The Periodic table n The periodic table is

The Periodic table n The periodic table is a way of keeping track of all of the elements. Each element has a symbol. Some symbols are only one capital letter (Example hydrogen – H), but some sumbols are two letters. A two letter element symbol is always a capital letter followed by a small letter (Example Calcium- Ca)

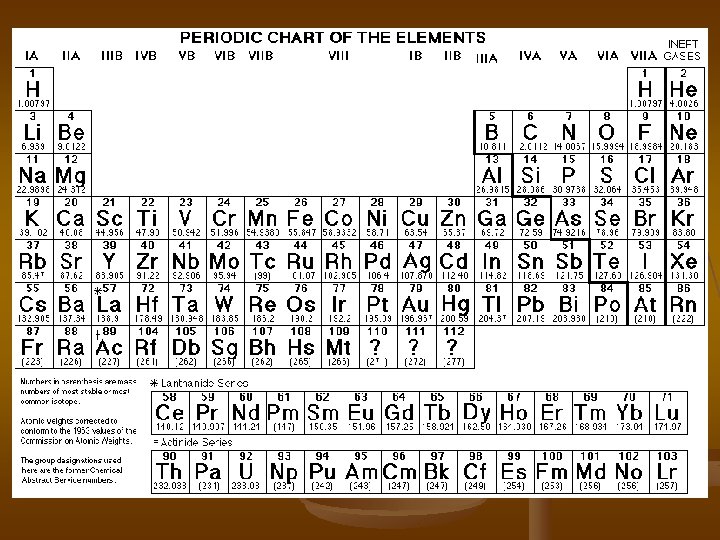
The Periodic Table The periodic table is arranged in the way it is for a reason! Look at the imaginary periodic table you drew Bohr diagrams on. What do you notice about all the elements in the same column?
n n The columns on the periodic table are called groups or families. Each family has the same number of electrons in their outer shell.

n n n Each family also has a number (at the top of the column). This number is the same as # of outer shell electrons. This is an easy way to double check your work when drawing Bohr models.

n n n Some of the important families have names: Alkali metals Group 1 Alkaline Earth Metals - Group 2 Chalcogens Group 6 A (16) Halogens Group 7 A (17) Noble Gases Group 8 A (18)

n n n The families react the same way, chemically, in most situations. The only difference is, as you go further down the periodic table, they get more reactive. How quickly and strongly something reacts with something else is called reactivity.

n n n The rows across the periodic table are also numbered. There are called rows or periods (hence the name). Look at your Bohr models again. What do you notice about the numbers of rows?

n n That’s right!! The number of the rows is equal to the # of shells we need to put electrons in. Another easy way to check your work!

n n n Now look at the periodic table in the back of your text. Green symbols represent metals. Think about metals you have seen. List some properties of metals. Shiny Ductile Malleable Conduct heat & electricity

n n n The orange elements are called nonmetals. List some properties of non metals. Dull Brittle Poor conductors (insulators)

n n The purple elements are called metalloids. This means that they sometimes act like metals, and sometimes act like non-metals. They can’t make up their minds!

n n Right through the metalloids runs something called the staircase. This separates metals (on the left side) from the non-metals (on the right side).
- Slides: 13