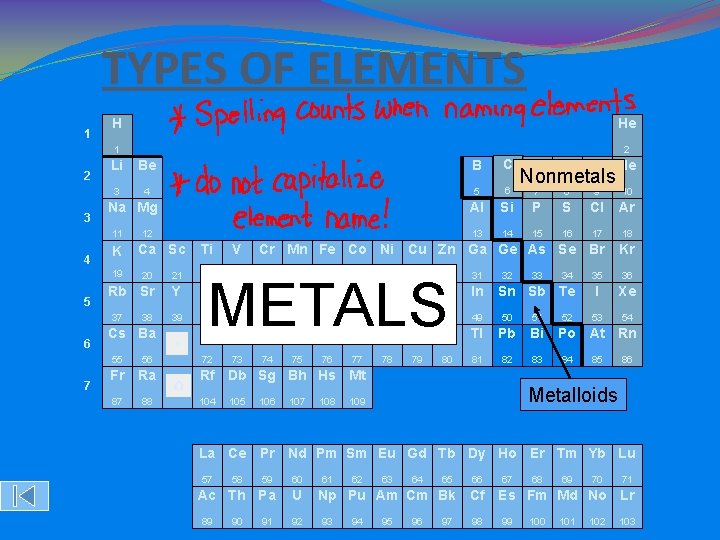
TYPES OF ELEMENTS �There are three types of elements on the periodic table: �Metals �Nonmetals �Metalloids �The STAIRCASE LINE on the periodic table divides the METALS and the NONMETALS. �The ratio of metals to nonmetals is about 4: 1.

ORGANIZATION. . . �GROUP 1 – Alkali Reactivity Soft, silver, react violently with water �GROUP 2 – Alkaline Earth Metals Reactivity Link �Shiny, silver, light, reactive �GROUP 17 – Halogens Info Link �Extremely reactive, poisonous �GROUP 18 – Noble Gases Info Link �INERT Not reactive under normal laboratory conditions
ORGANIZATION

Organization. . . �REPRESENTATIVE ELEMENTS �AKA Group A elements �Groups IA-VIIIA, or 1, 2, 13 -18 �These elements illustrate the ENTIRE RANGE OF PROPERTIES OF ELEMENTS.

ORGANIZATION. . . �TRANSITION ELEMENTS �AKA Group B elements �Group IB-VIIIB, or the “lower” groups �Metallic elements that exhibit some different properties due to their electron arrangements.

ORGANIZATION. . . �INNER TRANSITION ELEMENTS �Removed from the main table as a matter of convenience in organizing table �Two names: � 57 -71 LANTHANIDES (rare earth) � 89 -103 ACTINIDES (radioactive)
TYPES OF ELEMENTS 1 2 3 H He 1 2 Li Be B C 3 4 5 6 Al 13 Na Mg 11 4 K 19 5 7 Ca Sc O F Ne 7 8 9 10 Si P S Cl Ar 14 15 16 17 18 Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr 23 24 35 36 I Xe 53 54 20 21 22 Rb Sr Y Zr Nb Mo Tc Ru Rh Pd Ag Cd In 39 40 41 42 49 50 51 Hf Ta W 72 73 74 37 6 12 N Nonmetals 38 Cs Ba 55 56 Fr Ra 87 88 * W 25 26 27 28 29 30 METALS 43 44 Re Os 75 76 47 32 33 46 Ir Pt Au Hg Tl Pb Bi 77 78 81 82 83 80 34 Sn Sb Te 45 79 48 31 52 Po At Rn 84 85 86 Rf Db Sg Bh Hs Mt 104 105 106 107 108 Metalloids 109 La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb Lu 57 59 60 Ac Th Pa U 89 58 90 91 92 61 62 63 64 65 66 Np Pu Am Cm Bk Cf 93 94 95 96 97 98 67 68 69 70 71 Es Fm Md No Lr 99 100 101 102 103

METALS �Substances found on the Left Hand Side of the staircase on the periodic table. �Physical properties include: �Shiny �Solids at room temperature (except Hg) �Good conductors of electricity �Ductile (can be stretched into wire) �Malleable (can be hammered in shapes)

NON-METALS �Substances found on the Right Hand Side of the staircase on the periodic table. �Physical properties include: �Dull �Brittle �S, L, or G at room temperature �Poor conductors/good insulators

METALLOIDS �AKA semi-metals �Substances that are found ON the staircase on the periodic table. �Have properties of BOTH metals and nonmetals. �In general, �Hard, but sometimes brittle �Solid �High melting points �Weak (or not at all) conductors of electricity

HYDROGEN �The “rebel” of the periodic table! �It does not really “belong” anywhere. �Hydrogen is usually located on the top left hand corner of the table, even though it has both metallic and nonmetallic properties. �Hydrogen Experiment Review of the Periodic Table

�Periodic table song �Periodic table rap

Element Symbol Atomic # # Electrons #Atomic Mass #Protons # Neutrons fluorine zinc arsenic Atomic # = # electrons = # protons Always round atomic mass to nearest whole number when determining the number of neutrons! # neutrons = Atomic Mass - # protons Periodic Table worksheet
- Slides: 21