The Periodic Table Foldable Notes Alkali Metals Color

The Periodic Table Foldable Notes
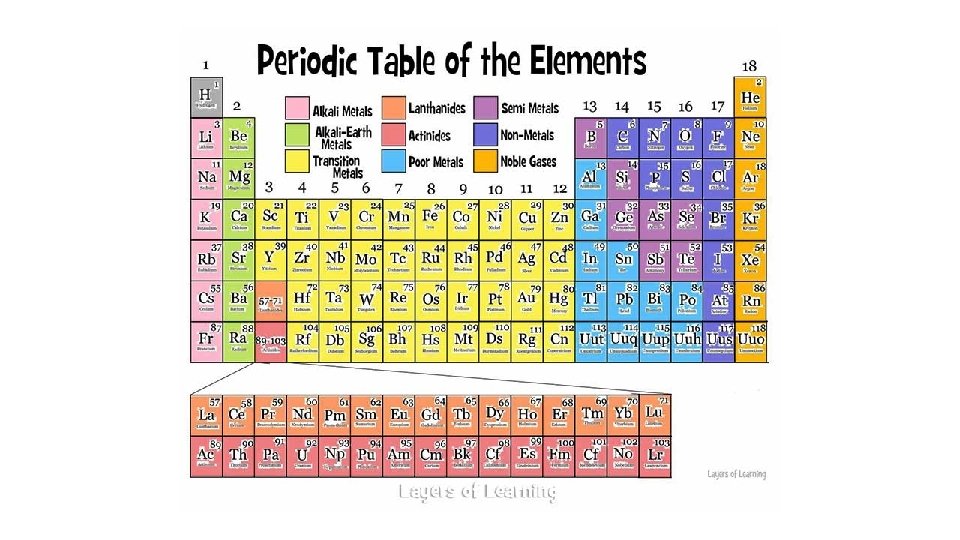
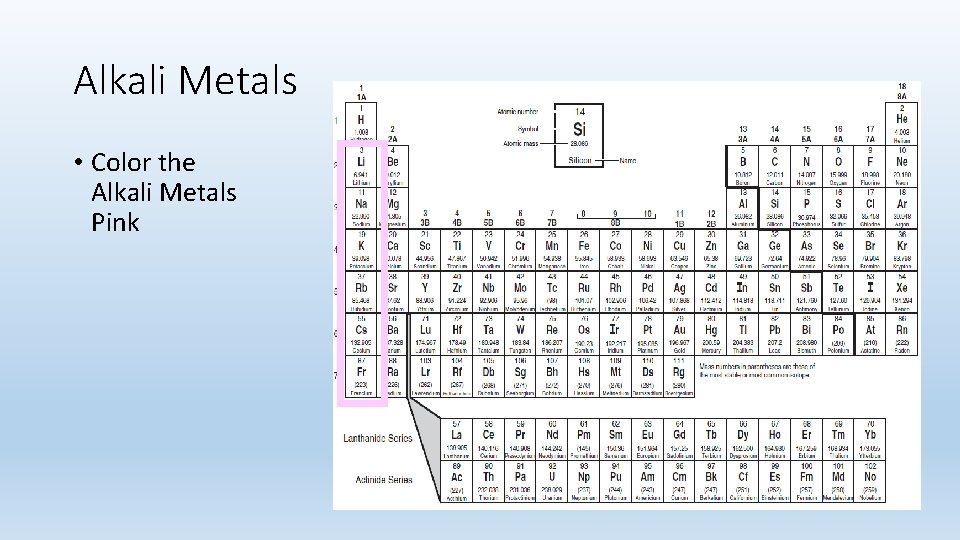
Alkali Metals • Color the Alkali Metals Pink
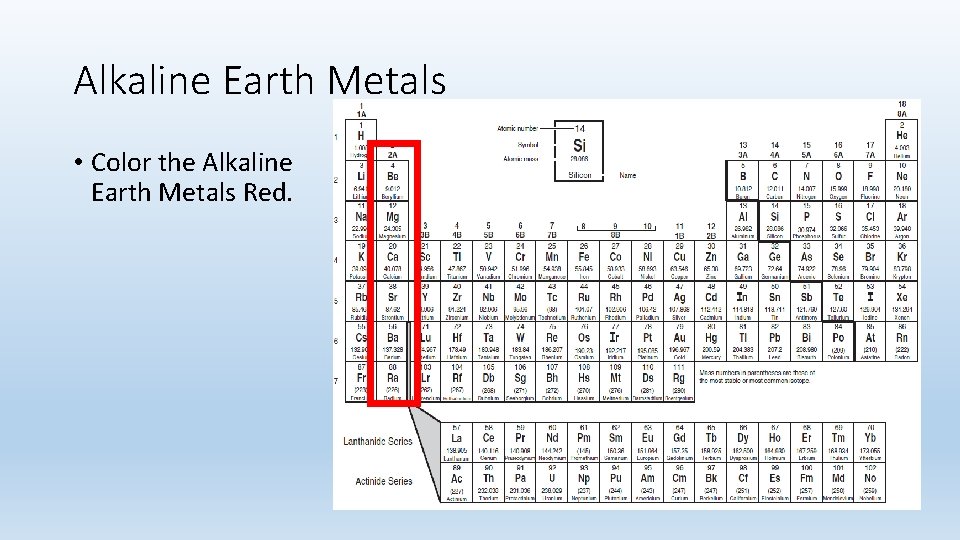
Alkaline Earth Metals • Color the Alkaline Earth Metals Red.
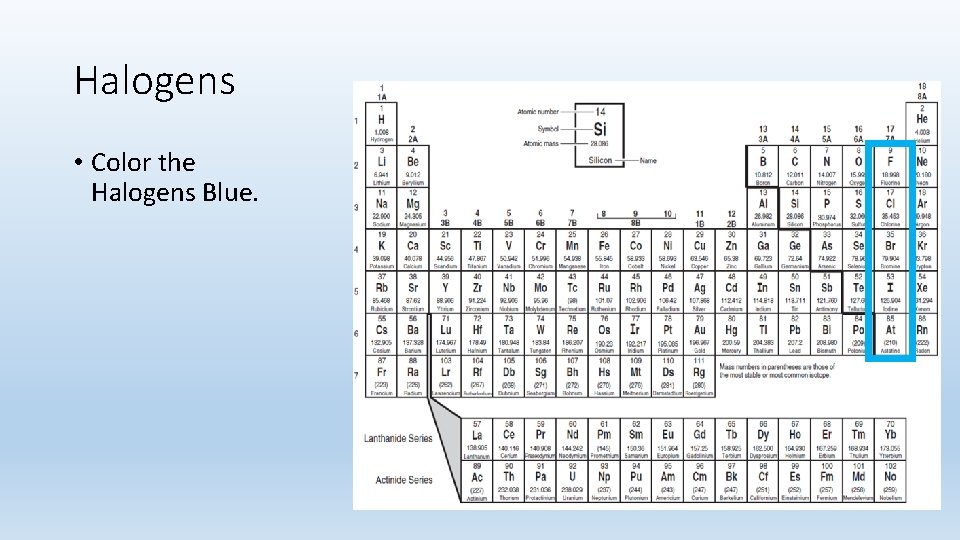
Halogens • Color the Halogens Blue.
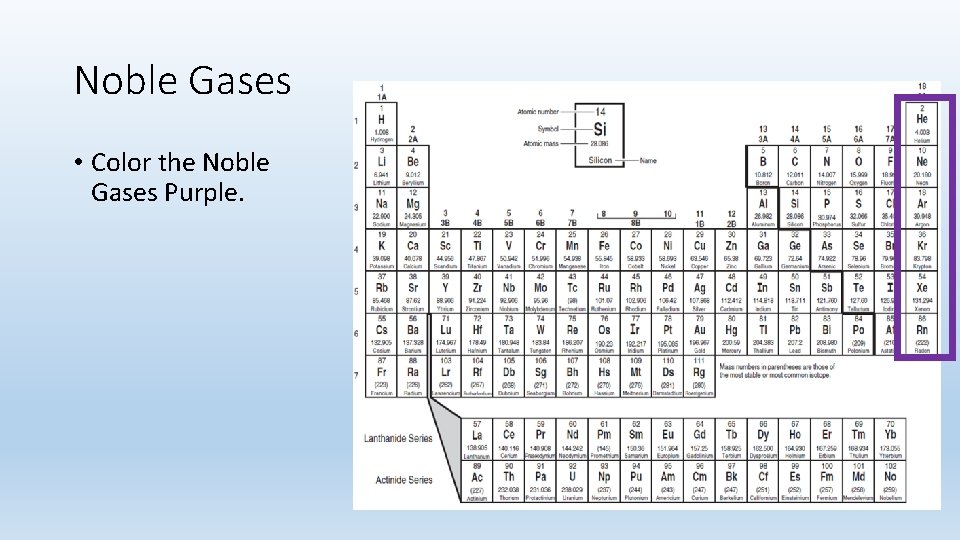
Noble Gases • Color the Noble Gases Purple.
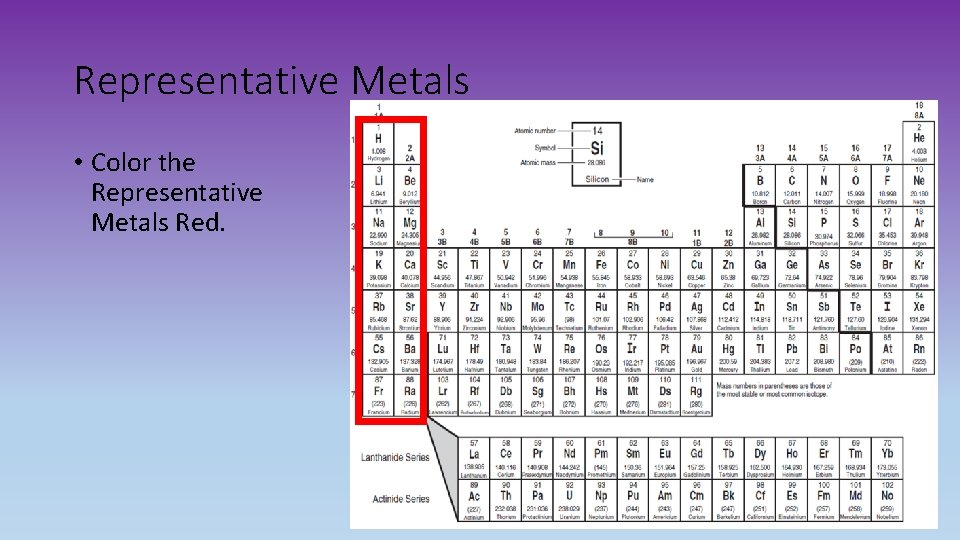
Representative Metals • Color the Representative Metals Red.
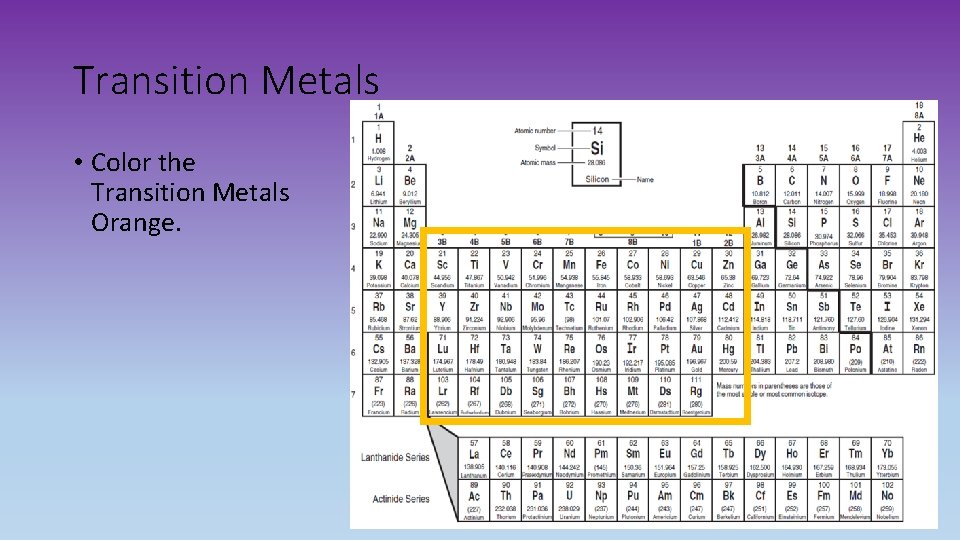
Transition Metals • Color the Transition Metals Orange.
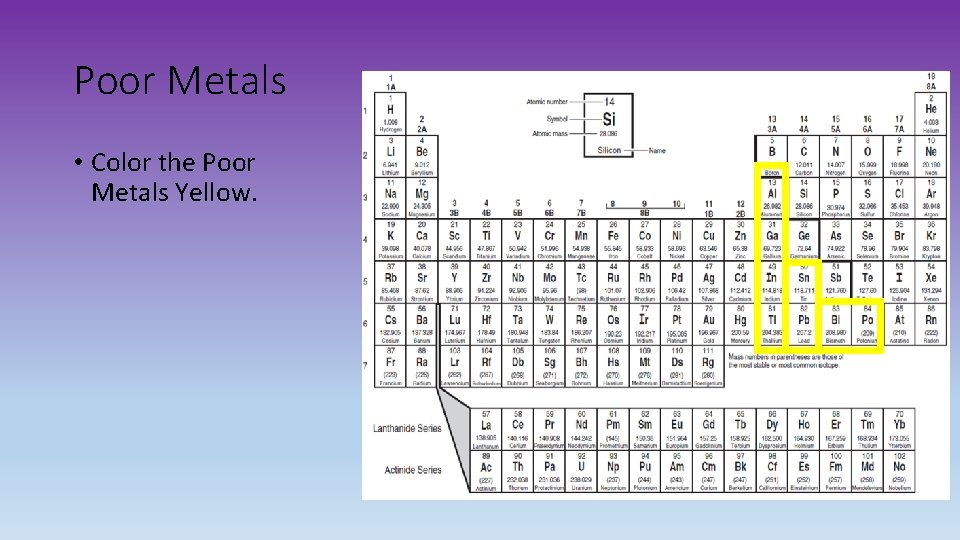
Poor Metals • Color the Poor Metals Yellow.
INNER TRANSITION METALS • Color the Inner Transition Metals Purple
Metalloids • Color the Metalloids Light/lime green.
Nonmetals • Color the Nonmetals Blue.

Alkali Metals • Group 1 A. • Most reactive of all the metals. • Shiny, malleable, ductile. • Good conductors of heat and electricity. • 1 Valence Electron.

Alkaline Earth Metals • Group 2 A. • They are never found uncombined in nature. • 2 valence electrons. • Not as reactive as Alkali Metals.

Halogens • Group 7 A • Only group with all three states of matter @ room temperature • 7 valence electrons • Very reactive; fluorine is the most reactive element and combines with most other elements • The name halogen comes from the combination of the Greek work hals, meaning “salt” and the Latin word genesis, meaning “to be born. ” • Compounds containing these are called “salts”

Noble Gases • Group 8 A • Colorless gases that are extremely un-reactive. • Inactive- because their outermost energy level is full. • 8 valence electrons • The s and p sublevels are completely filled with electrons—two electrons in the s sublevel and six electrons in the p sublevel.

Transition Metals • Good conductors of heat and electricity. • Are the Group B elements that are usually displayed in the main body of a periodic table (3 B-8 B, 1 B-2 B) • In atoms of a transition metal, the highest occupied s sublevel and a nearby d sublevel contain electrons • These elements are characterized by the presence of electrons in d orbitals. Poor Metals • Softer than transition metals. • Melting and boiling points are lower. Inner Transition Metals Lanthanides and Actinides • The 30 rare Earth elements. • Most elements are synthetic or man made. • In atoms of these elements, the highest occupied s sublevel and a nearby f sublevel generally contain electrons. • The inner transition metals are characterized by the presence of electrons in f orbitals.

Nonmetals • Poor conductors of heat and electricity. • Not ductile or malleable. • Brittle and break easily. • They are dull. • Many non-metals are gases. Hydrogen • The hydrogen square sits atop Family AI, but it is not a member of that family. Hydrogen is in a class of its own. • It’s a gas at room temperature. • It has one proton and one electron in its one and only energy level. • Hydrogen only needs 2 electrons to fill up its valence shell.

Metalloids • Have properties of both metals and non-metals. • They are solids that can be shiny or dull. • They conduct heat and electricity better than non-metals but not as well as metals. • They are ductile and malleable.

6. 2 Classifying the Elements > Electron Configurations in Groups The Representative Elements This figure shows a portion of the periodic table containing Groups 1 A through 7 A. • Elements in Groups 1 A through 7 A are often referred to as representative elements because they display a wide range of physical and chemical properties. 20

6. 2 Classifying the Elements > Electron Configurations in Groups The Representative Elements In atoms of representative elements, the s and p sublevels of the highest occupied energy level are not filled. • In atoms of these Group 1 A elements, there is only one electron in the highest occupied energy level. – The electron is in an s sublevel. Lithium (Li) 1 s 22 s 1 Sodium (Na) 1 s 22 p 63 s 1 Potassium (K) 1 s 22 p 63 s 23 p 64 s 1 21
- Slides: 21