The Periodic Table History of the Periodic Table

The Periodic Table

History of the Periodic Table n 1871 Mendeleev: The First Periodic Table n Arranged elements by increasing Atomic Mass Noticed repetition, or periodic pattern, in the chemical and physical properties of known elements n Blank spaces – Predicted the existence of elements not yet discovered n based on trends in properties n 1913 n n n Moseley Determined that atoms of each element contain a unique number of protons in their nuclei – Number of Protons = Atomic Number Arranged table by increasing Atomic Number Clear periodic pattern observed
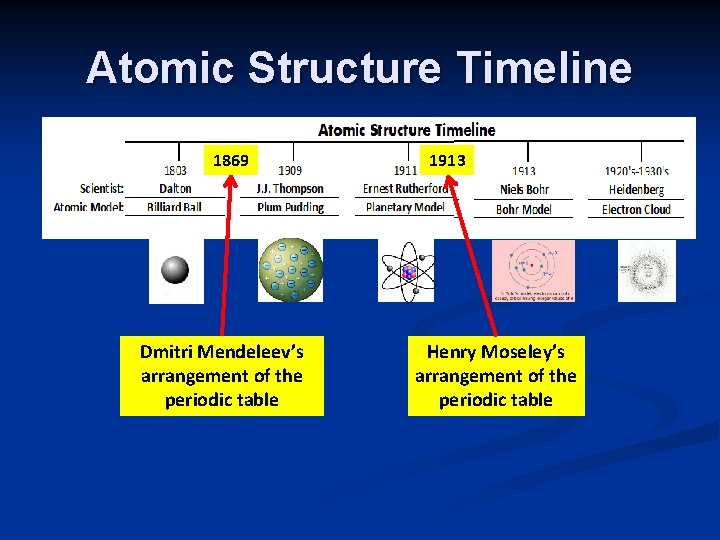
Atomic Structure Timeline 1869 Dmitri Mendeleev’s arrangement of the periodic table 1913 Henry Moseley’s arrangement of the periodic table

PERIODIC LAW When elements are arranged in order of increasing atomic number: There is a periodic repetition of their physical and chemical properties You can determine the properties of an element based on its location on the periodic table Write this on the back of your paper (up at the top)

Locate the Periodic Table on the bottom of the page n n n Color in the mini periodic table like you will see it on the next slide Be sure to fill in the key with the colors you chose Next to the word metal, put: (cation + charge) Next to the work nonmetal, put: (anion - charge) Next to the word group, put the definition on the following slide Next to the word period, put the definition on the following slide
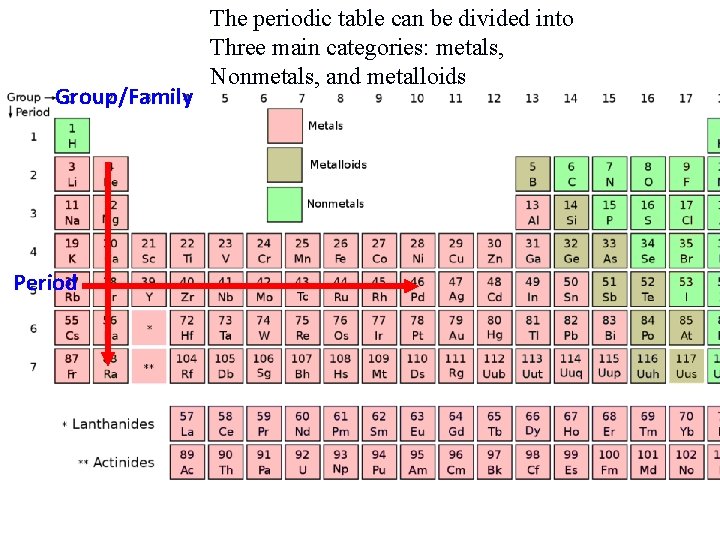
Group/Family Period The periodic table can be divided into Three main categories: metals, Nonmetals, and metalloids

The Modern Periodic Table n 18 Groups/Families Group/Family Vertical Columns n Elements have similar physical and chemical properties and reactivities Period n 7 Periods Horizontal Rows Determines number of energy levels

Locate the Periodic Table on the TOP of the page n n In the appropriate space on the top of the page, write the definition for both valence electrons and oxidation number Fill in the oxidation numbers and valence electrons for each of the groups on the periodic table

Oxidation Number The charge on an ion of an element; The number of electrons lost, gained, or shared during chemical bonding Oxidation # +1 -1 +2 +3 +/-4 -3 -2 0

Valence Electrons in the outermost energy level (valence shell) Valence Electrons 1 7 2 3 4 5 6 8

Locate the Periodic Table on the TOP of the page n n For each group that is on the page, write the important properties next to the group name and color in the group whatever color you choose. Make sure to fill in the key with the appropriate colors that you chose
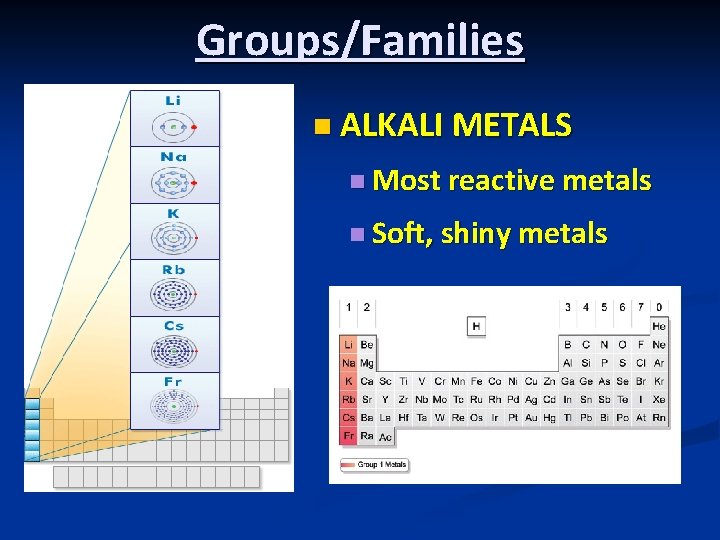
Groups/Families n ALKALI METALS n Most reactive metals n Soft, shiny metals

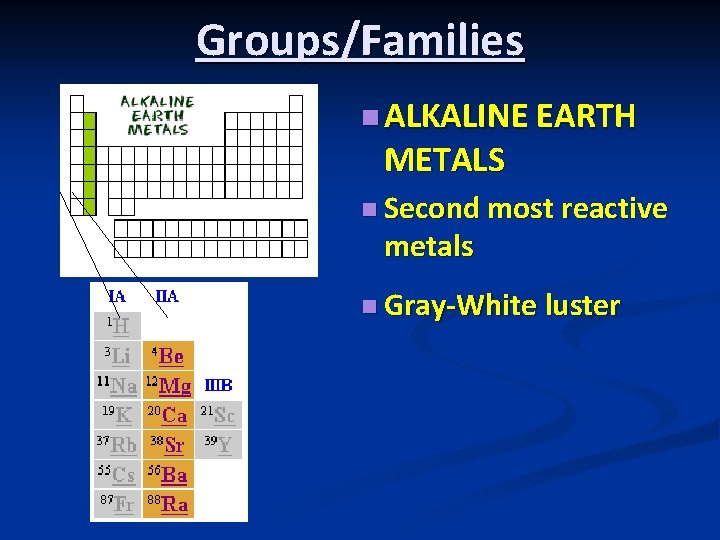
Groups/Families n ALKALINE EARTH METALS n Second most reactive metals n Gray-White luster

Groups/Families n LANTHANIDES n Rare Earth Elements n Soft, malleable metals n Great conductors

Groups/Families n ACTINIDES n Rare Earth Elements n Radioactive n Almost all are manmade

Groups/Families n TRANSITION METALS n Less active than Group 1 or 2 metals n Have variable numbers of valence electrons

Groups/Families n HALOGEN FAMILY n n Most reactive Non -Metals Fluorine most REACTIVE element!!
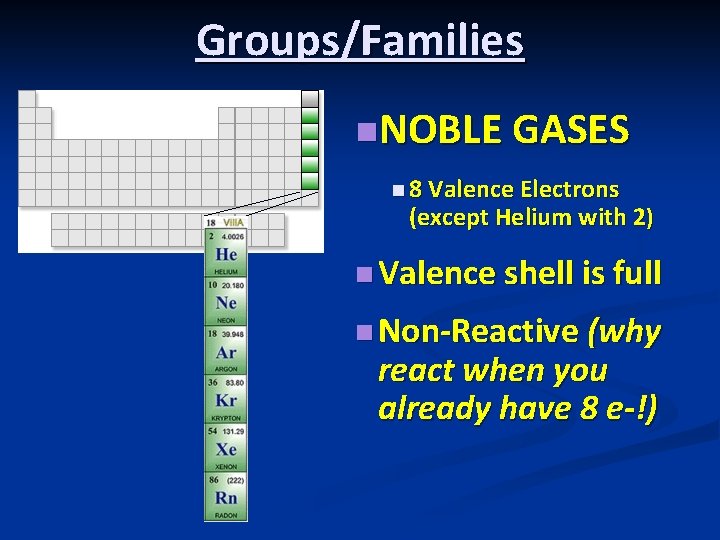
Groups/Families n. NOBLE GASES n 8 Valence Electrons (except Helium with 2) n Valence shell is full n Non-Reactive (why react when you already have 8 e-!)

Turn to the back of the page n n Label each mini periodic table with the trends. Draw and label all arrows and any extra information that I added to the slide Explanation of the trend – the third to the last slide has a summary of all of the trends for you to look over and summarize in that box.

Ionic Radius n Size of an ion n distance from nucleus to outermost e-
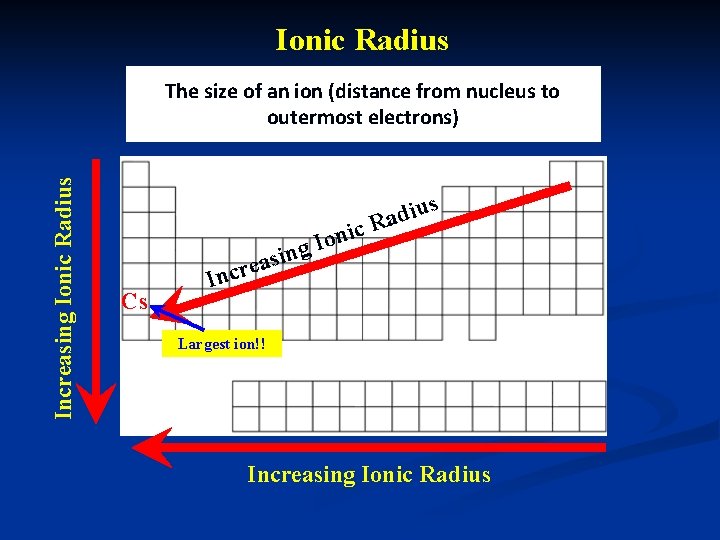
Ionic Radius Increasing Ionic Radius The size of an ion (distance from nucleus to outermost electrons) s iu d a c. R ni o I ng Cs i s a e ncr I Largest ion!! Increasing Ionic Radius

Atomic Radius n Size of an atom n distance from nucleus to outermost e-

Atomic Radius Increasing Atomic Radius The size of an atom (distance from nucleus to outermost electrons) s iu d a c. R i m o t A ng Cs i s a e ncr I Largest atom!! Increasing Atomic Radius

Ionization Energy n Ionization Energy: n Energy required to remove electrons from an atom n LOW ionization energy EASY for an atom to lose electrons n Ex: Group 1 elements n HIGH ionization energy HARD for an atom to lose electrons n Ex: Group 17 elements

Ionization Energy The energy required to remove electrons from an atom Increasing Ionization Energy n Increasing Ionization Energy In ng i s a cre tio a z i Ion y g r e En

Electronegativity n Electronegativity: n Tendency for an atom to take electrons n HIGH Electronegativity Tends to GAIN electrons easily n LOW Electronegativity electrons Tends to LOSE
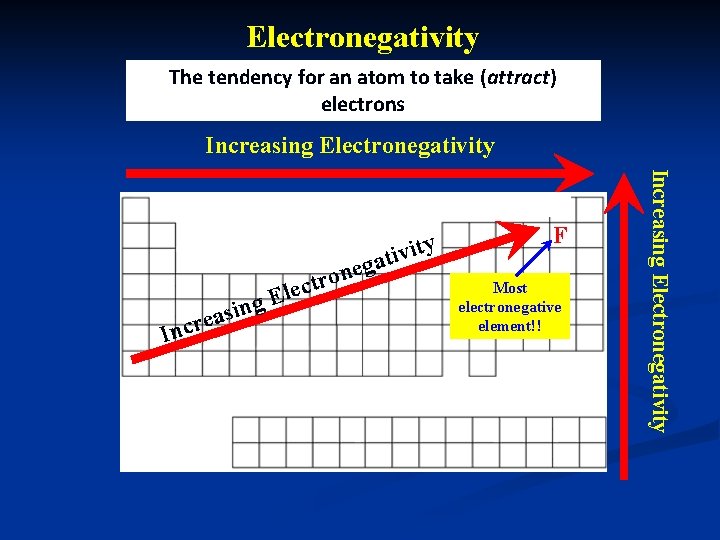
Electronegativity The tendency for an atom to take (attract) electrons Increasing Electronegativity si a e r Inc ng E ne o r t lec F Most electronegative element!! Increasing Electronegativity v i t ga

- Slides: 29