The Periodic Table Mendeleevs Periodic Table By the
“The Periodic Table”

Mendeleev’s Periodic Table • By the mid-1800 s, about 70 elements were known to exist • Dmitri Mendeleev – a Russian chemist and teacher • Arranged elements in order of increasing atomic mass • Thus, the first “Periodic Table. ”

The Periodic Law says: • Horizontal rows = periods –There are 7 periods –Same number of energy levels • Vertical column = group (or family) There are 18 groups. –Similar physical & chemical prop. –Identified by number & letter (IA, IIA)

Areas of the periodic table Three classes of elements are: 1)metals, 2)nonmetals, and 3)metalloids 1) Metals: electrical conductors, have luster, ductile, malleable. Are on the left side of the periodic table. 2) Nonmetals: generally brittle and nonlustrous, poor conductors of heat and electricity

Areas of the periodic table – Some nonmetals are gases (O, N, Cl); some are brittle solids (S); one is a fuming dark red liquid (Br) – Are on the right side of the periodic table Notice the heavy, stair-step line? 3) Metalloids: border the line-2 sides – Properties are intermediate between metals and nonmetals – Live on or below the bold line

A. Metallic Character • Metals • Nonmetals • Metalloids

Squares in the Periodic Table • The periodic table displays the symbols and names of the elements, along with information about the structure of their atoms: • Atomic number and atomic mass • Black symbol = solid; red = gas; blue = liquid • (from the Periodic Table on our classroom wall)

Groups of elements - family names • Group IA – alkali metals – Forms a “base” (or alkali) when reacting with water (not just dissolved!) – have 1 electron in their outermost shell • Group 2 A – alkaline earth metals – Also form bases with water; do not dissolve well, hence “earth metals” – have 2 electrons in their outermost shell • Group 3 A – boron family – have 3 electrons in their outermost shell • Group 4 A – carbon family – have 4 electrons in their outermost shell

Groups of elements - family names • Group 5 A – nitrogen family (pnitogen) – have 5 electrons in their outermost shell • Group 6 A –oxygen family – have 6 electrons in their outermost shell • Group 7 A – halogens – Means “salt-forming” – have 7 electrons in their outermost shell • Group 8 A – noble gases – Have 8 electrons in their outermost shell
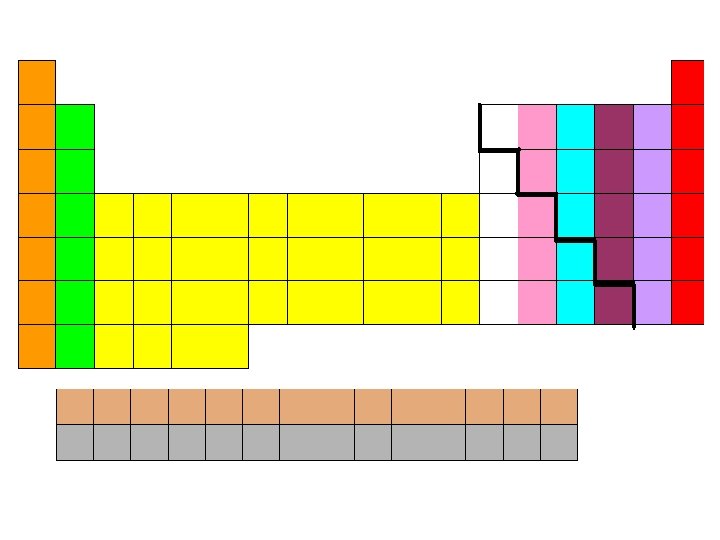

Electron Configurations in Groups Elements can be sorted into 4 different groupings based on their electron configurations: 1) Transition metals Filling D orbital 2) Inner transition metals Filling F orbital 3) Representative elements Filling S or P orbital 4) Noble gases FULL outer orbital
- Slides: 11