The Periodic Table of Elements Dmitri Mendeleev published

The Periodic Table of Elements • Dmitri Mendeleev published the first periodic table in 1869 • Made it possible for scientists to predict the existence of elements that had not been discovered yet • http: //www. youtube. com/watch? v=z. UDDi. Wt. Ft. EM periodic table song

Organization Periodic means- happening at regular intervals • Arranged in increasing atomic mass and atomic number • Periods are the horizontal rows of elements • Same number of energy levels for electrons • Groups or families are the vertical column of elements • Share chemical properties • Same # of valence electrons
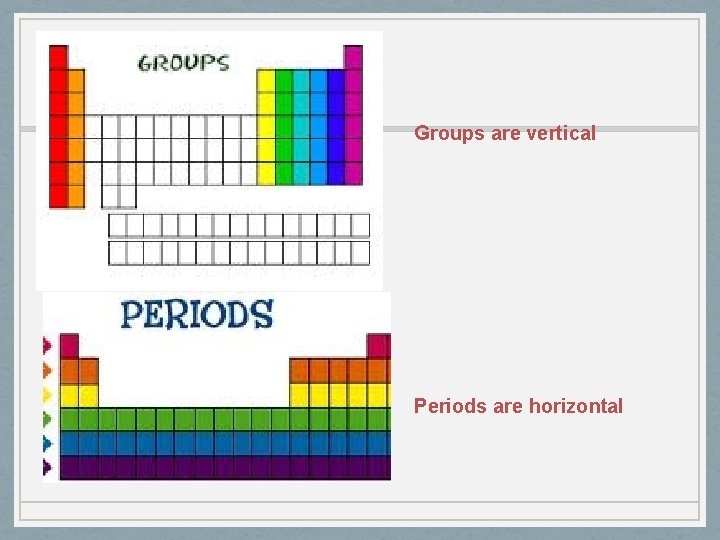
Groups are vertical Periods are horizontal

Periodic Law • The repeating chemical and physical properties of elements change periodically with the element atomic number

Metals, Nonmetals, & Metalloids • Metals • Most elements are metals • Found to the left of the zigzag line • Properties: malleable or ductile, good electric & thermal conductors
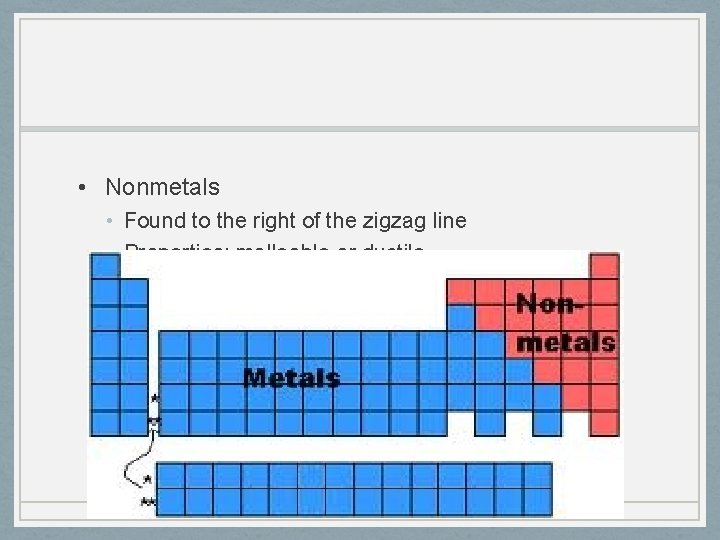
• Nonmetals • Found to the right of the zigzag line • Properties: malleable or ductile

• Metalloids • Border the zigzag line • Properties of metals and nonmetals • Properties: shiny, brittle, hard, good conductors of electric current

Groups • Alkali metals- found in group 1 of the periodic table • • • Contains metals Electrons in outer energy level 1 Electrons in the out level (VE) 1 Very reactive Properties: soft, silver in color, shiny, low density

Alkaline-Earth Metals- found in group 2 of the periodic table • Contains metals • Electrons in outer level (VE) 2 • Very reactive, but less than alkali metals • Properties: silver in color, higher density than alkali metals

Transition Metals- found in groups 3 -12 of the periodic table • Contains metals • Electrons in the outer energy level (VE) 1 or 2 • Less reactive than alkaline earth metal group • Properties: shiny, good conductors of thermal energy & electric current, higher densities and melting points of groups 1 and 2

Halogens- found in group 17 of the periodic table • Contains nonmetals • Electrons in the outer energy level (VE) 7 • Very reactive • Properties: poor conductors of electric current, violent reactions with alkali metals to form salts, never in pure form in nature

Nobel Gases- found in group 18 of the periodic table • Contains nonmetals • Electrons in the outer energy level (VE) 8 • Nonreactive • Properties: colorless, odorless, gases at room temperature

Vocabulary • Make index cards for the following terms for homework. Periodic alkali metal Period alkaline-earth metal Group halogen Periodic law noble gas Valence electron
- Slides: 13