The Periodic Table Electron Configurations Periodic Law when
The Periodic Table & Electron Configurations
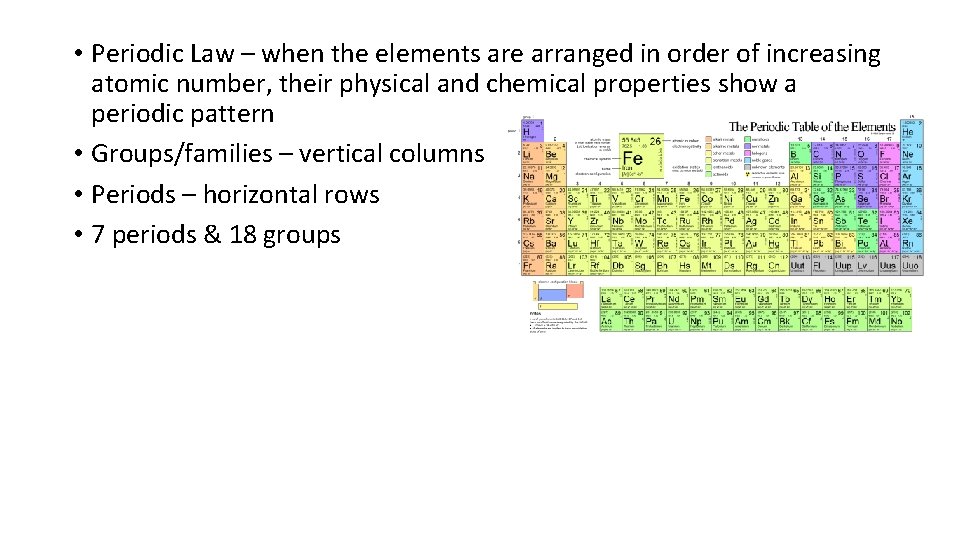
• Periodic Law – when the elements are arranged in order of increasing atomic number, their physical and chemical properties show a periodic pattern • Groups/families – vertical columns • Periods – horizontal rows • 7 periods & 18 groups
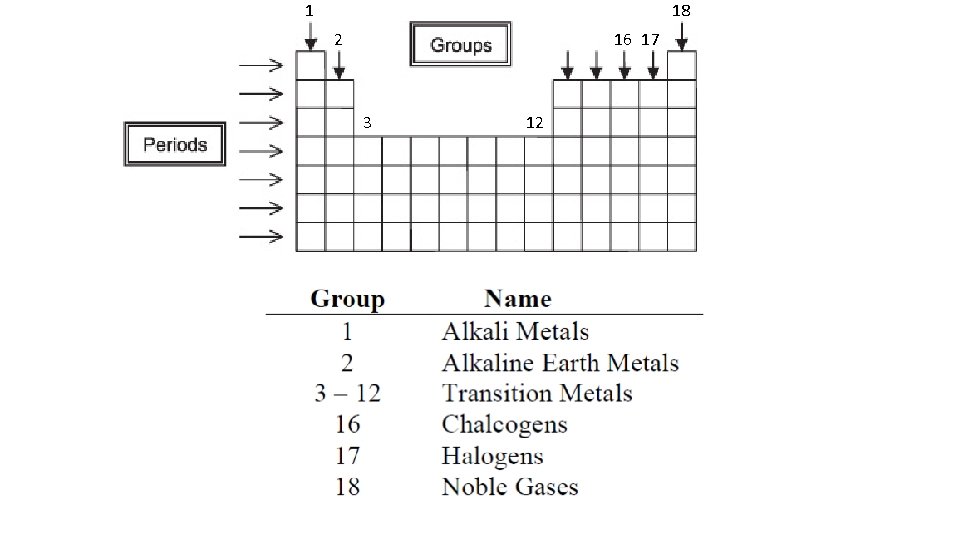
1 18 2 16 17 3 12

Metals, Nonmetals and Semimetals • Mostly solid at RT (exception Hg) • Shiny, malleable, ductile • Good conductors • Nonmetals • Often gases at RT, sometimes liquid or solid • Not shiny • Poor conductors • Metalloids (semimetals) • Properties between metals and non-metals (so identity disputed)
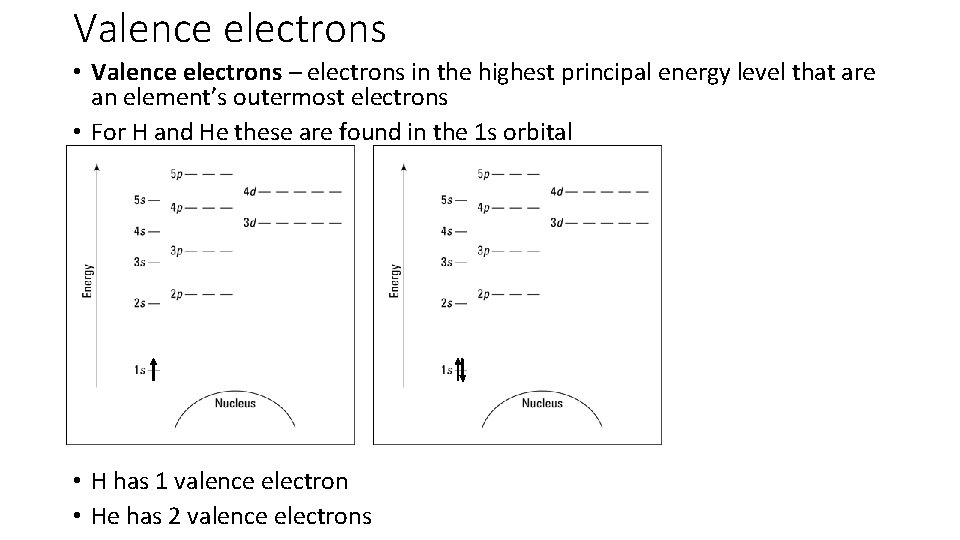
Valence electrons • Valence electrons – electrons in the highest principal energy level that are an element’s outermost electrons • For H and He these are found in the 1 s orbital • H has 1 valence electron • He has 2 valence electrons

Valence Electrons • Oxygen has 6 valence electrons in its n=2 orbitals (outermost)

Valence Electrons • Aluminum has 3 valence electrons in its n=3 orbitals (outermost)
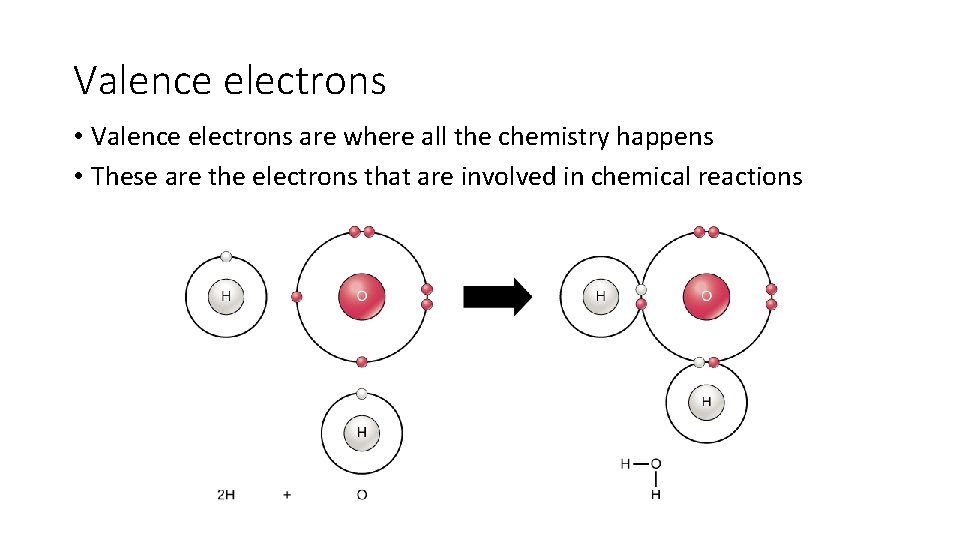
Valence electrons • Valence electrons are where all the chemistry happens • These are the electrons that are involved in chemical reactions

Electron Configuration: Noble Gas Notation • What is the electron configuration of He? • 1 s 2 • This can also be written as: [He] • This is understood to mean: 1 s 2 • Li = [He]2 s 1 • Be = [He]2 s 2 • B = [He]2 s 22 p 1 etc • Until [He] 2 s 22 p 6 = [Ne]

Write the configurations for the first 4 alkali metals using sublevel and noble gas notation Element Li Na K Rb Sublevel Notation Noble Gas Notation

Write the configurations for the first 4 alkali metals using sublevel and noble gas notation Element Li Na K Rb Sublevel Notation 1 s 22 s 1 1 s 22 s 22 p 63 s 23 p 64 s 1 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 1 Noble Gas Notation [He] 2 s 1 [Ne] 3 s 1 [Ar] 4 s 1 [Kr] 5 s 1 • What pattern do you observe? • The configuration of the valence electrons is the same for all of them. • This accounts for the similar chemical behavior of the alkali metals • This also explains group chemistry
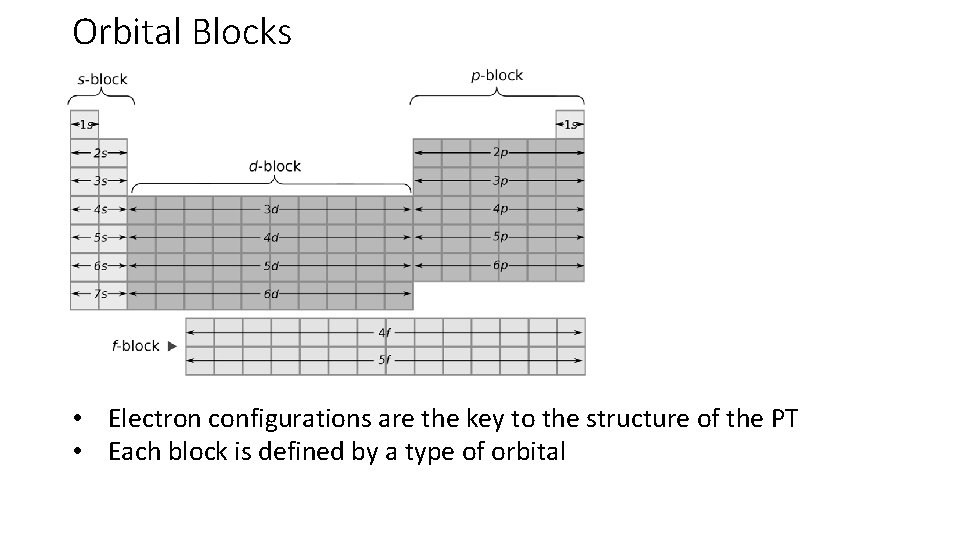
Orbital Blocks • Electron configurations are the key to the structure of the PT • Each block is defined by a type of orbital

s-Block • Alkali metals and alkaline earth metals plus hydrogen and Helium • Valence electrons only in s orbitals • Two groups b/c max of 2 electrons

p-Block • Groups 13 -18 (except He) • No p sublevel in first principal energy level • Thus no p sublevel in period 1 • 6 groups because p orbitals in a level contain a max of 6 electrons

d-Block • Groups 3 -12 • First d orbital is 3 d • Up to 10 electrons so d block is 10 elements wide

f-Block • Placed below main body of PT • 28 elements • f sublevel contains up to 14 electrons so 14 elements wide • Lanthanides insert after Lanthanum • Actinides insert after Actinium • Adding electron to the f-block does not follow the nice rules we have for elements further up the periodic table
- Slides: 16