THE PERIODIC TABLE History of the periodic table

THE PERIODIC TABLE History of the periodic table and the creation of the modern design

Overview RECALL: • The periodic table lists the 110+ elements • All matter is made up of a different combinations of elements • Elements are pure substances and cannot be broken down into simpler substances • Compounds are elements formed into molecules.

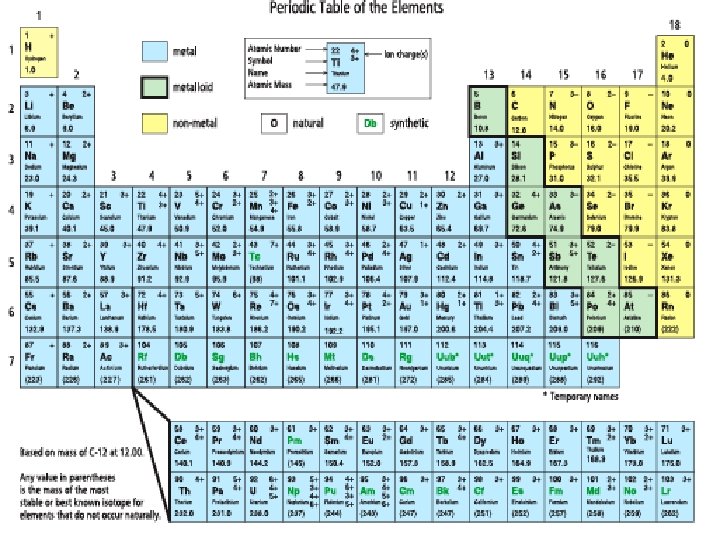
Elements • Each element on the • table explains the following information: Atomic number = # of protons in the nucleus of that atom (notice how the elements increase in atomic number, 1 is at the top right and increases as you move to the right and down the table)

Elements Each element on the table explains the following information: • The element’s symbol and name § Short hand (one or two letters) and name • Atomic Mass: § Mass of average atom of element § # of protons + # of neutrons

Elements Each element on the table explains the following information: • Ion Charge: § The charge that is created when an element gains or loses electrons § An atom that gains an electron gets a negative charge. Why? § An atom that loses an electron gets a positive charge. Why? § If element has more than one charge it can form more than one type of bond.
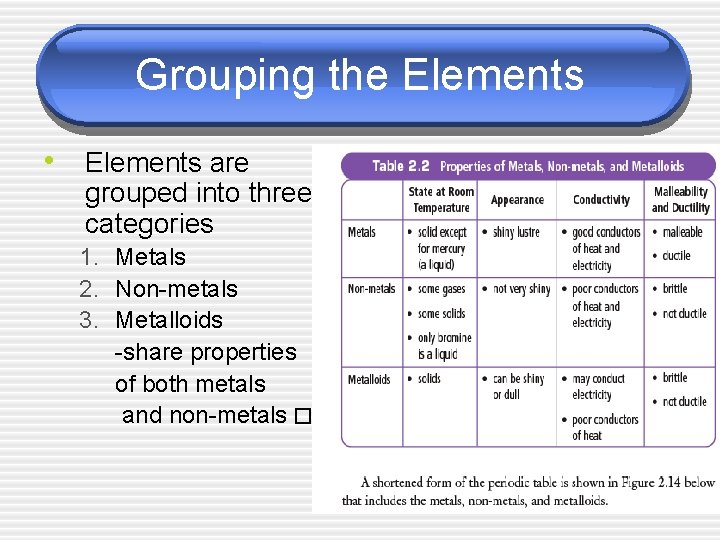
Grouping the Elements • Elements are grouped into three categories 1. Metals 2. Non-metals 3. Metalloids -share properties of both metals and non-metals � �

How the table is organized Horizontally • Each horizontal row is called a PERIOD. • Periods are numbered 1 -7 moving down the table Vertically • Each vertical column is called a FAMILY or GROUP. • Elements in the same family have similar properties and characteristics. • Families are numbered 1 -18.
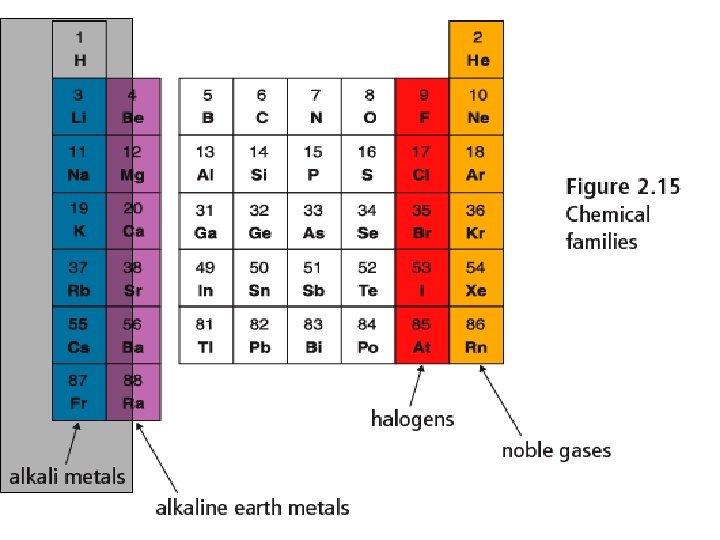

Alkali Metals - Group 1 • Alkali Metals § Include Lithium, Sodium and Potassium § Reactive metals that increase in reactivity as you move down the table. React vigorously with water. § Reactivity § Soft metals that you can cut with a knife § Very low melting points
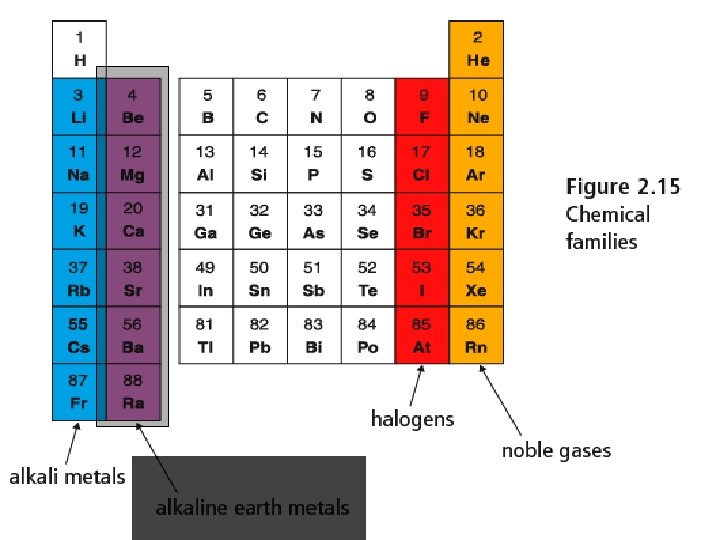

Alkaline Earth Metals Group 2 • Less reactive than • • • Alkali metals Burn in air if heated Produce bright flames and used in fireworks Will react with water, but less than alkali metals.
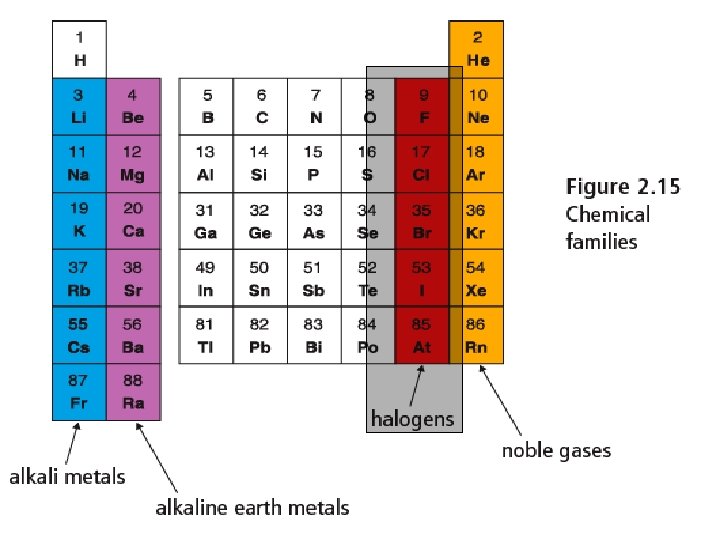

Halogens - Group 17 • Non-metals and highly • • • reactive Fluorine and Chlorine are gases, Bromine is a liquid and Iodine is a solid at room temperature. Fluorine is most reactive, iodine is least Often used as bleaching agents for cleaning (kills bacteria)
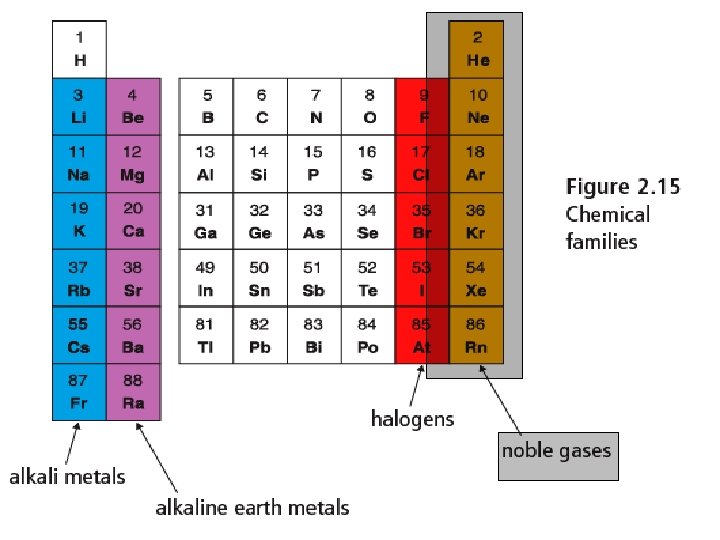

Noble Gases - Group 18 • Most stable and un • • reactive elements Colourless, odourless gases at room temp. Neon and argon used in light fixture and glow in distinctive colours.
Homework • Chapter 2. 2 Review Pg 63 #2, 4, 5, 8, 9, 1114, 17 -21.
- Slides: 18