The Periodic Table of Elements Periodic Table l

The Periodic Table of Elements

Periodic Table l Arranged by Russian chemist Dimitri Mendeleev (late 1800’s) l Originally arranged by increasing atomic mass l Later arranged by increasing atomic # (Henry Moseley)

Columns l Known as Groups or Families l. A group or family has similar but not identical properties l Example: Group 1 or Alkali Metals l. Properties – soft, white, shiny metals, highly reactive

We Are Family!
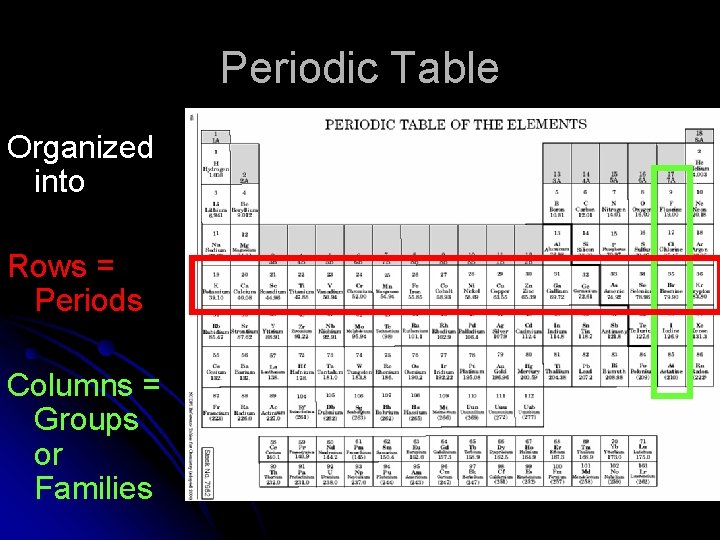
Periodic Table Organized into Rows = Periods Columns = Groups or Families

Special Groups Group 1 – Alkali Metals l Group 2 – Alkaline Earth Metals l Group 17 – Halogens l Group 18 – Noble Gases l

Metals/Non-metals Notice the dark stairstep line. Separates metals from nonmetals

Metals/Metalloids/Nonmetals l Metalloids l Properties between metals and nonmetals l Have a side touching the stairstep line EXCEPT Al (metal) l Nonmetals

Rows l Known as periods l Are not alike in properties, but there is a pattern l. The first element in a period is almost always a reactive solid l. The last element is an inert or inactive gas

Group Practice l Give the group number for each element: l Calcium l Tellurium l Cesium l Chlorine l Argon

Rows l Each period tells you how many energy levels each atom has l. Each energy level can hold a certain number of electrons l. Energy level 1 - 2 electrons l. Energy level 2 - 10 electrons l. Energy level 3 - 18 electrons

Practice Give the element from the periodic table: l Period 2, Group 14 l Period 4, Group 3 l Period 3, Group 17 l Period 1, Group 8 l Period 4, Group 2 l Period 2, Group 4 l

Why do we care about how many electrons an atom has? l The reactivity of an element is due to its valence electrons l Valence electrons are the electrons found in the outer shell of an atom. Nucleus Shell or Energy Level Valence Electrons

Trick to the Dot Diagrams l Some atoms are very easy to figure out how many electrons are in the valence shell l Group 1 has 1 l Group 2 has 2 l Group 13 has 3 l Group 14 has 4 (are you noticing a pattern yet? ) l Group 15 has 5 l Group 16 has 6 l Group 17 has 7 l Group 18 has 8 l Groups 3 -12 always have 2 electrons

Dot Diagrams l A diagram to represent electrons in the outer energy level of an atom l Uses the atomic symbol and dots l Example: Hydrogen H • Beryllium Be: Carbon : C:

Dot Diagram How-To Figure out how many valence electrons the atom has. 2. Write the symbol for the element. 3. Add dots in pairs on each side of the symbol (imagine that it is inside a box) to represent the valence electrons. Example: l Silicon l 4 valence electrons 1.

Dot Diagram Practice Draw Dot Diagrams for each of the following: l Hydrogen l Chlorine l Calcium l Nitrogen l Aluminum l
- Slides: 17