The Periodic Table https www youtube comwatch vt

The Periodic Table

https: //www. youtube. com/watch? v=t. OGM 2 g. OHKPc
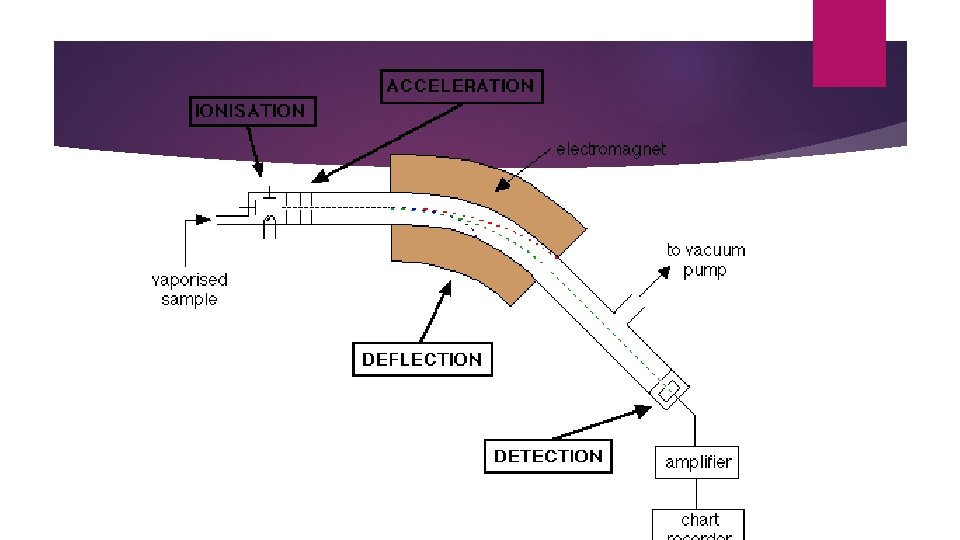

THE MASS SPECTROMETER Atoms can be deflected by magnetic fields - provided the atom is first turned into an ion.

Stage 1: Vaporisation Gas or liquid injected into the sample inlet. A good vacuum turns the liquid to gas. If solid then it may need to be heated first.
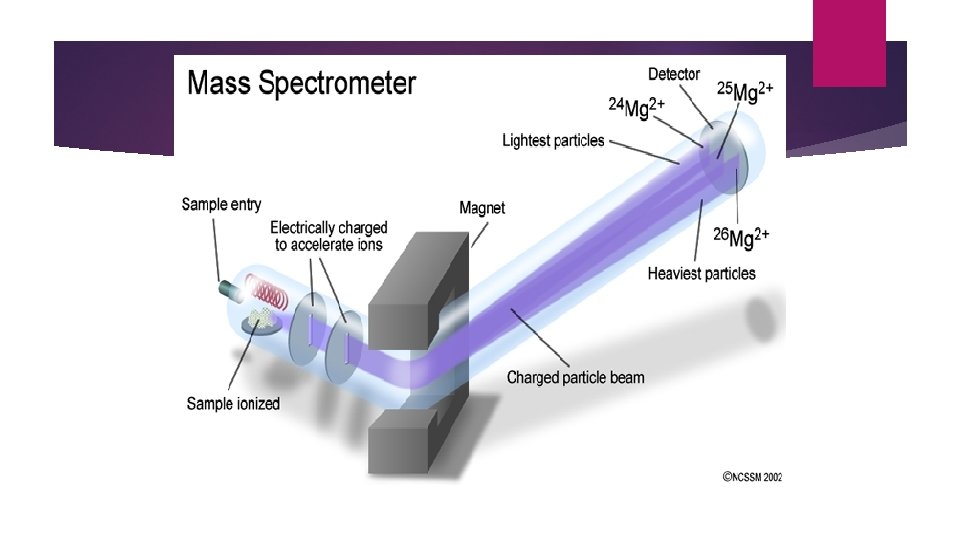

Stage 2: Ionisation The atom is ionised by knocking one or more electrons off to give a positive ion using an electron gun in an ionization chamber. X + e- X+ +2 e-

Stage 3: Acceleration The positive ions are accelerated using negatively charged plates in an accelerator so that they all have the same kinetic energy.

Stage 4: separation The beam is passed into an analyser unit. The positive ions are made to move in curved paths due to the magnetic field from the electromagnet. Lighter particles are deflected further meaning the ions are separated according to their masses. This is the principle of mass spectrometry.

Stage 5: Detection The positive ions fall on the detector based on their masses. The signal is amplified into an electric current and viewed as a series of different sized peaks called a mass spectrum.

Uses of mass spectrometry Identify isotopes and their abundances. Measure mass. Identify relative atomic mass and molecular unknown compounds e. g drugs.

Match up!
- Slides: 12